|
PEG-150 Jojoba
PEG-150 hydrogenated jojoba is an ethoxylated version of fully hydrogenated jojoba wax and the most water soluble form of jojoba available. This versatile emollient functions as a secondary emulsifier and provides viscosity adjustments in hair care formulas and fragrance oil solubilization in aqueous solutions. PEG-150 Hydrogenated Jojoba is commonly used in cosmetics formulations. Physical properties PEG-150 hydrogenated jojoba is a solid, flaked, free-flowing material with a melting point of . Testing in a 5% aqueous solution shows that it has a neutral pH, high clarity in water, and low viscosity The viscosity of a fluid is a measure of its resistance to deformation at a given rate. For liquids, it corresponds to the informal concept of "thickness": for example, syrup has a higher viscosity than water. Viscosity quantifies the inte .... The HLB value is approximately 18. References Cosmetics chemicals {{cosmetics-chem-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
International Nomenclature Of Cosmetic Ingredients
The International Nomenclature of Cosmetic Ingredients, abbreviated INCI, are the unique identifiers for cosmetic ingredients such as waxes, oils, pigments, and other chemicals that are assigned in accordance with rules established by the Personal Care Products Council (PCPC), previously the Cosmetic, Toiletry, and Fragrance Association (CTFA). INCI names often differ greatly from systematic chemical nomenclature or from more common trivial names and is a mixture of conventional scientific names, Latin and English words. INCI nomenclature conventions "are continually reviewed and modified when necessary to reflect changes in the industry, technology, and new ingredient developments". INCI and CAS The relationship between a CAS Registry Number and an INCI name is not always one-to-one. In some cases, more than one INCI name may have the same CAS number, or more than one CAS number may apply to an INCI name. For example, the CAS number 1245638-61-2 has the CA Index Name of 2-Propeno ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
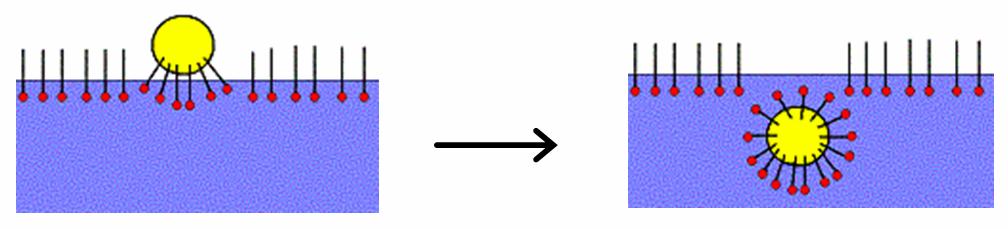
Ethoxylation
Ethoxylation is a chemical reaction in which ethylene oxide adds to a substrate. It is the most widely practiced alkoxylation, which involves the addition of epoxides to substrates. In the usual application, alcohols and phenols are converted into R(OC2H4)nOH where n ranges from 1 to 10. Such compounds are called alcohol ethoxylates. Alcohol ethoxylates are often converted to related species called ethoxysulfates. Alcohol ethoxylates and ethoxysulfates are surfactants, used widely in cosmetic and other commercial products. The process is of great industrial significance with more than 2,000,000 metric tons of various ethoxylates produced worldwide in 1994. Production The process was developed at the Ludwigshafen laboratories of IG Farben by Conrad Schöller and during the 1930s. Alcohol ethoxylates Industrial ethoxylation is primarily performed upon fatty alcohols in order to generate fatty alcohol ethoxylates (FAE's), which are a common form of nonionic surfactant (e.g. octa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogenated
Hydrogenation is a chemical reaction between molecular hydrogen (H2) and another compound or element, usually in the presence of a catalyst such as nickel, palladium or platinum. The process is commonly employed to reduce or saturate organic compounds. Hydrogenation typically constitutes the addition of pairs of hydrogen atoms to a molecule, often an alkene. Catalysts are required for the reaction to be usable; non-catalytic hydrogenation takes place only at very high temperatures. Hydrogenation reduces double and triple bonds in hydrocarbons. Process Hydrogenation has three components, the unsaturated substrate, the hydrogen (or hydrogen source) and, invariably, a catalyst. The reduction reaction is carried out at different temperatures and pressures depending upon the substrate and the activity of the catalyst. Related or competing reactions The same catalysts and conditions that are used for hydrogenation reactions can also lead to isomerization of the alkenes from cis to ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Jojoba Ester
Jojoba esters are the hydrogenation or transesterification product of Jojoba oil. Jojoba Esters are commonly used in cosmetic formulations as an emollient, due to its remarkable similarity to the natural oils produced by the human skin, and its high oxidative stability. Fully hydrogenated jojoba esters are most often small beads used to exfoliate the skin. Chemical structure Jojoba esters are proper waxes; there is no triglyceride component of jojoba esters. Chemically, jojoba esters are a complex mixture of long chain fatty acids and fatty alcohols joined by an ester bond, resulting in a total length of 36 to 46 carbon atoms. Jojoba esters are produced by the interesterification of jojoba oil, hydrogenated jojoba oil, or a mixture of the two. Pure jojoba oil and pure hydrogenated jojoba oil are also correctly described as jojoba esters. The CTFA does not regard ''partially''-hydrogenated jojoba oil as jojoba esters. For this reason, jojoba esters must not contain any trans-un ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Soluble
In chemistry, solubility is the ability of a substance, the solute, to form a solution with another substance, the solvent. Insolubility is the opposite property, the inability of the solute to form such a solution. The extent of the solubility of a substance in a specific solvent is generally measured as the concentration of the solute in a saturated solution, one in which no more solute can be dissolved. At this point, the two substances are said to be at the solubility equilibrium. For some solutes and solvents, there may be no such limit, in which case the two substances are said to be "miscible in all proportions" (or just "miscible"). The solute can be a solid, a liquid, or a gas, while the solvent is usually solid or liquid. Both may be pure substances, or may themselves be solutions. Gases are always miscible in all proportions, except in very extreme situations,J. de Swaan Arons and G. A. M. Diepen (1966): "Gas—Gas Equilibria". ''Journal of Chemical Physics'', vo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Jojoba
Jojoba (; botanical name: ''Simmondsia chinensis'')also commonly called goat nut, deer nut, pignut, wild hazel, quinine nut, coffeeberry, and gray box bushis native to the Southwestern United States. ''Simmondsia chinensis'' is the sole species of the family Simmondsiaceae, placed in the order Caryophyllales. Jojoba is grown commercially to produce jojoba oil, a liquid wax ester extracted from its seed. Distribution The plant is a native shrub of the Sonoran Desert, Colorado Desert, Baja California Desert, and California chaparral and woodlands habitats in the Peninsular Ranges and San Jacinto Mountains. It is found in southern California, Arizona, and Utah (U.S.), and Baja California state (Mexico). Jojoba is endemic to North America, and occupies an area of approximately between latitudes 25° and 31° North and between longitudes 109° and 117° West. Description ''Simmondsia chinensis'', or jojoba, typically grows to tall, with a broad, dense crown, but there have bee ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Emollient
A moisturizer, or emollient, is a cosmetic preparation used for protecting, moisturizing, and lubricating the skin. These functions are normally performed by sebum produced by healthy skin. The word "emollient" is derived from the Latin verb ''mollire'', to soften. Mechanism of action In the human body, water constantly evaporates from the deeper layers of the skin through an effect known as transepidermal water loss (TEWL). By regulating its water content, human skin naturally maintains a dry, easily shed surface as a barrier against pathogens, dirt, or damage, while protecting itself from drying out and becoming brittle and rigid. The ability to retain moisture depends on the lipid bilayer between the corneocytes. Moisturizers modify the rate of water loss, with active ingredients of moisturizers falling into one of two categories: occlusives and humectants. Occlusives form a coating on the surface of the skin, keeping moisture from escaping. The more occlusive the formula ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Emulsifier
An emulsion is a mixture of two or more liquids that are normally immiscible (unmixable or unblendable) owing to liquid-liquid phase separation. Emulsions are part of a more general class of two-phase systems of matter called colloids. Although the terms ''colloid'' and ''emulsion'' are sometimes used interchangeably, ''emulsion'' should be used when both phases, dispersed and continuous, are liquids. In an emulsion, one liquid (the dispersed phase) is dispersed in the other (the continuous phase). Examples of emulsions include vinaigrettes, homogenized milk, liquid biomolecular condensates, and some cutting fluids for metal working. Two liquids can form different types of emulsions. As an example, oil and water can form, first, an oil-in-water emulsion, in which the oil is the dispersed phase, and water is the continuous phase. Second, they can form a water-in-oil emulsion, in which water is the dispersed phase and oil is the continuous phase. Multiple emulsions are also p ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Viscosity
The viscosity of a fluid is a measure of its resistance to deformation at a given rate. For liquids, it corresponds to the informal concept of "thickness": for example, syrup has a higher viscosity than water. Viscosity quantifies the internal frictional force between adjacent layers of fluid that are in relative motion. For instance, when a viscous fluid is forced through a tube, it flows more quickly near the tube's axis than near its walls. Experiments show that some stress (such as a pressure difference between the two ends of the tube) is needed to sustain the flow. This is because a force is required to overcome the friction between the layers of the fluid which are in relative motion. For a tube with a constant rate of flow, the strength of the compensating force is proportional to the fluid's viscosity. In general, viscosity depends on a fluid's state, such as its temperature, pressure, and rate of deformation. However, the dependence on some of these properties is ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Solubilization
Micellar solubilization (solubilization) is the process of incorporating the solubilizate (the component that undergoes solublization) into or onto micelles. Solublization may occur in a system consisting of a solvent, an association colloid (a colloid that forms micelles), and at least one other solubilizate. Usage of the term Solubilization is distinct from dissolution because the resulting fluid is a colloidal dispersion involving an association colloid. This suspension is distinct from a true solution, and the amount of the solubilizate in the micellar system can be different (often higher) than the regular solubility of the solubilizate in the solvent. In non-chemical literature and in everyday language, the term "solubilization" is sometimes used in a broader meaning as "to bring to a solution or (non- sedimenting) suspension" by any means, e.g., leaching by a reaction with an acid. Application Micellar solubilization is widely utilized, e.g. in laundry washing using de ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aqueous
An aqueous solution is a solution in which the solvent is water. It is mostly shown in chemical equations by appending (aq) to the relevant chemical formula. For example, a solution of table salt, or sodium chloride (NaCl), in water would be represented as . The word ''aqueous'' (which comes from ''aqua'') means pertaining to, related to, similar to, or dissolved in, water. As water is an excellent solvent and is also naturally abundant, it is a ubiquitous solvent in chemistry. Since water is frequently used as the solvent in experiments, the word solution refers to an aqueous solution, unless the solvent is specified. A ''non-aqueous solution'' is a solution in which the solvent is a liquid, but is not water. (See also Solvent and Inorganic nonaqueous solvent.) Characteristics Substances that are ''hydrophobic'' ('water-fearing') do not dissolve well in water, whereas those that are ''hydrophilic'' ('water-friendly') do. An example of a hydrophilic substance is sodium chlori ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cosmetics
Cosmetics are constituted mixtures of chemical compounds derived from either natural sources, or synthetically created ones. Cosmetics have various purposes. Those designed for personal care and skin care can be used to cleanse or protect the body or skin. Cosmetics designed to enhance or alter one's appearance (makeup) can be used to conceal blemishes, enhance one's natural features (such as the eyebrows and eyelashes), add color to a person's face, or change the appearance of the face entirely to resemble a different person, creature or object. Cosmetics can also be designed to add fragrance to the body. Definition and etymology The word ''cosmetics'' derives from the Greek (), meaning "technique of dress and ornament", from (), "skilled in ordering or arranging" and that from (), meaning "order" and "ornament". Cosmetics are constituted from a mixture of chemical compounds derived from either natural sources, or synthetically created ones. Legal definition T ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |