|
OxFA Process
The OxFA process is a process to produce formic acid from biomass by catalytic oxidation using molecular oxygen or air. Polyoxometalates of the Keggin-type are used as catalysts. OxFA-Process General description Formic acid is obtained by aqueous catalytic partial oxidation of wet biomass. A Keggin structure, Keggin-type polyoxometalate (H5PV2Mo10O40) is used as the homogeneous catalyst to convert sugars, wood, waste paper or cyanobacteria to formic acid and CO2 as the sole byproduct. Yield (chemistry), Yields of up to 53% formic acid can be achieved. It is possible to convert water-insoluble biomass in a suspension with the catalyst. After the reaction, formic acid is separated from the reaction mixture. Depending on the separation process, the formic acid can be further purified or used as it is. The remaining solution of the catalyst, residual formic acid and additives are recycled to the reaction. In this step, solids (e.g. soil from dirty biomass) or unreactive inorganics (e.g ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Formic Acid
Formic acid (), systematically named methanoic acid, is the simplest carboxylic acid, and has the chemical formula HCOOH and structure . It is an important intermediate in chemical synthesis and occurs naturally, most notably in some ants. Esters, salts and the anion derived from formic acid are called formates. Industrially, formic acid is produced from methanol. Natural occurrence In nature, formic acid is found in most ants and in stingless bees of the genus ''Oxytrigona''. Wood ants from the genus ''Formica'' can spray formic acid on their prey or to defend the nest. The puss moth caterpillar (''Cerura vinula'') will spray it as well when threatened by predators. It is also found in the trichomes of stinging nettle (''Urtica dioica''). Apart from that, this acid is incorporated in many fruits such as pineapple (0.21mg per 100g), apple (2mg per 100g) and kiwi (1mg per 100g), as well as in many vegetables, namely onion (45mg per 100g), eggplant (1.34 mg per 100g) and, in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
P-Toluenesulfonic Acid
''p''-Toluenesulfonic acid (PTSA or ''p''TsOH) or tosylic acid (TsOH) is an organic compound with the formula CH3 C6H4 SO3H. It is a white extremely hygroscopic solid that is soluble in water, alcohols, and other polar organic solvents. The CH3C6H4SO2 group is known as the tosyl group and is often abbreviated as Ts or Tos. Most often, TsOH refers to the monohydrate, TsOH.H2O. As with other aryl sulfonic acids, TsOH is a strong organic acid. It is about one million times stronger than benzoic acid. It is one of the few strong acids that is solid and therefore is conveniently weighed and stored. Preparation and uses TsOH is prepared on an industrial scale by the sulfonation of toluene. Common impurities include benzenesulfonic acid and sulfuric acid. TsOH monohydrate contains an amount of water. To estimate the total moisture present as impurity, the Karl Fischer method is used. Impurities can be removed by recrystallization from its concentrated aqueous solution followed by az ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen Storage
Hydrogen storage can be accomplished by several existing methods of holding hydrogen for later use. These include mechanical approaches such as using high pressures and low temperatures, or employing chemical compounds that release H2 upon demand. While large amounts of hydrogen are produced by various industries, it is mostly consumed at the site of production, notably for the synthesis of ammonia. For many years hydrogen has been stored as compressed gas or cryogenic liquid, and transported as such in cylinders, tubes, and cryogenic tanks for use in industry or as propellant in space programs. Interest in using hydrogen for on-board storage of energy in zero-emissions vehicles is motivating the development of new methods of storage, more adapted to this new application. The overarching challenge is the very low boiling point of H2: it boils around 20.268 K (−252.882 °C or −423.188 °F). Achieving such low temperatures requires expending significant energy. Es ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ethyl Acetate
Ethyl acetate ( systematically ethyl ethanoate, commonly abbreviated EtOAc, ETAC or EA) is the organic compound with the formula , simplified to . This colorless liquid has a characteristic sweet smell (similar to pear drops) and is used in glues, nail polish removers, and in the decaffeination process of tea and coffee. Ethyl acetate is the ester of ethanol and acetic acid; it is manufactured on a large scale for use as a solvent. Production and synthesis Ethyl acetate was first synthesized by the Count de Lauraguais in 1759 by distilling a mixture of ethanol and acetic acid. In 2004, an estimated 1.3 million tonnes were produced worldwide. The combined annual production in 1985 of Japan, North America, and Europe was about 400,000 tonnes. The global ethyl acetate market was valued at $3.3 billion in 2018. Ethyl acetate is synthesized in industry mainly via the classic Fischer esterification reaction of ethanol and acetic acid. This mixture converts to the ester in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methylisobutylketone
Methyl isobutyl ketone (MIBK) is the common name for the organic compound 4-methylpentan-2-one, condensed chemical formula (CH3)2CHCH2C(O)CH3. This colourless liquid, a ketone, is used as a solvent for gums, resins, paints, varnishes, lacquers, and nitrocellulose. Production At laboratory scale, MIBK can be produced via a three-step process using acetone as the starting material. Self-condensation, a type of aldol reaction, produces diacetone alcohol, which readily dehydrates to give 4-methylpent-3-en-2-one (commonly, mesityl oxide). Mesityl oxide is then hydrogenated to give MIBK. : Industrially, these three steps are combined. Acetone is treated with a strongly acidic, palladium catalyst-doped cation exchange resin under medium pressure of hydrogen. Several million kilograms are produced annually.Stylianos Sifniades, Alan B. Levy, "Acetone" in ''Ullmann’s Encyclopedia of Industrial Chemistry'', Wiley-VCH, Weinheim, 2005. Uses MIBK is used as a solvent for nitrocellulose ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dibutyl Ether
Dibutyl ether is a chemical compound belonging to the ether family with the molecular formula of . It is colorless, volatile, and flammable liquid and has peculiar ethereal smell. Liquid dibutyl ether is lighter than water. On the other hand, the vapor is heavier than air. It is not soluble in water, but it is soluble in acetone and many other organic solvents. Due to this property, dibutyl ether is used as solvent in various chemical reactions and processes. For example, phenyllithium is commercially available as a ca. 1.8M solution in dibutyl ether. Because of the formation of peroxides, it should be protected from heat, light and air. Synthesis Dibutyl ether is obtained from dehydration of 1-butanol with sulfuric acid as a catalyst and dehydrating agent: :2 → + Industrially, dibutyl ether can be obtained by dehydration of 1-butanol on alumina at 300 °C. Reactions This compound is generally stable to oxidation, reduction, and base. Strong acids like HI and HBr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Partition Coefficient
In the physical sciences, a partition coefficient (''P'') or distribution coefficient (''D'') is the ratio of concentrations of a compound in a mixture of two immiscible solvents at equilibrium. This ratio is therefore a comparison of the solubilities of the solute in these two liquids. The partition coefficient generally refers to the concentration ratio of un-ionized species of compound, whereas the distribution coefficient refers to the concentration ratio of all species of the compound (ionized plus un-ionized). In the chemical and pharmaceutical sciences, both phases usually are solvents. Most commonly, one of the solvents is water, while the second is hydrophobic, such as 1-octanol. Hence the partition coefficient measures how hydrophilic ("water-loving") or hydrophobic ("water-fearing") a chemical substance is. Partition coefficients are useful in estimating the distribution of drugs within the body. Hydrophobic drugs with high octanol-water partition coefficients are m ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
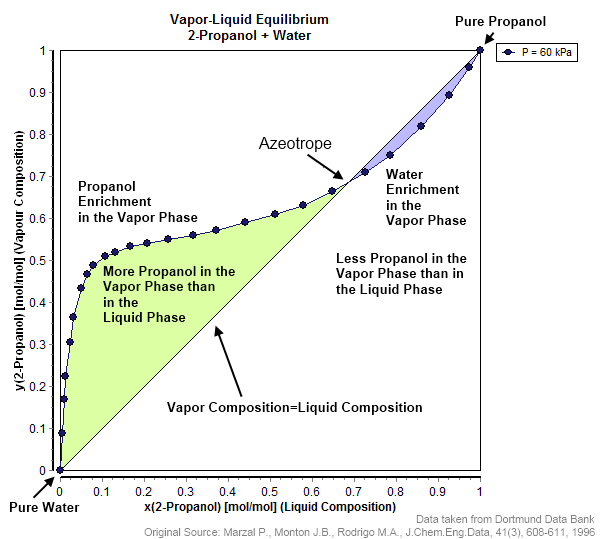
Azeotrope
An azeotrope () or a constant heating point mixture is a mixture of two or more liquids whose proportions cannot be altered or changed by simple distillation.Moore, Walter J. ''Physical Chemistry'', 3rd e Prentice-Hall 1962, pp. 140–142 This happens when an azeotrope is boiled, the vapour has the same proportions of constituents as the unboiled mixture. Because their composition is unchanged by distillation, azeotropes are also called (especially in older texts) ''constant boiling point mixtures''. Some azeotropic mixtures of pairs of compounds are known, and many azeotropes of three or more compounds are also known. In such a case it is not possible to separate the components by fractional distillation and azeotropic distillation is usually used instead. There are two types of azeotropes: minimum boiling azeotrope and maximum boiling azeotrope. A Solution (chemistry), solution that shows greater positive deviation from Raoult's law forms a minimum boiling azeotrope at a speci ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Extraction (chemistry)
Extraction in chemistry is a separation process consisting of the separation of a substance from a matrix. Common examples include '' liquid-liquid extraction'', and ''solid phase extraction''. The distribution of a solute between two phases is an equilibrium condition described by partition theory. This is based on exactly how the analyte moves from the initial solvent into the extracting solvent. The term ''washing'' may also be used to refer to an extraction in which impurities are extracted from the solvent containing the desired compound. Types of extraction * Liquid–liquid extraction * Solid-phase extraction * Acid-base extraction * Supercritical fluid extraction * Ultrasound-assisted extraction * Heat reflux extraction * Mechanochemical-assisted extraction * Maceration * Microwave-assisted extraction * Instant controlled pressure drop extraction (DIC, from the French, Détente instantanée contrôlée) * Perstraction Laboratory applications and examples Liquid ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Distillation
Distillation, or classical distillation, is the process of separation process, separating the components or substances from a liquid mixture by using selective boiling and condensation, usually inside an apparatus known as a still. Dry distillation is the heating of solid materials to produce gaseous products (which may condense into liquids or solids); this may involve chemical changes such as destructive distillation or Cracking (chemistry), cracking. Distillation may result in essentially complete separation (resulting in nearly pure components), or it may be a partial separation that increases the concentration of selected components; in either case, the process exploits differences in the relative volatility of the mixture's components. In Chemical industry, industrial applications, distillation is a unit operation of practically universal importance, but is a physical separation process, not a chemical reaction. An installation used for distillation, especially of distilled ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen Peroxide
Hydrogen peroxide is a chemical compound with the formula . In its pure form, it is a very pale blue liquid that is slightly more viscous than water. It is used as an oxidizer, bleaching agent, and antiseptic, usually as a dilute solution (3%–6% by weight) in water for consumer use, and in higher concentrations for industrial use. Concentrated hydrogen peroxide, or " high-test peroxide", decomposes explosively when heated and has been used as a propellant in rocketry. Hydrogen peroxide is a reactive oxygen species and the simplest peroxide, a compound having an oxygen–oxygen single bond. It decomposes slowly when exposed to light, and rapidly in the presence of organic or reactive compounds. It is typically stored with a stabilizer in a weakly acidic solution in a dark bottle to block light. Hydrogen peroxide is found in biological systems including the human body. Enzymes that use or decompose hydrogen peroxide are classified as peroxidases. Properties The boiling poi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |