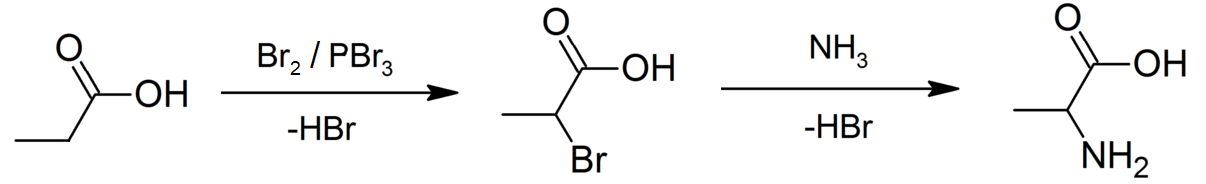
|
Piceol Ethers
Piceol is a phenolic compound found in the needles and in mycorrhizal roots of Norway spruces (''Picea abies''). Picein is the glucoside of piceol. Uses Piceol is used in the synthesis of several pharmaceutical drugs including octopamine, sotalol, bamethan, and dyclonine. Piceol can be used to make acetaminophen by oxime formation with hydroxylamine and subsequent Beckmann rearrangement in acid. Anticonvulsants are also possible by Mannich reaction: Metabolism Diprenylated derivatives of piceol can be isolated from ''Ophryosporus macrodon''. 4-Hydroxyacetophenone monooxygenase is an enzyme that transforms piceol into O-acetylhydroquinone. This enzyme is found in ''Pseudomonas fluorescens''. See also *Paroxypropione, where the acetyl group is replaced by a propionyl group. *Apocynin Apocynin, also known as acetovanillone, is a natural organic compound structurally related to vanillin. It has been isolated from a variety of plant sources and is being studied for its vari ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Picea Abies
''Picea abies'', the Norway spruce or European spruce, is a species of spruce native to Northern Europe, Northern, Central Europe, Central and Eastern Europe. It has branchlets that typically hang downwards, and the largest cones of any spruce, 9–17 cm long. It is very closely related to the Siberian spruce (''Picea obovata''), which replaces it east of the Ural Mountains, and with which it hybridizes freely. The Norway spruce has a wide distribution for it being planted for its wood, and is the species used as the main Christmas tree in several countries around the world. It was the first gymnosperm to have its genome sequenced. The Latin binomial nomenclature, specific epithet ''abies'' means “like ''Abies'', Fir tree” Description Norway spruce is a large, fast-growing evergreen coniferous tree growing tall and with a trunk diameter of 1 to 1.5 m. It can grow fast when young, up to 1 m per year for the first 25 years under good conditions, but becomes slower once ov ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Prenylation
Prenylation (also known as isoprenylation or lipidation) is the addition of hydrophobic molecules to a protein or a biomolecule. It is usually assumed that prenyl groups (3-methylbut-2-en-1-yl) facilitate attachment to cell membranes, similar to lipid anchors like the GPI anchor, though direct evidence of this has not been observed. Prenyl groups (also called isoprenyl groups, having one hydrogen atom more than isoprene) have been shown to be important for protein–protein binding through specialized prenyl-binding domains. Protein prenylation Protein prenylation involves the transfer of either a farnesyl or a geranylgeranyl moiety to C-terminal cysteine(s) of the target protein. There are three enzymes that carry out prenylation in the cell, farnesyl transferase, Caax protease and geranylgeranyl transferase I. Farnesylation is a type of prenylation, a post-translational modification of proteins by which an isoprenyl group is added to a cysteine residue. It is an important pro ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Apocynin
Apocynin, also known as acetovanillone, is a natural organic compound structurally related to vanillin. It has been isolated from a variety of plant sources and is being studied for its variety of pharmacological properties. History Apocynin was first described by Oswald Schmiedeberg, a German pharmacologist, in 1883 and was first isolated by Horace Finnemore, in 1908, from the root of Canadian hemp ('' Apocynum cannabinum''). At the time, this plant was already used for its known effectiveness against edema and heart problems. In 1971, apocynin was also isolated from ''Picrorhiza kurroa'', a small plant that grows at high altitudes in the western Himalayas. ''P. kurroa'' was used for ages as a treatment for liver and heart problems, jaundice, and asthma. In 1990, Simons et al. isolated apocynin to a pharmacologically useful level using an actively guided isolation procedure. Apocynin's observed anti-inflammatory capabilities proved to be a result of its ability to selectively ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Propionyl
Propionic acid (, from the Greek words πρῶτος : ''prōtos'', meaning "first", and πίων : ''píōn'', meaning "fat"; also known as propanoic acid) is a naturally occurring carboxylic acid with chemical formula CH3CH2CO2H. It is a liquid with a pungent and unpleasant smell somewhat resembling body odor. The anion CH3CH2CO2− as well as the salts and esters of propionic acid are known as propionates or propanoates. History Propionic acid was first described in 1844 by Johann Gottlieb, who found it among the degradation products of sugar. Over the next few years, other chemists produced propionic acid by different means, none of them realizing they were producing the same substance. In 1847, French chemist Jean-Baptiste Dumas established all the acids to be the same compound, which he called propionic acid, from the Greek words πρῶτος (prōtos), meaning ''first'', and πίων (piōn), meaning ''fat'', because it is the smallest H(CH2)''n''COOH acid that exhib ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acetyl
In organic chemistry, acetyl is a functional group with the chemical formula and the structure . It is sometimes represented by the symbol Ac (not to be confused with the element actinium). In IUPAC nomenclature, acetyl is called ethanoyl, although this term is barely heard. The acetyl group contains a methyl group () single-bonded to a carbonyl (). The carbonyl center of an acyl radical has one nonbonded electron with which it forms a chemical bond to the remainder ''R'' of the molecule. The acetyl moiety is a component of many organic compounds, including acetic acid, the neurotransmitter acetylcholine, acetyl-CoA, acetylcysteine, acetaminophen (also known as paracetamol), and acetylsalicylic acid (also known as aspirin). Acetylation In nature The introduction of an acetyl group into a molecule is called acetylation. In biological organisms, acetyl groups are commonly transferred from acetyl-CoA to other organic molecules. Acetyl-CoA is an intermediate both ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Paroxypropione
Paroxypropione, also known as paraoxypropiophenone, is a synthetic nonsteroidal estrogen which has been used medically as an antigonadotropin in Spain and Italy but appears to no longer be marketed. It was first synthesized in 1902. The antigonadotropic properties of the drug were discovered in 1951 and it entered clinical use shortly thereafter. Pharmacology Pharmacodynamics Paroxypropione is closely related structurally to ''p''-hydroxybenzoic acid and parabens such as methylparaben, and also bears a close resemblance to diethylstilbestrol (which, in fact, produces paroxypropione as an active metabolite) and alkylphenols like nonylphenol, all of which are also estrogens. The drug possesses relatively low affinity for the estrogen receptor and must be given at high dosages to achieve significant estrogenic and antigonadotropic effects, for instance, 0.8 to 1.6 g/day. It possesses 0.1% of the estrogenic activity and less than 0.5% of the antigonadotropic potency of estrone. Ch ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pseudomonas Fluorescens
''Pseudomonas fluorescens'' is a common Gram-negative, rod-shaped bacterium. It belongs to the ''Pseudomonas'' genus; 16S rRNA analysis as well as phylogenomic analysis has placed ''P. fluorescens'' in the ''P. fluorescens'' group within the genus, Text was copied from this source, which is available under Creative Commons Attribution 4.0 International License to which it lends its name. General characteristics ''Pseudomonas fluorescens'' has multiple flagella. It has an extremely versatile metabolism, and can be found in the soil and in water. It is an obligate aerobe, but certain strains are capable of using nitrate instead of oxygen as a final electron acceptor during cellular respiration. Optimal temperatures for growth of ''P. fluorescens'' are 25–30° C. It tests positive for the oxidase test. It is also a nonsaccharolytic bacterial species. Heat-stable lipases and proteases are produced by ''P. fluorescens'' and other similar pseudomonads. These enzymes cause m ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydroquinone
Hydroquinone, also known as benzene-1,4-diol or quinol, is an aromatic organic compound that is a type of phenol, a derivative of benzene, having the chemical formula C6H4(OH)2. It has two hydroxyl groups bonded to a benzene ring in a ''para'' position. It is a white granular solid. Substituted derivatives of this parent compound are also referred to as hydroquinones. The name "hydroquinone" was coined by Friedrich Wöhler in 1843. Production Hydroquinone is produced industrially in two main ways.Phillip M. Hudnall "Hydroquinone" in Ullmann's Encyclopedia of Industrial Chemistry 2002, Wiley-VCH, Weinheim. 2005 Wiley-VCH, Weinheim. . * The most widely used route is similar to the cumene process in reaction mechanism and involves the dialkylation of benzene with propene to give 1,4-diisopropylbenzene. This compound reacts with air to afford the bis(hydroperoxide), which is structurally similar to cumene hydroperoxide and rearranges in acid to give acetone and hydroquinone. * A se ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ophryosporus Macrodon
''Ophryosporus'' is a genus of South American flowering plants in the tribe Eupatorieae within the family Asteraceae The family Asteraceae, alternatively Compositae, consists of over 32,000 known species of flowering plants in over 1,900 genera within the order Asterales. Commonly referred to as the aster, daisy, composite, or sunflower family, Compositae w ....Meyen, Franz Julius Ferdinand. 1834. Reise um die Erde 1: 402 ; Species ; formerly included Several once considered part of ''Ophryosporus'' now regarded as better suited to other genera: '' Decachaeta Koanophyllon Mikania Stomatanthes'' References External links Asteraceae genera Flora of South America Taxonomy articles created by Polbot {{Eupatorieae-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mannich Reaction
In organic chemistry, the Mannich reaction is a three-component organic reaction that involves the amino alkylation of an acidic proton next to a carbonyl () functional group by formaldehyde () and a primary or secondary amine () or ammonia (). The final product is a β-amino-carbonyl compound also known as a Mannich base. Reactions between aldimines and α-methylene carbonyls are also considered Mannich reactions because these imines form between amines and aldehydes. The reaction is named after Carl Mannich. center, 500px, Scheme 1 - Ammonia or an amine reacts with formaldehyde and an alpha acidic proton of a carbonyl compound to a beta amino carbonyl compound. The Mannich reaction starts with the nucleophilic addition of an amine to a carbonyl group followed by dehydration to the Schiff base. The Schiff base is an electrophile which reacts in a second step in an electrophilic addition with an enol formed from a carbonyl compound containing an acidic alpha-proton. The Mann ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Picein
Picein is a phenolic compound found in mycorrhizal roots of Norway spruces (''Picea abies''). It is the glucoside of piceol Piceol is a phenolic compound found in the needles and in mycorrhizal roots of Norway spruces (''Picea abies''). Picein is the glucoside of piceol. Uses Piceol is used in the synthesis of several pharmaceutical drugs including octopamine, sota .... See also * Pungenin References Phenol glucosides Aromatic ketones {{phenol-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |