|
Promethium(III) Oxide
Promethium(III) oxide is a compound with the formula Pm2O3. It is the most common form of promethium Promethium is a chemical element with the symbol Pm and atomic number 61. All of its isotopes are radioactive; it is extremely rare, with only about 500–600 grams naturally occurring in Earth's crust at any given time. Promethium is one of onl .... Crystal structure Promethium oxide exists in three major crystalline forms: *a, b and c are lattice parameters, Z is the number of formula units per unit cell, density is calculated from X-ray data. The low-temperature cubic form converts to the monoclinic structure upon heating to 750–800 °C, and this transition can only be reversed by melting the oxide. The transition from the monoclinic to hexagonal form occurs at 1740 °C. References Promethium compounds Sesquioxides {{inorganic-compound-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Promethium(III) Chloride
Promethium(III) chloride is a chemical compound of promethium and chlorine with the formula PmCl3. It is an ionic, water soluble, crystalline salt that glows in the dark with a pale blue or green light due to promethium's intense radioactivity. Applications Promethium(III) chloride (with 147Pm) has been used to generate long-lasting glow in signal lights and buttons. This application relied on the unstable nature of promethium, which emitted beta radiation (electrons) with a half-life of several years. The electrons were absorbed by a phosphor A phosphor is a substance that exhibits the phenomenon of luminescence; it emits light when exposed to some type of radiant energy. The term is used both for fluorescent or phosphorescent substances which glow on exposure to ultraviolet or vi ..., generating visible glow. Unlike many other radioactive nuclides, promethium-147 does not emit alpha particles that would degrade the phosphor. References Promethium compounds Chlorid ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
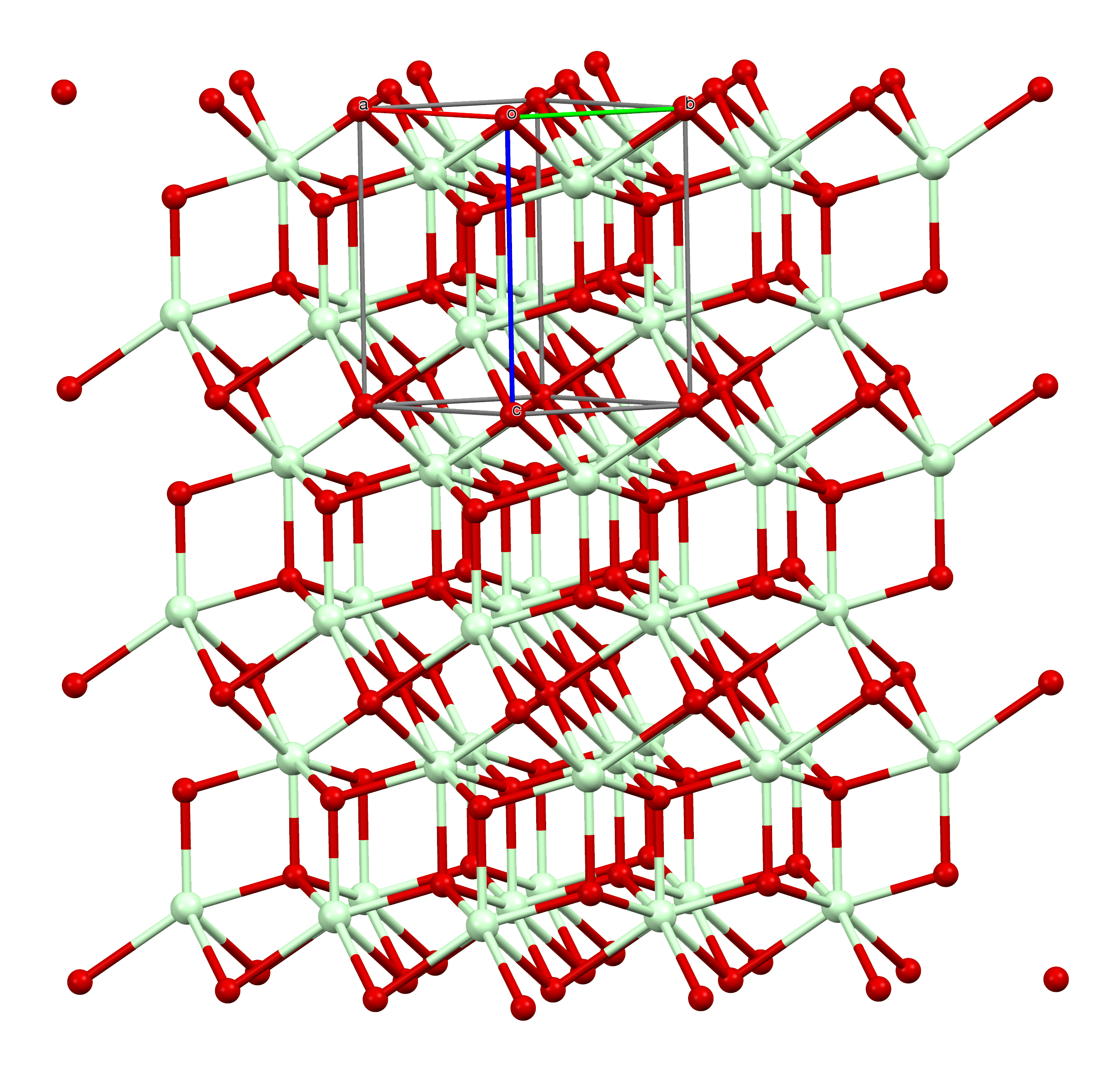
Neodymium(III) Oxide
Neodymium(III) oxide or neodymium sesquioxide is the chemical compound composed of neodymium and oxygen with the formula Nd2O3. It forms very light grayish-blue hexagonal crystals. The rare-earth mixture didymium, previously believed to be an element, partially consists of neodymium(III) oxide. Uses Neodymium(III) oxide is used to dope glass, including sunglasses, to make solid-state lasers, and to color glasses and enamels. Neodymium-doped glass turns purple due to the absorbance of yellow and green light, and is used in welding goggles. Some neodymium-doped glass is dichroic; that is, it changes color depending on the lighting. One kind of glass named for the mineral alexandrite appears blue in sunlight and red in artificial light. About 7000 tonnes of neodymium(III) oxide are produced worldwide each year. Neodymium(III) oxide is also used as a polymerization catalyst. Reactions Neodymium(III) oxide is formed when neodymium(III) nitride or neodymium(III) hydroxide is ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Samarium(III) Oxide
Samarium(III) oxide ( Sm2 O3) is a chemical compound. Samarium oxide readily forms on the surface of samarium metal under humid conditions or temperatures in excess of 150°C in dry air. Similar to rust on metallic iron, this oxide layer spalls off the surface of the metal, exposing more metal to continue the reaction. The oxide is commonly white to off yellow in color and is often encountered as a highly fine dust like powder. Uses Samarium(III) oxide is used in optical and infrared absorbing glass to absorb infrared radiation. Also, it is used as a neutron absorber in control rods for nuclear power reactors. The oxide catalyzes the dehydration and dehydrogenation of primary and secondary alcohols. Another use involves preparation of other samarium salts. Pradyot Patnaik. ''Handbook of Inorganic Chemicals''. McGraw-Hill, 2002, Preparations Samarium(III) oxide may be prepared by two methods: 1. thermal decomposition of samarium(III) carbonate, hydroxide, nitrate, oxalate or ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Neptunium(III) Oxide
Neptunium is a chemical element with the symbol Np and atomic number 93. A radioactive actinide metal, neptunium is the first transuranic element. Its position in the periodic table just after uranium, named after the planet Uranus, led to it being named after Neptune, the next planet beyond Uranus. A neptunium atom has 93 protons and 93 electrons, of which seven are valence electrons. Neptunium metal is silvery and tarnishes when exposed to air. The element occurs in three allotropic forms and it normally exhibits five oxidation states, ranging from +3 to +7. It is radioactive, poisonous, pyrophoric, and capable of accumulating in bones, which makes the handling of neptunium dangerous. Although many false claims of its discovery were made over the years, the element was first synthesized by Edwin McMillan and Philip H. Abelson at the Berkeley Radiation Laboratory in 1940. Since then, most neptunium has been and still is produced by neutron irradiation of uranium in nuclear re ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
.png)