|
Polonides
A polonide is a chemical compound of the radioactive element polonium with any element less electronegative than polonium. Polonides are usually prepared by a direct reaction between the elements at temperatures of around 300–400 °C... They are amongst the most chemically stable compounds of polonium,. and can be divided into two broad groups: *ionic polonides, which appear to contain the Po2− anion; *intermetallic polonides, in which the bonding is more complex. Some polonides are intermediate between these two cases and others are non-stoichiometric compounds. Alloys containing polonium are also classed as polonides. As polonium is immediately below tellurium in the periodic table, there are many chemical and structural similarities between polonides and tellurides. Naturally occurring polonides Lead polonide (PbPo) occurs naturally, as lead is produced in the alpha decay of polonium. Ionic polonides The polonides of the most electropositive metals show classic ionic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Calcium Polonide
Calcium polonide (CaPo) is an intermetallic compound made up of calcium and polonium. Rather than being found in nature, the compound is entirely synthetic, and difficult to study, due to polonium's high vapor pressure, radioactivity, and easy oxidation in air. Structure At atmospheric pressure, it crystalizes in the cubic rock crystal salt structure. At a high pressure of around 16.7 GPa, the structure is predicted to transform to the caesium chloride-type crystal structure. Electronic properties Based on theoretical calculations, calcium polonide is predicted to be a semiconductor. See also * Magnesium polonide Magnesium polonide (MgPo) is a salt of magnesium and polonium. It is a polonide, a set of very chemically stable compounds of polonium. Preparation Magnesium polonide can be produced by heating a mixture of elemental magnesium and polonium at 300� ... * Potassium polonide References Polonides Calcium compounds Rock salt crystal structure {{inorganic-com ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Magnesium Polonide
Magnesium polonide (MgPo) is a salt of magnesium and polonium. It is a polonide, a set of very chemically stable compounds of polonium. Preparation Magnesium polonide can be produced by heating a mixture of elemental magnesium and polonium at 300–400 °C. Structure Magnesium polonide has the nickeline (NiAs) structure. It is unusual among polonides in not being isomorphous with the corresponding sulfide, selenide A selenide is a chemical compound containing a selenium anion with oxidation number of −2 (Se2−), much as sulfur does in a sulfide. The chemistry of the selenides and sulfides is similar. Similar to sulfide, in aqueous solution, the selenide ion ... and telluride; only mercury polonide (HgPo) shares this property. References {{inorganic-chemistry-stub Magnesium compounds Polonides Nickel arsenide structure type ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Polonide
Sodium polonide is a radioactive chemical compound with the formula Na2 Po. This salt is a polonide, a set of very chemically stable compounds of polonium.. Due to the difference in electronegativity (ΔEN) between sodium and polonium (≈ 1.1 under the Pauling system) and the slight non-metallic character of polonium, it is intermediate between intermetallic phases and ionic compounds. Production This salt may be produced from the reaction between aqueous hydrogen polonide and sodium metal: :H2Po + 2 Na → Na2Po + H2 This method of synthesis is hampered by the chemical instability of hydrogen polonide. Sodium polonide may also be produced by heating sodium and polonium together at 300–400 °C. Crystal structure Like lithium polonide and potassium polonide, sodium polonide has the antifluorite In solid state chemistry, the fluorite structure refers to a common motif for compounds with the formula MX2. The X ions occupy the eight tetrahedral interstitial sites whereas M ion ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lanthanide Contraction
The lanthanide contraction is the greater-than-expected decrease in atomic radii/ionic radii of the elements in the lanthanide series from atomic number 57, lanthanum, to 71, lutetium, which results in smaller than otherwise expected atomic radii/ionic radii for the subsequent elements starting with 72, hafnium. Jolly, William L. ''Modern Inorganic Chemistry'', McGraw-Hill 1984, p. 22 The term was coined by the Norwegian geochemist Victor Goldschmidt in his series "Geochemische Verteilungsgesetze der Elemente" (Geochemical distribution laws of the elements). Goldschmidt, Victor M. "Geochemische Verteilungsgesetze der Elemente", Part V "Isomorphie und Polymorphie der Sesquioxyde. Die Lanthaniden-Kontraktion und ihre Konsequenzen", Oslo, 1925 Cause The effect results from poor shielding of nuclear charge (nuclear attractive force on electrons) by 4f electrons; the 6s electrons are drawn towards the nucleus, thus resulting in a smaller atomic radius. In single-electron atoms ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ionic Radius
Ionic radius, ''r''ion, is the radius of a monatomic ion in an ionic crystal structure. Although neither atoms nor ions have sharp boundaries, they are treated as if they were hard spheres with radii such that the sum of ionic radii of the cation and anion gives the distance between the ions in a crystal lattice. Ionic radii are typically given in units of either picometers (pm) or angstroms (Å), with 1 Å = 100 pm. Typical values range from 31 pm (0.3 Å) to over 200 pm (2 Å). The concept can be extended to solvated ions in liquid solutions taking into consideration the solvation shell. Trends Ions may be larger or smaller than the neutral atom, depending on the ion's electric charge. When an atom loses an electron to form a cation, the other electrons are more attracted to the nucleus, and the radius of the ion gets smaller. Similarly, when an electron is added to an atom, forming an anion, the added electron increases the size of the e ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
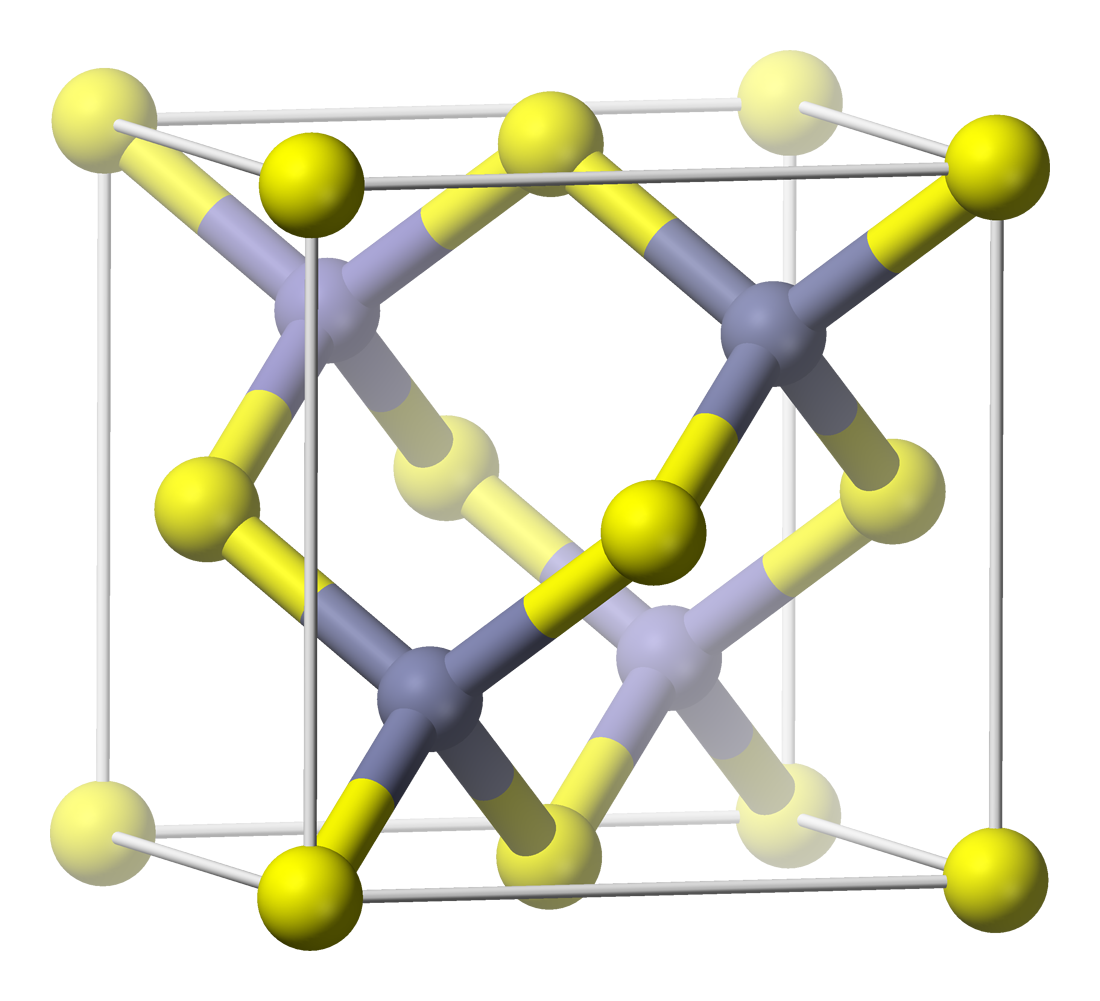
Zinc Polonide
Zinc is a chemical element with the Symbol (chemistry), symbol Zn and atomic number 30. Zinc is a slightly brittle metal at room temperature and has a shiny-greyish appearance when oxidation is removed. It is the first element in group 12 element, group 12 (IIB) of the periodic table. In some respects, zinc is chemically similar to magnesium: both elements exhibit only one normal oxidation state (+2), and the Zn2+ and Mg2+ ions are of similar size.The elements are from different metal groups. See periodic table. Zinc is the 24th most abundant Abundance of elements in Earth's crust, element in Earth's crust and has five stable isotopes. The most common zinc ore is sphalerite (zinc blende), a zinc sulfide mineral. The largest workable lodes are in Australia, Asia, and the United States. Zinc is refined by froth flotation of the ore, Roasting (metallurgy), roasting, and final extractive metallurgy, extraction using electricity (electrowinning). Zinc is an essential trace element fo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cadmium Polonide
Cadmium is a chemical element with the symbol Cd and atomic number 48. This soft, silvery-white metal is chemically similar to the two other stable metals in group 12, zinc and mercury. Like zinc, it demonstrates oxidation state +2 in most of its compounds, and like mercury, it has a lower melting point than the transition metals in groups 3 through 11. Cadmium and its congeners in group 12 are often not considered transition metals, in that they do not have partly filled ''d'' or ''f'' electron shells in the elemental or common oxidation states. The average concentration of cadmium in Earth's crust is between 0.1 and 0.5 parts per million (ppm). It was discovered in 1817 simultaneously by Stromeyer and Hermann, both in Germany, as an impurity in zinc carbonate. Cadmium occurs as a minor component in most zinc ores and is a byproduct of zinc production. Cadmium was used for a long time as a corrosion-resistant plating on steel, and cadmium compounds are used as red, orange ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sphalerite
Sphalerite (sometimes spelled sphaelerite) is a sulfide mineral with the chemical formula . It is the most important ore of zinc. Sphalerite is found in a variety of deposit types, but it is primarily in Sedimentary exhalative deposits, sedimentary exhalative, Carbonate-hosted lead-zinc ore deposits, Mississippi-Valley type, and Volcanogenic massive sulfide ore deposit, volcanogenic massive sulfide deposits. It is found in association with galena, chalcopyrite, pyrite (and other sulfide mineral, sulfides), calcite, dolomite (mineral), dolomite, quartz, rhodochrosite, and fluorite. German geologist Ernst Friedrich Glocker discovered sphalerite in 1847, naming it based on the Greek word ''sphaleros'', meaning "deceiving", due to the difficulty of identifying the mineral. In addition to zinc, sphalerite is an ore of cadmium, gallium, germanium, and indium. Miners have been known to refer to sphalerite as ''zinc blende'', ''black-jack'', and ''ruby blende''. Marmatite is an opaque ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Beryllium Polonide
Beryllium is a chemical element with the symbol Be and atomic number 4. It is a steel-gray, strong, lightweight and brittle alkaline earth metal. It is a divalent element that occurs naturally only in combination with other elements to form minerals. Notable gemstones high in beryllium include beryl ( aquamarine, emerald) and chrysoberyl. It is a relatively rare element in the universe, usually occurring as a product of the spallation of larger atomic nuclei that have collided with cosmic rays. Within the cores of stars, beryllium is depleted as it is fused into heavier elements. Beryllium constitutes about 0.0004 percent by mass of Earth's crust. The world's annual beryllium production of 220 tons is usually manufactured by extraction from the mineral beryl, a difficult process because beryllium bonds strongly to oxygen. In structural applications, the combination of high flexural rigidity, thermal stability, thermal conductivity and low density (1.85 times that of water) ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nickeline
Nickeline or niccolite is a mineral consisting primarily of nickel arsenide (NiAs). The naturally-occurring mineral contains roughly 43.9% nickel and 56.1% arsenic by mass, but composition of the mineral may vary slightly. Small quantities of sulfur, iron and cobalt are usually present, and sometimes the arsenic is largely replaced by antimony. This last forms an isomorphous series with breithauptite (nickel antimonide). Etymology and history Medieval miners looking for copper in the German Erzgebirge ("Ore Mountains") would sometimes find a red mineral, superficially resembling copper ore. Upon attempting extraction, no copper was produced, and subsequently, the miners would be afflicted with mysterious illness. They blamed a mischievous sprite of German mythology, Nickel (similar to ''Old Nick'') for besetting the copper (German: Kupfer). This German equivalent of "copper-nickel" was used as early as 1694 (other old German synonyms are ''Rotnickelkies'' and ''Arsennickel' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nickeline
Nickeline or niccolite is a mineral consisting primarily of nickel arsenide (NiAs). The naturally-occurring mineral contains roughly 43.9% nickel and 56.1% arsenic by mass, but composition of the mineral may vary slightly. Small quantities of sulfur, iron and cobalt are usually present, and sometimes the arsenic is largely replaced by antimony. This last forms an isomorphous series with breithauptite (nickel antimonide). Etymology and history Medieval miners looking for copper in the German Erzgebirge ("Ore Mountains") would sometimes find a red mineral, superficially resembling copper ore. Upon attempting extraction, no copper was produced, and subsequently, the miners would be afflicted with mysterious illness. They blamed a mischievous sprite of German mythology, Nickel (similar to ''Old Nick'') for besetting the copper (German: Kupfer). This German equivalent of "copper-nickel" was used as early as 1694 (other old German synonyms are ''Rotnickelkies'' and ''Arsennickel' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |