|
Pentyl Propionate
Pentyl propanoate (also known as amyl propionate) is an organic ester formed by the condensation of pentan-1-ol and propanoic acid Propionic acid (, from the Greek words πρῶτος : ''prōtos'', meaning "first", and πίων : ''píōn'', meaning "fat"; also known as propanoic acid) is a naturally occurring carboxylic acid with chemical formula CH3CH2CO2H. It is a liqu .... It is a colorless liquid with an apple-like odor, that floats on water. References External links Propionate esters {{ester-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ester
In chemistry, an ester is a compound derived from an oxoacid (organic or inorganic) in which at least one hydroxyl group () is replaced by an alkoxy group (), as in the substitution reaction of a carboxylic acid and an alcohol. Glycerides are fatty acid esters of glycerol; they are important in biology, being one of the main classes of lipids and comprising the bulk of animal fats and vegetable oils. Esters typically have a pleasant smell; those of low molecular weight are commonly used as fragrances and are found in essential oils and pheromones. They perform as high-grade solvents for a broad array of plastics, plasticizers, resins, and lacquers, and are one of the largest classes of synthetic lubricants on the commercial market. Polyesters are important plastics, with monomers linked by ester moieties. Phosphoesters form the backbone of DNA molecules. Nitrate esters, such as nitroglycerin, are known for their explosive properties. '' Nomenclature Etymology Th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Propyl Propanoate
Propyl propanoate (propyl propionate) is the organic compound with the molecular formula C6 H12 O2. It is the ester of propanol and propionic acid. Like most esters, propyl propanoate is a colorless liquid with a fruity odor. The scent of propyl propionate is described as a chemically tinged pineapple or pear. It is used in perfumery and as a solvent. The refractive index at 20 °C is 1.393. Because n-propyl propionate is a low-odor, moderately volatile, non-HAP ester solvent with good solvent activity and versatility, on April 1, 2006 it is considered a safer substitute for toluene
Toluene (), also known as toluol ( ...
[...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Butyl Propanoate
Butyl propionate is a butyl ester of propionic acid. This ester has the chemical formula . Occurrence in nature Butyl propionate is a plant metabolite, a human metabolite and an insect attractant. Butyl propionate is a natural ester found in Armenian apricot. Preparation Butyl propionate is derived by esterification of propionic acid with butanol. : Properties Butyl propionate is a colorless liquid with a pear drops-like or apple-like odor. The liquid is less dense than water. Its vapor is 4.5 times denser than the air at the mean ocean level. Uses Butyl propionate is used to make fragrances, perfumes and as a flavoring. It is also used in paints and primers for auto body or engine, appliance coatings (paints designed specifically for painting household items and vehicles like microwave ovens, refrigerators and automobiles), enamels, lacquers, and printing inks, as a solvent for adhesives and nitrocellulose, and in polymerization reactions for acrylic resins. Hazards an ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hexyl Propanoate
In chemistry, an ester is a compound derived from an acid (organic or inorganic) in which the hydrogen atom (H) of at least one acidic hydroxyl group () of that acid is replaced by an organyl group (). Analogues derived from oxygen replaced by other chalcogens belong to the ester category as well (i.e. esters of acidic , , , and groups). According to some authors, organyl derivatives of acidic hydrogen of other acids are esters as well (e.g. amides), but not according to the IUPAC. An example of an ester formation is the substitution reaction between a carboxylic acid () and an alcohol (R'OH), forming an ester (), where R and R′ are organyl groups, or H in the case of esters of formic acid. Glycerides, which are fatty acid esters of glycerol, are important esters in biology, being one of the main classes of lipids, and making up the bulk of animal fats and vegetable oils. Esters of carboxylic acids with low molecular weight are commonly used as fragrances and found in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pentyl Acetate
Amyl acetate (pentyl acetate) is an organic compound and an ester with the chemical formula CH3COO H2sub>4CH3 and the molecular weight 130.19g/mol. It is colorless and has a scent similar to bananas and apples. The compound is the condensation product of acetic acid and 1-pentanol. However, esters formed from other pentanol isomers (amyl alcohols), or mixtures of pentanols, are often referred to as amyl acetate. The symptoms of exposure to amyl acetate in humans are dermatitis, central nervous system depression, narcosis and irritation to the eyes and nose. Uses It is used as a flavoring agent, as a paint and lacquer solvent, and in the preparation of penicillin. It is an inactive ingredient in liquid bandages. It is used as a fuel in the Hefner lamp. See also *Isoamyl acetate, also known as banana oil. *Ester In chemistry, an ester is a compound derived from an oxoacid (organic or inorganic) in which at least one hydroxyl group () is replaced by an alkoxy group (), a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pentyl Butanoate
Pentyl butyrate, also known as pentyl butanoate or amyl butyrate, is an ester that is formed when pentanol is reacted with butyric acid,N-AMYL BUTYRATE ''Cameo Chemicals'', usually in the presence of sulfuric acid as a . This ester has a smell reminiscent of |
Ester
In chemistry, an ester is a compound derived from an oxoacid (organic or inorganic) in which at least one hydroxyl group () is replaced by an alkoxy group (), as in the substitution reaction of a carboxylic acid and an alcohol. Glycerides are fatty acid esters of glycerol; they are important in biology, being one of the main classes of lipids and comprising the bulk of animal fats and vegetable oils. Esters typically have a pleasant smell; those of low molecular weight are commonly used as fragrances and are found in essential oils and pheromones. They perform as high-grade solvents for a broad array of plastics, plasticizers, resins, and lacquers, and are one of the largest classes of synthetic lubricants on the commercial market. Polyesters are important plastics, with monomers linked by ester moieties. Phosphoesters form the backbone of DNA molecules. Nitrate esters, such as nitroglycerin, are known for their explosive properties. '' Nomenclature Etymology Th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pentan-1-ol
1-Pentanol, (or ''n''-pentanol, pentan-1-ol), is an alcohol with five carbon atoms and the molecular formula C5H11OH. CRC Handbook of Chemistry and Physics 65Th Ed. 1-Pentanol is a colourless liquid with a distinctive aroma. It is the straight-chain form of amyl alcohol, one of 8 isomers with that formula. The hydroxyl group (OH) is the active site of many reactions. The ester formed from 1-pentanol and butyric acid is pentyl butyrate, which smells like apricot. The ester formed from 1-pentanol and acetic acid is amyl acetate (also called pentyl acetate), which smells like banana. In 2014, a study was conducted comparing the performance of diesel fuel blends with various proportions of pentanol as an additive. While gaseous emissions increased with higher concentrations of pentanol, particulate emissions decreased.Wei, Liangjie & Cheung, C.s & Huang, Zuohua. (2014). Effect of n-pentanol addition on the combustion, performance and emission characteristics of a direct ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
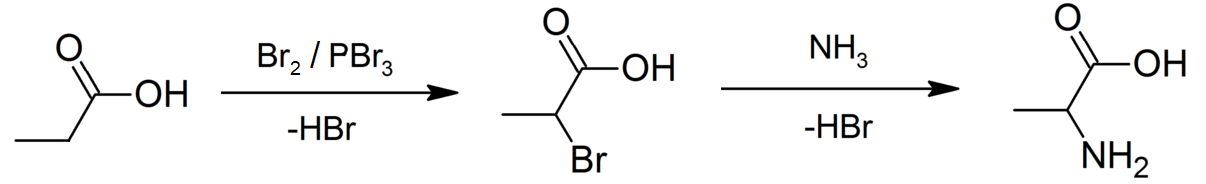
Propanoic Acid
Propionic acid (, from the Greek words πρῶτος : ''prōtos'', meaning "first", and πίων : ''píōn'', meaning "fat"; also known as propanoic acid) is a naturally occurring carboxylic acid with chemical formula CH3CH2CO2H. It is a liquid with a pungent and unpleasant smell somewhat resembling body odor. The anion CH3CH2CO2− as well as the salts and esters of propionic acid are known as propionates or propanoates. History Propionic acid was first described in 1844 by Johann Gottlieb, who found it among the degradation products of sugar. Over the next few years, other chemists produced propionic acid by different means, none of them realizing they were producing the same substance. In 1847, French chemist Jean-Baptiste Dumas established all the acids to be the same compound, which he called propionic acid, from the Greek words πρῶτος (prōtos), meaning ''first'', and πίων (piōn), meaning ''fat'', because it is the smallest H(CH2)''n''COOH acid that exhibits ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |