|
Selenophene
Selenophene is an unsaturated organic compound containing a five-member ring with selenium with formula C4H4Se. It is a metallole with reduced aromatic character compared to thiophene. Nomenclature Atoms in selenophene are numbered sequentially around the ring, starting with the selenium atom as number 1 following normal systematic nomenclature rules. Oxidized forms include selenophene 1,1-dioxide. Related ring structures include those with only one double bond ( 2-selenolene and 3-selenolene) and the fully saturated structure selenolane. Production Although Ida Foa claimed to have made selenophene in 1909, the first confirmed production was by Mazza and Solazzo in 1927. They heated acetylene and selenium together at about 300 °C. The selenium burst into flame, and up to 15% selenophene was formed, along with selenonaphthene. Another way to make it is from furan heated with hydrogen selenide and aluminium at 400 °C. Substituted selenophenes can be made using a Fie ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thiophene
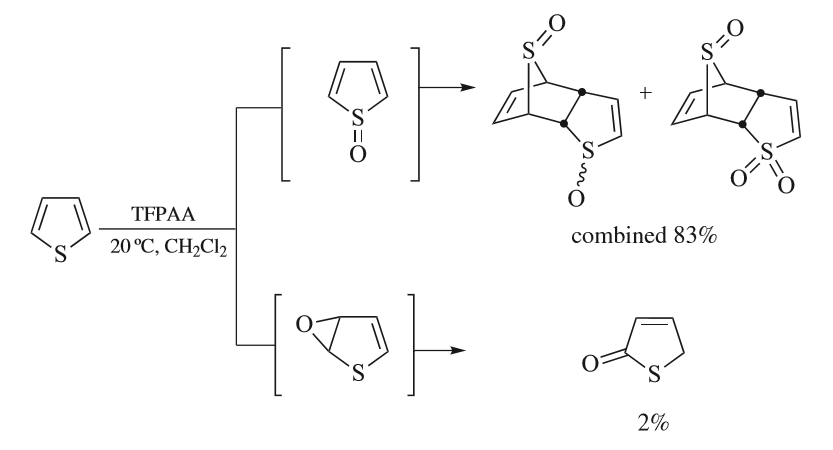
Thiophene is a heterocyclic compound with the formula C4H4S. Consisting of a planar five-membered ring, it is aromatic as indicated by its extensive substitution reactions. It is a colorless liquid with a benzene-like odor. In most of its reactions, it resembles benzene. Compounds analogous to thiophene include furan (C4H4O), selenophene (C4H4Se) and pyrrole (C4H4NH), which each vary by the heteroatom in the ring. Isolation and occurrence Thiophene was discovered as a contaminant in benzene. It was observed that isatin (an indole) forms a blue dye if it is mixed with sulfuric acid and crude benzene. The formation of the blue indophenin had long been believed to be a reaction of benzene itself. Viktor Meyer was able to isolate thiophene as the actual substance responsible for this reaction. Thiophene and especially its derivatives occur in petroleum, sometimes in concentrations up to 1–3%. The thiophenic content of oil and coal is removed via the hydrodesulfurization (HDS) ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Metallole
Metalloles are derivatives of cyclopentadiene in which the carbon atom at position 5, the saturated carbon, is replaced by a heteroatom. In contrast to its parent compound, the numbering of the metallole starts at the heteroatom. Some of these compounds are described as organometallic compounds, but in the list below quite a number of metalloids are present too. Many metalloles are fluorescent. Polymeric derivatives of pyrrole and thiophene are of interest in molecular electronics. Metalloles, which can also be viewed as structural analogs of pyrrole, include: * Arsole, a moderately-aromatic arsenic analog * Bismole, a bismuth analog * Borole, a boron analog * Furan (oxole), an oxygen analog * Gallole, a gallium analog * Germole, a germanium analog * Phosphole, a phosphorus analog * Pyrrole (azole), a nitrogen analog * Selenophene, a selenium analog * Silole, a silicon analog * Stannole, a tin analog * Stibole, an antimony analog * Tellurophene, a tellurium analog *Plumbole, a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tellurophene
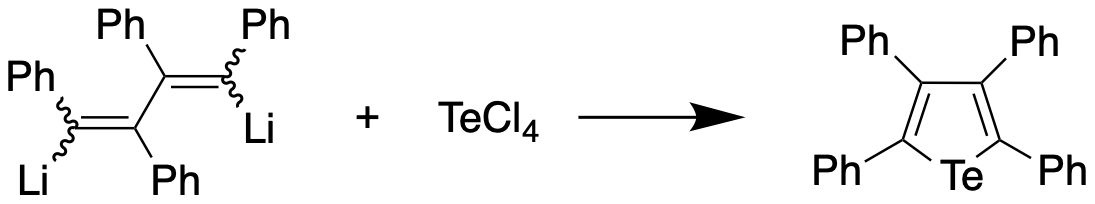
Tellurophenes are the tellurium analogue of thiophenes and selenophenes. Synthesis The first preparation of a tellurophene, tetraphenyltellurophene, was reported in 1961 by Braye et al. by reacting 1,4-dilithiotetraphenylbutadiene with tellurium tetrachloride, with the former synthesized by reaction of diphenylacetylene and lithium metal. The tellurophene, upon recrystallization from a dichloromethane/ethanol mixture, was obtained in 56% yield, and found to appear as yellow-orange crystals with a melting point of 239-239.5 °C. The same compound was obtained from 1,4-diiodotetraphenylbutadiene and lithium telluride in 82% yield. In 1966, Mack report a synthesis of an unsubstituted tellurophene through the reaction of sodium telluride with diacetylene in methanol at 20 °C. This method could be generalised to prepare 2,5-derivatives of tellurophene by selecting a suitably-substituted diacetylene precursor. The product was obtained as a pale yellow liquid with a meltin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Heterocyclic Compound
A heterocyclic compound or ring structure is a cyclic compound that has atoms of at least two different elements as members of its ring(s). Heterocyclic chemistry is the branch of organic chemistry dealing with the synthesis, properties, and applications of these heterocycles. Examples of heterocyclic compounds include all of the nucleic acids, the majority of drugs, most biomass (cellulose and related materials), and many natural and synthetic dyes. More than half of known compounds are heterocycles. 59% of US FDA-approved drugs contain nitrogen heterocycles. Classification The study of heterocyclic chemistry focuses especially on unsaturated derivatives, and the preponderance of work and applications involves unstrained 5- and 6-membered rings. Included are pyridine, thiophene, pyrrole, and furan. Another large class of heterocycles refers to those fused to benzene rings. For example, the fused benzene derivatives of pyridine, thiophene, pyrrole, and furan are quinol ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Saturated And Unsaturated Compounds
In chemistry, a saturated compound is a chemical compound (or ion) that resists the addition reactions, such as hydrogenation, oxidative addition, and binding of a Lewis acids and bases, Lewis base. The term is used in many contexts and for many classes of chemical compounds. Overall, saturated compounds are less reactive than unsaturated compounds. Saturation is derived from the Latin word ''saturare'', meaning 'to fill'. Organic chemistry Unsaturated compounds generally carry out typical addition reactions that are not possible with saturated compounds such as alkanes. A saturated organic compound has only single bonds between carbon atoms. An important class of saturated compounds are the alkanes. Many saturated compounds have functional groups, e.g., alcohols. Unsaturated organic compounds The concept of saturation can be described using various naming systems, formulas, and Analytical chemistry, analytical tests. For instance, IUPAC nomenclature of organic chemistry, IU ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electrophilic Substitution
Electrophilic substitution reactions are chemical reactions in which an electrophile displaces a functional group in a compound, which is typically, but not always, aromatic. Aromatic substitution reactions are characteristic of aromatic compounds and are common ways of introducing functional groups into benzene rings. Some aliphatic compounds can undergo electrophilic substitution as well. Electrophilic aromatic substitution In electrophilic substitution in aromatic compounds, an atom appended to the aromatic ring, usually hydrogen, is replaced by an electrophile. The most important reactions of this type that take place are aromatic nitration, aromatic halogenation, aromatic sulfonation and acylation and alkylating Friedel-Crafts reactions. It further consists of alkylation and acylation. Electrophilic aliphatic substitution In electrophilic substitution in aliphatic compounds, an electrophile displaces a functional group. This reaction is similar to nucleophilic aliphati ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ethyl Bromoacetate
Ethyl bromoacetate is the chemical compound with the formula CH2BrCO2C2H5. It is the ethyl ester of bromoacetic acid and is prepared in two steps from acetic acid. It is a lachrymator and has a fruity, pungent odor. It is also a highly toxic alkylating agent and may be fatal if inhaled. Applications Ethyl bromoacetate is listed by the World Health Organization as a riot control agent, and was first employed for that purpose by French police in 1912. The French army used rifle grenades 'grenades lacrymogènes' filled with this gas against the Germans beginning in August 1914, but the weapons were largely ineffective, even though ethyl bromoacetate is twice as toxic as chlorine. In the early months of the war the British also used the weaponized use of tear gas agents and more toxic gasses including sulfur dioxide. The German army then used these attacks to justify their subsequent employment of it as odorant or warning agent in odorless, toxic gases and chemical weapons in 1915 und ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Selenide
Sodium selenide is an inorganic compound of sodium and selenium with the chemical formula Na2Se. Preparation This colourless solid is prepared by the reaction of selenium with a solution of sodium in liquid ammonia at −40 °C.Brauer, G. ed. (1963) ''Handbook of Preparative Inorganic Chemistry'', 2nd Ed., Academic Press, NY., Vol. 1. p. 421. Alternatively, sodium selenide can be prepared by the reaction of gaseous hydrogen selenide with metallic sodium at 100 °C. Reactions Like other alkali metal chalcogenides, this material is highly sensitive to water, easily undergoing hydrolysis to give mixtures of sodium biselenide (NaSeH) and hydroxide. This hydrolysis occurs because of the extreme basicity of the Se2− ion. :Na2Se + H2O → NaHSe + NaOH Similarly, sodium selenide is readily oxidized to polyselenides, a conversion signaled by off-white samples. Sodium selenide reacts with acids to produce toxic hydrogen selenide gas. :Na2Se + 2 HCl → H2Se + 2 NaCl The compound r ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen Selenide
Hydrogen selenide is an inorganic compound with the formula H2Se. This hydrogen chalcogenide is the simplest and most commonly encountered hydride of selenium. H2Se is a colorless, flammable gas under standard conditions. It is the most toxic selenium compoundhttp://www.epa.gov/ttnatw01/hlthef/selenium.html, US Environmental Protection Agency, Air Toxins website with an exposure limit of 0.05 ppm over an 8-hour period.https://www.cdc.gov/niosh/idlh/7783075.html, Documentation of Immediately Dangerous to Life or Health Concentrations: Hydrogen Selenide, The National Institute for Occupational Safety and Healthhttps://www.cdc.gov/niosh/docs/81-123/pdfs/0336.pdf Occupational Health Guideline for Hydrogen Selenide, The National Institute for Occupational Safety and Health, 1978 Even at extremely low concentrations, this compound has a very irritating smell resembling that of decayed horseradish or 'leaking gas', but smells of rotten eggs at higher concentrations. Structure and propert ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acetylene
Acetylene (systematic name: ethyne) is the chemical compound with the formula and structure . It is a hydrocarbon and the simplest alkyne. This colorless gas is widely used as a fuel and a chemical building block. It is unstable in its pure form and thus is usually handled as a solution. Pure acetylene is odorless, but commercial grades usually have a marked odor due to impurities such as divinyl sulfide and phosphine.Compressed Gas Association (1995Material Safety and Data Sheet – Acetylene As an alkyne, acetylene is unsaturated because its two carbon atoms are bonded together in a triple bond. The carbon–carbon triple bond places all four atoms in the same straight line, with CCH bond angles of 180°. Discovery Acetylene was discovered in 1836 by Edmund Davy, who identified it as a "new carburet of hydrogen". It was an accidental discovery while attempting to isolate potassium metal. By heating potassium carbonate with carbon at very high temperatures, he produced a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
International Union Of Pure And Applied Chemistry
The International Union of Pure and Applied Chemistry (IUPAC ) is an international federation of National Adhering Organizations working for the advancement of the chemical sciences, especially by developing nomenclature and terminology. It is a member of the International Science Council (ISC). IUPAC is registered in Zürich, Switzerland, and the administrative office, known as the "IUPAC Secretariat", is in Research Triangle Park, North Carolina, United States. This administrative office is headed by IUPAC's executive director, currently Lynn Soby. IUPAC was established in 1919 as the successor of the International Congress of Applied Chemistry for the advancement of chemistry. Its members, the National Adhering Organizations, can be national chemistry societies, national academies of sciences, or other bodies representing chemists. There are fifty-four National Adhering Organizations and three Associate National Adhering Organizations. IUPAC's Inter-divisional Committee on ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |