|
Plant Sterol
Phytosterols are phytosteroids, similar to cholesterol, that serve as structural components of biological membranes of plants. They encompass plant sterols and stanol ester, stanols. More than 250 sterols and related compounds have been identified. Free phytosterols extracted from oils are insoluble in water, relatively insoluble in oil, and soluble in alcohols. Phytosterol-enriched foods and dietary supplements have been marketed for decades. Despite well-documented LDL cholesterol-lowering effects from long-term consumption of phytosterols, there is insufficient evidence for an effect on cardiovascular diseases, fasting blood sugar, glycated hemoglobin, or overall mortality rate. Structure They have a fused polycyclic structure and vary in carbon side chains and / or presence or absence of a double bond (saturation). They are divided into 4,4-dimethyl phytosterols, 4-monomethyl phytosterols, and 4-desmethyl phytosterols based on the location of methyl groups at the carbon-4 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
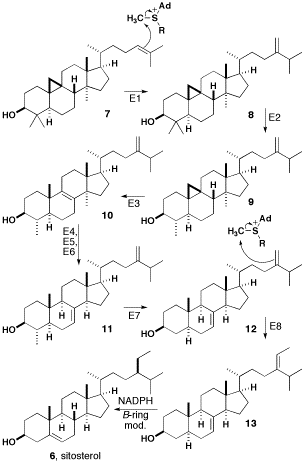
Sitosterol Structure
β-sitosterol (beta-sitosterol) is one of several phytosterols (plant sterols) with chemical structures similar to that of cholesterol. It is a white, waxy powder with a characteristic odor, and is one of the components of the food additive E499. Phytosterols are hydrophobic and soluble in alcohols. Natural occurrences and food β-sitosterol is widely distributed in the plant kingdom. It is found in vegetable oil, nuts, avocados, and derived prepared foods such as salad dressings. Human research β-sitosterol is being studied for its potential to reduce benign prostatic hyperplasia (BPH) and blood cholesterol levels. Genetic disorder While plant sterols are usually beneficial, there is a rare autosomal recessive genetic disorder phytosterolemia which causes over-absorption of phytosterols. Precursor of anabolic steroid boldenone Being a steroid, β-sitosterol is a precursor of anabolic steroid boldenone. Boldenone undecylenate is commonly used in veterinary medicine to induce ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Stigmasterol
Stigmasterol – a plant sterol (''phytosterol'') – is among the most abundant of plant sterols, having a major function to maintain the structure and physiology of cell membranes. In the European Union, it is a food additive listed with E number E499, and may be used in food manufacturing to increase the phytosterol content, potentially lowering the levels of LDL cholesterol. Discovery Once called ''Wulzen factor'' in the mid-20th century, stigmasterol was discovered by the University of California physiologist Rosalind Wulzen (born 1886). Natural occurrences Stigmasterol is an unsaturated phytosterol occurring in the plant fats or oils of numerous plants, such as soybean, calabar bean, and rape seed, and in herbs used in herbalism practices, including the Chinese herbs ''Ophiopogon japonicus'' (Mai men dong), in ''Mirabilis jalapa''. Stigmasterol is a constituent of various vegetables, legumes, nuts, seeds, and unpasteurized milk. Pasteurization will inactivate stig ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ergosterol
Ergosterol (ergosta-5,7,22-trien-3β-ol) is a sterol found in cell membranes of fungi and protozoa, serving many of the same functions that cholesterol serves in animal cells. Because many fungi and protozoa cannot survive without ergosterol, the enzymes that synthesize it have become important targets for drug discovery. In human nutrition, ergosterol is a provitamin form of vitamin D2; exposure to ultraviolet (UV) light causes a chemical reaction that produces vitamin D2. Role in fungi Ergosterol (ergosta-5,7,22-trien-3β-ol) is a sterol found in fungi, and named for ergot, the common name of members of the fungal genus '' Claviceps'' from which ergosterol was first isolated. Ergosterol is a component of yeast and other fungal cell membranes, serving many of the same functions that cholesterol serves in animal cells. Its specificity in higher fungi is thought to be related to the climatic instabilities (highly varying humidity and moisture conditions) encountered by these orga ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cholesterol
Cholesterol is any of a class of certain organic molecules called lipids. It is a sterol (or modified steroid), a type of lipid. Cholesterol is biosynthesized by all animal cells and is an essential structural component of animal cell membranes. When chemically isolated, it is a yellowish crystalline solid. Cholesterol also serves as a precursor for the biosynthesis of steroid hormones, bile acid and vitamin D. Cholesterol is the principal sterol synthesized by all animals. In vertebrates, hepatic cells typically produce the greatest amounts. It is absent among prokaryotes (bacteria and archaea), although there are some exceptions, such as '' Mycoplasma'', which require cholesterol for growth. François Poulletier de la Salle first identified cholesterol in solid form in gallstones in 1769. However, it was not until 1815 that chemist Michel Eugène Chevreul named the compound "cholesterine". Etymology The word "cholesterol" comes from the Ancient Greek ''chole-'' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cycloartenol
Cycloartenol is an important triterpenoid of the sterol class which is found in plants. It is the starting point for the synthesis of almost all plant steroids, making them chemically distinct from the steroids of fungi and animals, which are, instead, produced from lanosterol. Synthesis The biosynthesis of cycloartenol starts from the triterpenoid squalene. It is the first precursor in the biosynthesis of other stanols and sterols, referred to as phytostanols and phytosterols in photosynthetic Photosynthesis is a process used by plants and other organisms to convert light energy into chemical energy that, through cellular respiration, can later be released to fuel the organism's activities. Some of this chemical energy is stored in c ... organisms and plants. The identities and distribution of phytostanols and phytosterols is characteristic of a plant species. References Sterols Triterpenes Cyclopropanes {{Alcohol-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Brassicasterol
Brassicasterol (24-methyl cholest-5,22-dien-3β-ol) is a 28-carbon sterol synthesised by several unicellular algae (phytoplankton) and some terrestrial plants, like rape. This compound has frequently been used as a biomarker for the presence of (marine) algal matter in the environment, and is one of the ingredients for E number E499. There is some evidence to suggest that it may also be a relevant additional biomarker in Alzheimer's disease. Chemical properties Solubility Brassicasterol has a low water solubility and, as a consequence, a high octanol-water partition coefficient. This means that, in most environmental systems, brassicasterol will be associated with the solid phase. Degradation In anaerobic sediments and soils, brassicasterol is stable for many hundreds of years, enabling it to be used as an indicator of past algal production (see below). Chemical analysis Since the molecule has a hydroxyl (-OH) group, it is frequently bound to other lipids including glycerols; m ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Campestanol
Campestanol is a natural phytosterol Phytosterols are phytosteroids, similar to cholesterol, that serve as structural components of biological membranes of plants. They encompass plant sterols and stanols. More than 250 sterols and related compounds have been identified. Free phyto .... External links Phytosterol effects on milk and yogurt microflora Phytosterols {{organic-chem-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Stigmastanol
Stigmastanol (sitostanol) is a phytosterol found in a variety of plant sources. Similar to sterol esters and stanol esters, stigmastanol inhibits the absorption of cholesterol from the diet. Animal studies suggest that it also inhibits biosynthesis of cholesterol in the liver. Stigmastanol is the product of the reduction of β-sitosterol and the hydrogenation of stigmasterol. See also * Stigmasterol Stigmasterol – a plant sterol (''phytosterol'') – is among the most abundant of plant sterols, having a major function to maintain the structure and physiology of cell membranes. In the European Union, it is a food additive listed with E numb ..., a closely related sterol References Phytosterols {{steroid-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Stigmasterol
Stigmasterol – a plant sterol (''phytosterol'') – is among the most abundant of plant sterols, having a major function to maintain the structure and physiology of cell membranes. In the European Union, it is a food additive listed with E number E499, and may be used in food manufacturing to increase the phytosterol content, potentially lowering the levels of LDL cholesterol. Discovery Once called ''Wulzen factor'' in the mid-20th century, stigmasterol was discovered by the University of California physiologist Rosalind Wulzen (born 1886). Natural occurrences Stigmasterol is an unsaturated phytosterol occurring in the plant fats or oils of numerous plants, such as soybean, calabar bean, and rape seed, and in herbs used in herbalism practices, including the Chinese herbs ''Ophiopogon japonicus'' (Mai men dong), in ''Mirabilis jalapa''. Stigmasterol is a constituent of various vegetables, legumes, nuts, seeds, and unpasteurized milk. Pasteurization will inactivate stig ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Campesterol
Campesterol is a phytosterol whose chemical structure is similar to that of cholesterol, and is one of the ingredients for E number E499. Natural occurrences Many vegetables, fruits, nuts, and seeds contain campesterol, but in low concentrations. Banana, pomegranate, pepper, coffee, grapefruit, cucumber, onion, oat, potato, and lemon grass (citronella) are few examples of common sources containing campesterol at roughly 1–7 mg/100 g of the edible portion. In contrast, canola and corn oils contain as much as 16–100 mg/100 g. Levels are variable and are influenced by geography and growing environment. In addition, different strains have different levels of plant sterols. A number of new genetic strains are currently being engineered with the goal of producing varieties high in campesterol and other plant sterols. It is also found in dandelion coffee. It is so named because it was first isolated from the rapeseed (''Brassica campestris''). It is thought to have a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sterol Ester
Sterol esters are a heterogeneous group of chemical compounds. They are created when the hydroxyl group of a sterol and a fatty acid undergo an esterification reaction. They can be found in trace amounts in every cell type but are highly enriched in foam cells and are common components of human skin oil. Plant sterol esters have been shown to reduce the level of low-density lipoprotein (LDL) cholesterol in blood when ingested. Plant sterol esters used for dietary supplements are made from phytosterols and fatty acids also derived from plants. They are added to certain oil-containing products like margarine, milk, or yogurt to make functional foods for controlling cholesterol levels. Studies have indicated that consumption of about 2 grams per day of phytosterol esters provides a reduction in LDL cholesterol of around 10%. Sterol esters are added to certain Unilever products under the brand name Becel/Flora. See also * Phytosterols * Stanol ester * Sitosterolemia Sitosterolemia ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ergosterol
Ergosterol (ergosta-5,7,22-trien-3β-ol) is a sterol found in cell membranes of fungi and protozoa, serving many of the same functions that cholesterol serves in animal cells. Because many fungi and protozoa cannot survive without ergosterol, the enzymes that synthesize it have become important targets for drug discovery. In human nutrition, ergosterol is a provitamin form of vitamin D2; exposure to ultraviolet (UV) light causes a chemical reaction that produces vitamin D2. Role in fungi Ergosterol (ergosta-5,7,22-trien-3β-ol) is a sterol found in fungi, and named for ergot, the common name of members of the fungal genus '' Claviceps'' from which ergosterol was first isolated. Ergosterol is a component of yeast and other fungal cell membranes, serving many of the same functions that cholesterol serves in animal cells. Its specificity in higher fungi is thought to be related to the climatic instabilities (highly varying humidity and moisture conditions) encountered by these orga ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |