|
S-methyl-L-methionine
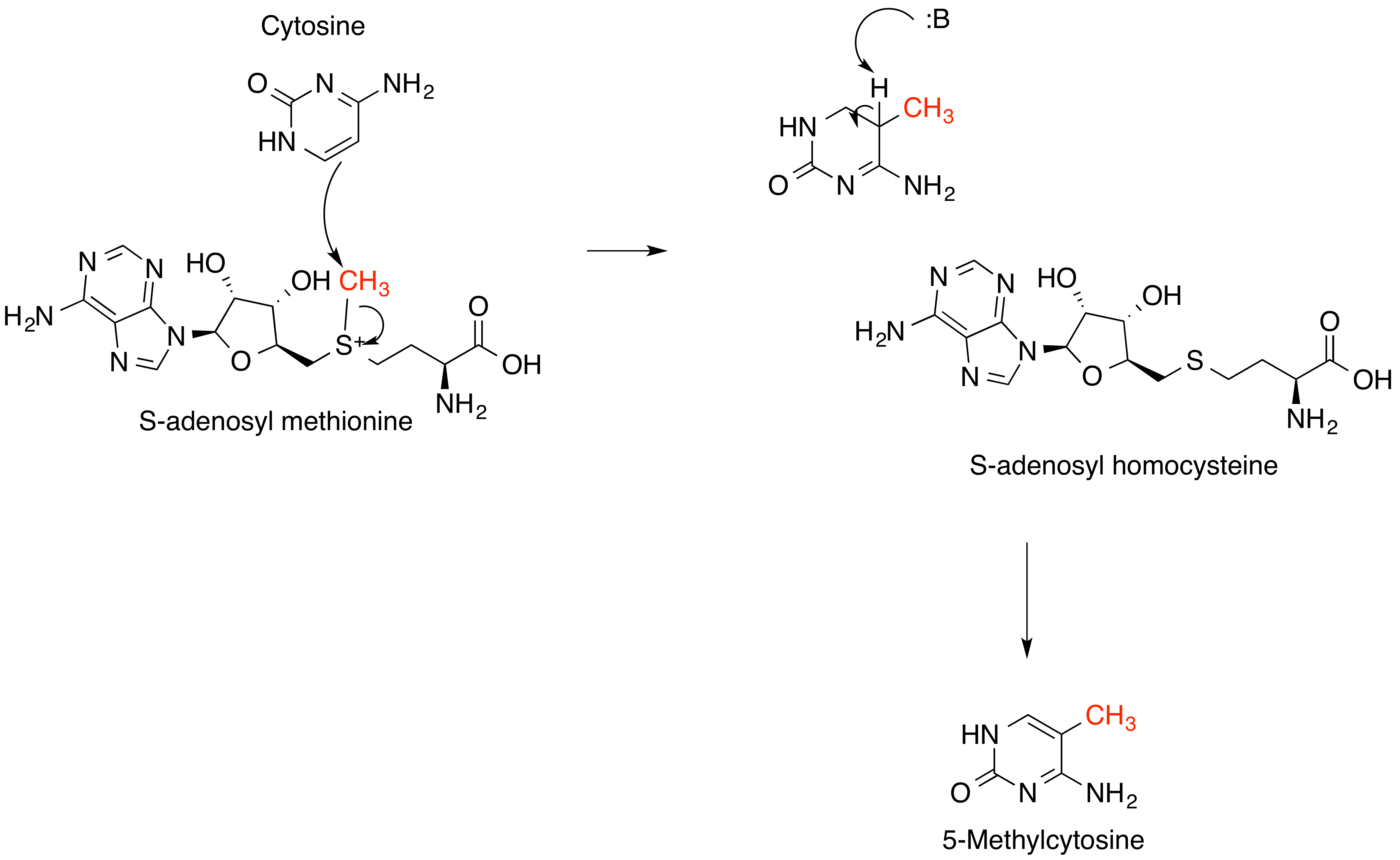
''S''-Methylmethionine (SMM) is a derivative of methionine with the chemical formula ( CH3)2S+CH2CH2CH(NH3+)CO2−. This cation is a naturally-occurring intermediate in many biosynthetic pathways owing to the sulfonium functional group. It is biosynthesized from L-methionine which is first converted to ''S''-adenosylmethionine. The subsequent conversion, involving replacement of the adenosyl group by a methyl group is catalyzed by the enzyme methionine ''S''-methyltransferase. ''S''-methylmethionine is particularly abundant in plants, being more abundant than methionine. ''S''-Methylmethionine is sometimes referred to as ''vitamin U'', but it is not considered a true vitamin. The term was coined in 1950 by Garnett Cheney for uncharacterized anti-ulcerogenic factors in raw cabbage juice that may help speed healing of peptic ulcers. Biosynthesis and biochemical function ''S''-Methylmethionine arises via the methylation of methionine by ''S''-adenosyl methionine (SAM). The copr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methionine S-methyltransferase
In enzymology, a methionine S-methyltransferase () is an enzyme that catalyzes the chemical reaction :S-adenosyl-L-methionine + L-methionine \rightleftharpoons S-adenosyl-L-homocysteine + S-methyl-L-methionine Thus, the two substrates of this enzyme are S-adenosyl methionine and L-methionine, whereas its two products are S-adenosylhomocysteine and S-methyl-L-methionine. This enzyme belongs to the family of transferases, specifically those transferring one-carbon group methyltransferases. The systematic name of this enzyme class is S-adenosyl-L-methionine:L-methionine S-methyltransferase. Other names in common use include S-adenosyl methionine:methionine methyl transferase, methionine methyltransferase, S-adenosylmethionine transmethylase, and S-adenosylmethionine-methionine methyltransferase. This enzyme participates in selenoamino acid metabolism. It has 2 cofactors: manganese, and zinc Zinc is a chemical element with the symbol Zn and atomic number 30. Zinc is a s ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Merck Index
''The Merck Index'' is an encyclopedia of chemical substance, chemicals, pharmaceutical drug, drugs and biomolecule, biologicals with over 10,000 monographs, monograph on single substances or groups of related chemical compound, compounds published online by the Royal Society of Chemistry. History The first edition of the Merck's Index was published in 1889 by the German chemical company Merck Group, Emanuel Merck and was primarily used as a sales catalog for Merck's growing list of chemicals it sold. The American subsidiary was established two years later and continued to publish it. During World War I the US government seized Merck's US operations and made it a separate American "Merck" company that continued to publish the Merck Index. In 2012 the Merck Index was licensed to the Royal Society of Chemistry. An online version of The Merck Index, including historic records and new updates not in the print edition, is commonly available through research libraries. It also include ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methylation
In the chemical sciences, methylation denotes the addition of a methyl group on a substrate, or the substitution of an atom (or group) by a methyl group. Methylation is a form of alkylation, with a methyl group replacing a hydrogen atom. These terms are commonly used in chemistry, biochemistry, soil science, and the biological sciences. In biological systems, methylation is catalyzed by enzymes; such methylation can be involved in modification of heavy metals, regulation of gene expression, regulation of protein function, and RNA processing. In vitro methylation of tissue samples is also one method for reducing certain histological staining artifacts. The reverse of methylation is demethylation. In biology In biological systems, methylation is accomplished by enzymes. Methylation can modify heavy metals, regulate gene expression, RNA processing and protein function. It has been recognized as a key process underlying epigenetics. Methanogenesis Methanogenesis, the process th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfur Ions
Sulfur (or sulphur in British English) is a chemical element with the symbol S and atomic number 16. It is abundant, multivalent and nonmetallic. Under normal conditions, sulfur atoms form cyclic octatomic molecules with a chemical formula S8. Elemental sulfur is a bright yellow, crystalline solid at room temperature. Sulfur is the tenth most abundant element by mass in the universe and the fifth most on Earth. Though sometimes found in pure, native form, sulfur on Earth usually occurs as sulfide and sulfate minerals. Being abundant in native form, sulfur was known in ancient times, being mentioned for its uses in ancient India, ancient Greece, China, and ancient Egypt. Historically and in literature sulfur is also called brimstone, which means "burning stone". Today, almost all elemental sulfur is produced as a byproduct of removing sulfur-containing contaminants from natural gas and petroleum.. Downloahere The greatest commercial use of the element is the production of su ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfur Amino Acids
Sulfur (or sulphur in British English) is a chemical element with the symbol S and atomic number 16. It is abundant, multivalent and nonmetallic. Under normal conditions, sulfur atoms form cyclic octatomic molecules with a chemical formula S8. Elemental sulfur is a bright yellow, crystalline solid at room temperature. Sulfur is the tenth most abundant element by mass in the universe and the fifth most on Earth. Though sometimes found in pure, native form, sulfur on Earth usually occurs as sulfide and sulfate minerals. Being abundant in native form, sulfur was known in ancient times, being mentioned for its uses in ancient India, ancient Greece, China, and ancient Egypt. Historically and in literature sulfur is also called brimstone, which means "burning stone". Today, almost all elemental sulfur is produced as a byproduct of removing sulfur-containing contaminants from natural gas and petroleum.. Downloahere The greatest commercial use of the element is the production of su ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Amino Acids
Amino acids are organic compounds that contain both amino and carboxylic acid functional groups. Although hundreds of amino acids exist in nature, by far the most important are the alpha-amino acids, which comprise proteins. Only 22 alpha amino acids appear in the genetic code. Amino acids can be classified according to the locations of the core structural functional groups, as Alpha and beta carbon, alpha- , beta- , gamma- or delta- amino acids; other categories relate to Chemical polarity, polarity, ionization, and side chain group type (aliphatic, Open-chain compound, acyclic, aromatic, containing hydroxyl or sulfur, etc.). In the form of proteins, amino acid '' residues'' form the second-largest component (water being the largest) of human muscles and other tissues. Beyond their role as residues in proteins, amino acids participate in a number of processes such as neurotransmitter transport and biosynthesis. It is thought that they played a key role in enabling lif ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dimethyl Sulfide
Dimethyl sulfide (DMS) or methylthiomethane is an organosulfur compound with the formula (CH3)2S. Dimethyl sulfide is a flammable liquid that boils at and has a characteristic disagreeable odor. It is a component of the smell produced from cooking of certain vegetables, notably maize, cabbage, beetroot, and seafoods. It is also an indication of bacterial contamination in malt production and brewing. It is a breakdown product of dimethylsulfoniopropionate (DMSP), and is also produced by the bacterial metabolism of methanethiol. Occurrence and production DMS originates primarily from DMSP, a major secondary metabolite in some marine algae. DMS is the most abundant biological sulfur compound emitted to the atmosphere. Emission occurs over the oceans by phytoplankton. DMS is also produced naturally by bacterial transformation of dimethyl sulfoxide (DMSO) waste that is disposed of into sewers, where it can cause environmental odor problems. DMS is oxidized in the marine atmos ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dimethylsulfoniopropionate
Dimethylsulfoniopropionate (DMSP), is an organosulfur compound with the formula (CH3)2S+CH2CH2COO−. This zwitterionic metabolite can be found in marine phytoplankton, seaweeds, and some species of terrestrial and aquatic vascular plants. It functions as an osmolyte as well as several other physiological and environmental roles have also been identified. DMSP was first identified in the marine red alga '' Polysiphonia fastigiata''. Biosynthesis In higher plants, DMSP is biosynthesized from ''S''-methylmethionine. Two intermediates in this conversion are dimethylsulfoniumpropylamine and dimethylsulfoniumpropionaldehyde. In algae, however, the biosynthesis starts with the replacement of the amino group in methionine by hydroxide. Degradation DMSP is broken down by marine microbes to form two major volatile sulfur products, each with distinct effects on the environment. One of its breakdown products is methanethiol (CH3SH), which is assimilated by bacteria into protein su ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Osmolyte
Osmolytes are low-molecular weight organic compounds that influence the properties of biological fluids. Their primary role is to maintain the integrity of cells by affecting the viscosity, melting point, and ionic strength of the aqueous solution. When a cell swells due to external osmotic pressure, membrane channels open and allow efflux of osmolytes which carry water with them, restoring normal cell volume. Osmolytes also interact with the constituents of the cell, e.g. they influence protein folding. Common osmolytes include amino acids, sugars and polyols, methylamines, methylsulfonium compounds, and urea. Case studies Natural osmolytes that can act as osmoprotectants include trimethylamine ''N''-oxide (TMAO), dimethylsulfoniopropionate, sarcosine, betaine, glycerophosphorylcholine, myo-inositol, taurine, glycine, and others. Remarkably, TMAO has the capacity to restore glucocorticoid binding to mutant receptors. Bacteria accumulate osmolytes for protection against a high o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
S-Adenosyl Methionine
''S''-Adenosyl methionine (SAM), also known under the commercial names of SAMe, SAM-e, or AdoMet, is a common cosubstrate involved in methyl group transfers, transsulfuration, and aminopropylation. Although these anabolic reactions occur throughout the body, most SAM is produced and consumed in the liver. More than 40 methyl transfers from SAM are known, to various substrates such as nucleic acids, proteins, lipids and secondary metabolites. It is made from adenosine triphosphate (ATP) and methionine by methionine adenosyltransferase. SAM was first discovered by Giulio Cantoni in 1952. In bacteria, SAM is bound by the SAM riboswitch, which regulates genes involved in methionine or cysteine biosynthesis. In eukaryotic cells, SAM serves as a regulator of a variety of processes including DNA, tRNA, and rRNA methylation; immune response; amino acid metabolism; transsulfuration; and more. In plants, SAM is crucial to the biosynthesis of ethylene, an important plant hormone and sig ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
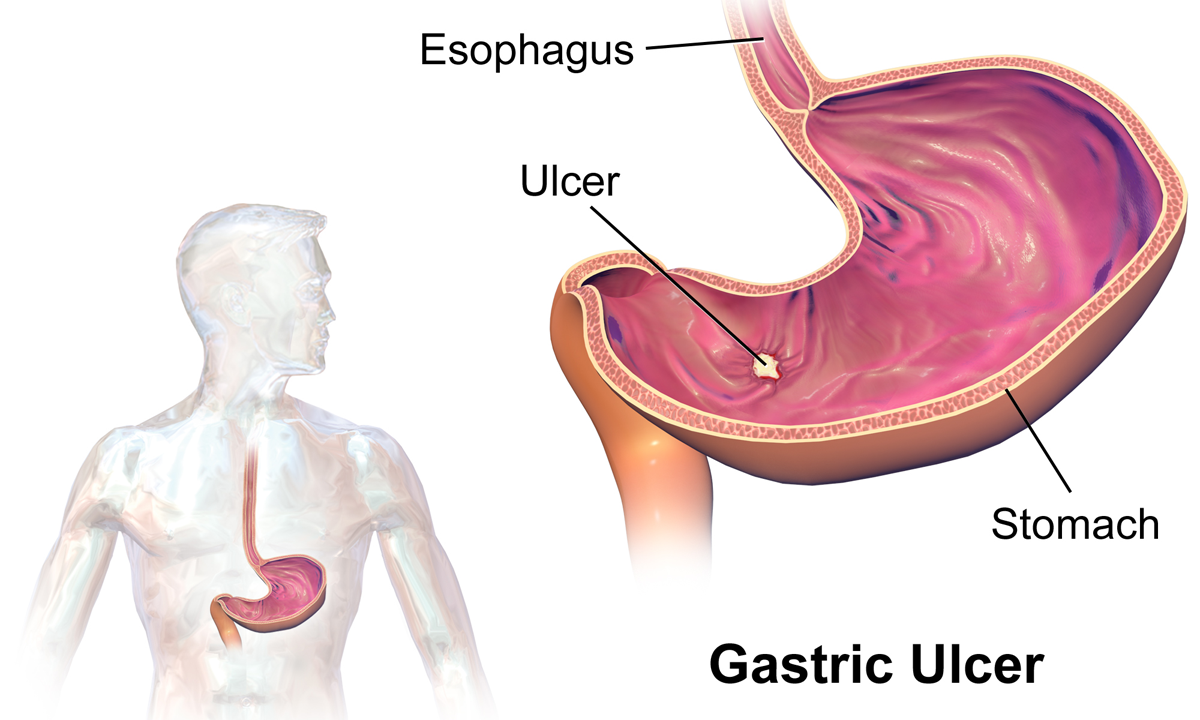
Peptic Ulcers
Peptic ulcer disease (PUD) is a break in the inner lining of the stomach, the first part of the small intestine, or sometimes the lower esophagus. An ulcer in the stomach is called a gastric ulcer, while one in the first part of the intestines is a duodenal ulcer. The most common symptoms of a duodenal ulcer are waking at night with upper abdominal pain and upper abdominal pain that improves with eating. With a gastric ulcer, the pain may worsen with eating. The pain is often described as a burning or dull ache. Other symptoms include belching, vomiting, weight loss, or poor appetite. About a third of older people have no symptoms. Complications may include bleeding, perforation, and blockage of the stomach. Bleeding occurs in as many as 15% of cases. Common causes include the bacteria ''Helicobacter pylori'' and non-steroidal anti-inflammatory drugs (NSAIDs). Other, less common causes include tobacco smoking, stress as a result of other serious health conditions, Behçet's di ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |