|
SNAP25
Synaptosomal-Associated Protein, 25kDa (SNAP-25) is a Target Soluble NSF (''N''-ethylmaleimide-sensitive factor) Attachment Protein Receptor (t-SNARE) protein encoded by the ''SNAP25'' gene found on chromosome 20p12.2 in humans. SNAP-25 is a component of the ''trans''-SNARE complex, which accounts for membrane fusion specificity and directly executes fusion by forming a tight complex that brings the synaptic vesicle and plasma membranes together. Structure and function SNAP-25, a Q-SNARE protein, is anchored to the cytosolic face of membranes via palmitoyl side chains covalently bound to cysteine amino acid residues in the central linker domain of the molecule. This means that SNAP-25 does not contain a trans-membrane domain. SNAP-25 has been identified to contribute two α-helices to the SNARE complex, a four-α-helix domain complex. The SNARE complex participates in vesicle fusion, which involves the docking, priming and merging of a vesicle with the cell membrane to in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Q-SNARE
SNARE proteins – " SNAP REceptor" – are a large protein family consisting of at least 24 members in yeasts, more than 60 members in mammalian cells, and some numbers in plants. The primary role of SNARE proteins is to mediate vesicle fusion – the fusion of vesicles with the target membrane; this notably mediates exocytosis, but can also mediate the fusion of vesicles with membrane-bound compartments (such as a lysosome). The best studied SNAREs are those that mediate the neurotransmitter release of synaptic vesicles in neurons. These neuronal SNAREs are the targets of the neurotoxins responsible for botulism and tetanus produced by certain bacteria. Types SNAREs can be divided into two categories: ''vesicle'' or ''v-SNAREs'', which are incorporated into the membranes of transport vesicles during budding, and ''target'' or ''t-SNAREs'', which are associated with nerve terminal membranes. Evidence suggests that t-SNAREs form stable subcomplexes which serve as guides f ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
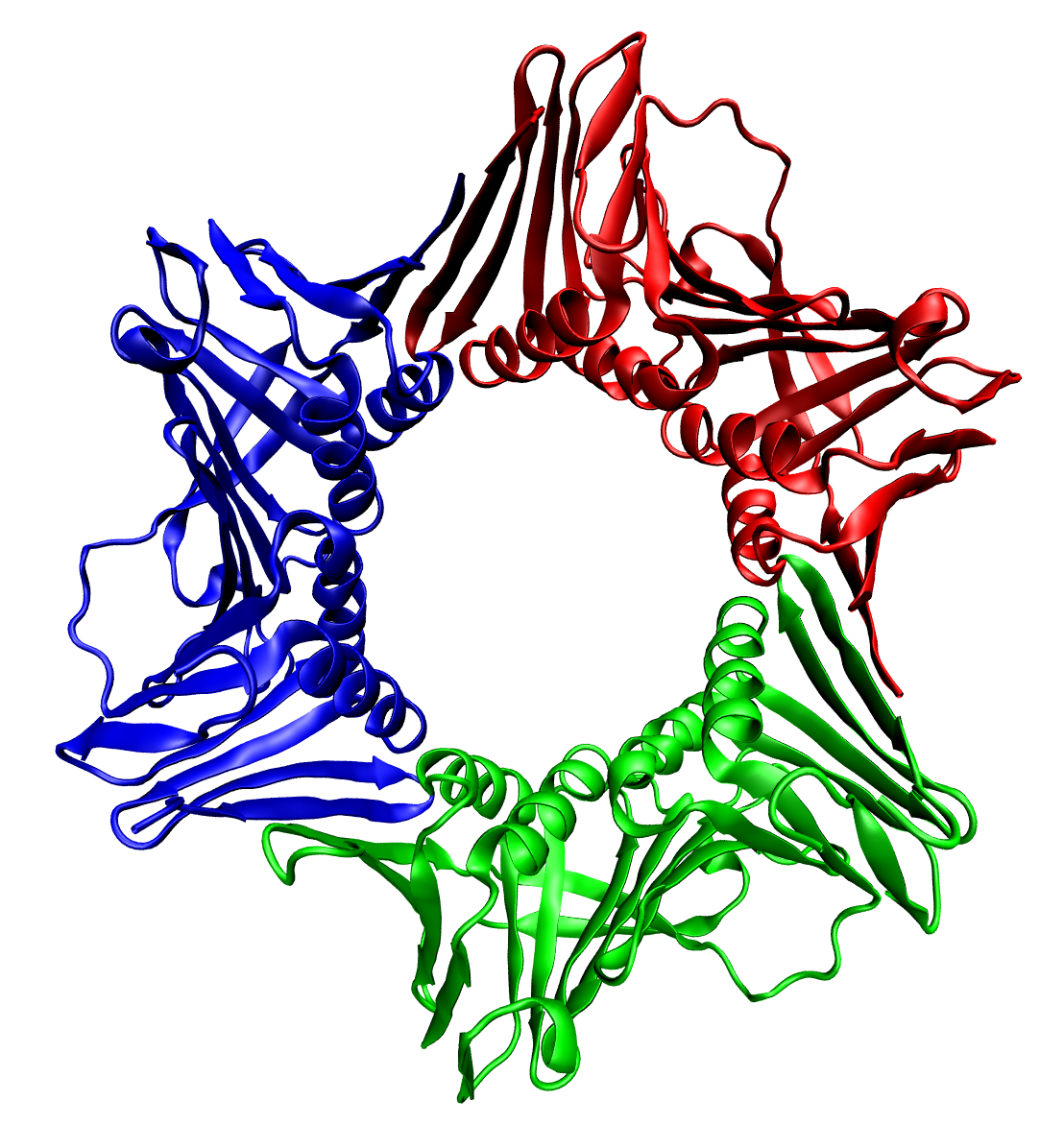
SNARE (protein)
SNARE proteins – " SNAP REceptor" – are a large protein family consisting of at least 24 members in yeasts, more than 60 members in mammalian cells, and some numbers in plants. The primary role of SNARE proteins is to mediate vesicle fusion – the fusion of vesicles with the target membrane; this notably mediates exocytosis, but can also mediate the fusion of vesicles with membrane-bound compartments (such as a lysosome). The best studied SNAREs are those that mediate the neurotransmitter release of synaptic vesicles in neurons. These neuronal SNAREs are the targets of the neurotoxins responsible for botulism and tetanus produced by certain bacteria. Types SNAREs can be divided into two categories: ''vesicle'' or ''v-SNAREs'', which are incorporated into the membranes of transport vesicles during budding, and ''target'' or ''t-SNAREs'', which are associated with nerve terminal membranes. Evidence suggests that t-SNAREs form stable subcomplexes which serve as guides f ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Palmitoyl Chain
Palmitoylation is the covalent attachment of fatty acids, such as palmitic acid, to cysteine (''S''-palmitoylation) and less frequently to serine and threonine (''O''-palmitoylation) residues of proteins, which are typically membrane proteins. The precise function of palmitoylation depends on the particular protein being considered. Palmitoylation enhances the hydrophobicity of proteins and contributes to their membrane association. Palmitoylation also appears to play a significant role in subcellular trafficking of proteins between membrane compartments, as well as in modulating protein–protein interactions. In contrast to prenylation and myristoylation, palmitoylation is usually reversible (because the bond between palmitic acid and protein is often a thioester bond). The reverse reaction in mammalian cells is catalyzed by acyl-protein thioesterases (APTs) in the cytosol and palmitoyl protein thioesterases in lysosomes. Because palmitoylation is a dynamic, post-translatio ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Exocytosis
Exocytosis () is a form of active transport and bulk transport in which a cell transports molecules (e.g., neurotransmitters and proteins) out of the cell ('' exo-'' + ''cytosis''). As an active transport mechanism, exocytosis requires the use of energy to transport material. Exocytosis and its counterpart, endocytosis, are used by all cells because most chemical substances important to them are large polar molecules that cannot pass through the hydrophobic portion of the cell membrane by passive means. Exocytosis is the process by which a large amount of molecules are released; thus it is a form of bulk transport. Exocytosis occurs via secretory portals at the cell plasma membrane called porosomes. Porosomes are permanent cup-shaped lipoprotein structure at the cell plasma membrane, where secretory vesicles transiently dock and fuse to release intra-vesicular contents from the cell. In exocytosis, membrane-bound secretory vesicles are carried to the cell membrane, where they ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Vesicle-associated Membrane Protein
Vesicle associated membrane proteins (VAMP) are a family of SNARE proteins with similar structure, and are mostly involved in vesicle fusion. * VAMP1 and VAMP2 proteins known as synaptobrevins are expressed in brain and are constituents of the synaptic vesicles, where they participate in neurotransmitter release. * VAMP3 (known as cellubrevin) is ubiquitously expressed and participates in regulated and constitutive exocytosis as a constituent of secretory granules and secretory vesicles. * VAMP5 and VAMP7 ( SYBL1) participate in constitutive exocytosis. ** VAMP5 is a constituent of secretory vesicles, myotubes and tubulovesicular structures. ** VAMP7 is found both in secretory granules and endosomes. * VAMP8 (known as endobrevin) participates in endocytosis and is found in early endosomes. VAMP8 also participates the regulated exocytosis in pancreatic acinar cells. *VAMP4 Vesicle-associated membrane protein 4 is a protein that in humans is encoded by the ''VAMP4'' gene. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Syntaxin
Syntaxins are a family of membrane integrated Q-SNARE proteins participating in exocytosis. Domains Syntaxins possess a single C-terminal transmembrane domain, a SNARE domain (known as H3), and an N-terminal regulatory domain (Habc). Syntaxin 17 may have two transmembrane domains. * The SNARE (H3) domain binds to both synaptobrevin and SNAP-25 forming the core SNARE complex. Formation of this stable SNARE core complex is believed to generate the free energy required to initiate fusion between the vesicle membrane and plasma membrane. * The N-terminal Habc domain is formed by 3 α-helices and when collapsed onto its own H3 helix forms an inactive "closed" syntaxin conformation. This closed conformation of syntaxin is believed to be stabilized by binding of Munc-18 (nSec1), although more recent data suggests that nSec1 may bind to other conformations of syntaxin, as well. The "open" syntaxin conformation is the conformation that is competent to form into SNARE core complexes. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Coiled Coil
A coiled coil is a structural motif in proteins in which 2–7 alpha-helices are coiled together like the strands of a rope. (Dimers and trimers are the most common types.) Many coiled coil-type proteins are involved in important biological functions, such as the regulation of gene expression — e.g., transcription factors. Notable examples are the oncoproteins c-Fos and c-Jun, as well as the muscle protein tropomyosin. Discovery The possibility of coiled coils for α-keratin was initially somewhat controversial. Linus Pauling and Francis Crick independently came to the conclusion that this was possible at about the same time. In the summer of 1952, Pauling visited the laboratory in England where Crick worked. Pauling and Crick met and spoke about various topics; at one point, Crick asked whether Pauling had considered "coiled coils" (Crick came up with the term), to which Pauling said he had. Upon returning to the United States, Pauling resumed research on the topic. He conc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Protein Quaternary Structure
Protein quaternary structure is the fourth (and highest) classification level of protein structure. Protein quaternary structure refers to the structure of proteins which are themselves composed of two or more smaller protein chains (also referred to as subunits). Protein quaternary structure describes the number and arrangement of multiple protein folding, folded protein subunits in a Multiprotein complex, multi-subunit complex. It includes organizations from simple protein dimer, dimers to large homooligomers and multiprotein complex, complexes with defined or variable numbers of subunits. In contrast to the first three levels of protein structure, not all proteins will have a quaternary structure since some proteins function as single units. Protein quaternary structure can also refer to biomolecular complexes of proteins with nucleic acids and other Cofactor (biochemistry), cofactors. Description and examples Many proteins are actually assemblies of multiple polypeptide c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
C-terminus
The C-terminus (also known as the carboxyl-terminus, carboxy-terminus, C-terminal tail, C-terminal end, or COOH-terminus) is the end of an amino acid chain (protein or polypeptide), terminated by a free carboxyl group (-COOH). When the protein is translated from messenger RNA, it is created from N-terminus to C-terminus. The convention for writing peptide sequences is to put the C-terminal end on the right and write the sequence from N- to C-terminus. Chemistry Each amino acid has a carboxyl group and an amine group. Amino acids link to one another to form a chain by a dehydration reaction which joins the amine group of one amino acid to the carboxyl group of the next. Thus polypeptide chains have an end with an unbound carboxyl group, the C-terminus, and an end with an unbound amine group, the N-terminus. Proteins are naturally synthesized starting from the N-terminus and ending at the C-terminus. Function C-terminal retention signals While the N-terminus of a protein often c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Synapse
Chemical synapses are biological junctions through which neurons' signals can be sent to each other and to non-neuronal cells such as those in muscles or glands. Chemical synapses allow neurons to form circuits within the central nervous system. They are crucial to the biological computations that underlie perception and thought. They allow the nervous system to connect to and control other systems of the body. At a chemical synapse, one neuron releases neurotransmitter molecules into a small space (the synaptic cleft) that is adjacent to another neuron. The neurotransmitters are contained within small sacs called synaptic vesicles, and are released into the synaptic cleft by exocytosis. These molecules then bind to neurotransmitter receptors on the postsynaptic cell. Finally, the neurotransmitters are cleared from the synapse through one of several potential mechanisms including enzymatic degradation or re-uptake by specific transporters either on the presynaptic cell o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
N-terminus
The N-terminus (also known as the amino-terminus, NH2-terminus, N-terminal end or amine-terminus) is the start of a protein or polypeptide, referring to the free amine group (-NH2) located at the end of a polypeptide. Within a peptide, the amine group is bonded to the carboxylic group of another amino acid, making it a chain. That leaves a free carboxylic group at one end of the peptide, called the C-terminus, and a free amine group on the other end called the N-terminus. By convention, peptide sequences are written N-terminus to C-terminus, left to right (in LTR writing systems). This correlates the translation direction to the text direction, because when a protein is translated from messenger RNA, it is created from the N-terminus to the C-terminus, as amino acids are added to the carboxyl end of the protein. Chemistry Each amino acid has an amine group and a carboxylic group. Amino acids link to one another by peptide bonds which form through a dehydration reaction that ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
P-type Calcium Channel
The P-type calcium channel is a type of voltage-dependent calcium channel. Similar to many other high-voltage-gated calcium channels, the α1 subunit determines most of the channel's properties. The 'P' signifies cerebellar Purkinje cells, referring to the channel's initial site of discovery. P-type calcium channels play a similar role to the N-type calcium channel in neurotransmitter release at the presynaptic terminal and in neuronal integration in many neuronal types. History The calcium channel experiments that led to the discovery of P-type calcium channels were initially completed by Rodolfo Llinas, Llinás and Sugimori in 1980. P type calcium channels were named in 1989 because they were discovered within mammalian Purkinje neurons. They were able to use an ''in vitro'' preparation to examine the ionic currents that account for Purkinje cells' electrophysiology, electrophysiological properties. They found that there are calcium dependent action potentials which rise slowly a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |