|
Organobromides
Organobromine compounds, also called organobromides, are organic compounds that contain carbon bonded to bromine. The most pervasive is the naturally produced bromomethane. One prominent application of synthetic organobromine compounds is the use of polybrominated diphenyl ethers as fire-retardants, and in fact fire-retardant manufacture is currently the major industrial use of the element bromine. A variety of minor organobromine compounds are found in nature, but none are biosynthesized or required by mammals. Organobromine compounds have fallen under increased scrutiny for their environmental impact. General properties Most organobromine compounds, like most organohalide compounds, are relatively nonpolar. Bromine is more electronegative than carbon (2.9 vs 2.5). Consequently, the carbon in a carbon–bromine bond is electrophilic, i.e. alkyl bromides are alkylating agents. Carbon–halogen bond strengths, or bond dissociation energies are of 115, 83.7, 72.1, and 57.6 kc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bromine
Bromine is a chemical element with the symbol Br and atomic number 35. It is the third-lightest element in group 17 of the periodic table (halogens) and is a volatile red-brown liquid at room temperature that evaporates readily to form a similarly coloured vapour. Its properties are intermediate between those of chlorine and iodine. Isolated independently by two chemists, Carl Jacob Löwig (in 1825) and Antoine Jérôme Balard (in 1826), its name was derived from the Ancient Greek (bromos) meaning "stench", referring to its sharp and pungent smell. Elemental bromine is very reactive and thus does not occur as a native element in nature but it occurs in colourless soluble crystalline mineral halide salts, analogous to table salt. In fact, bromine and all the halogens are so reactive that they form bonds in pairs—never in single atoms. While it is rather rare in the Earth's crust, the high solubility of the bromide ion (Br) has caused its accumulation in the oceans. Commercial ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
2-Phenylethylbromide
2-Phenylethyl bromide is an organobromide with the formula C6H5CH2CH2Br. It is a colorless liquid, although older samples appear yellow. Analogous to the preparation of most 1-bromoalkanes, it is prepared by free-radical addition of hydrogen bromide to styrene. These conditions lead to anti-Markovnikov addition, giving the 1-bromo derivatives. Upon reaction with hydrazine, phenelzine Phenelzine, sold under the brand name Nardil, among others, is a non-selective and irreversible monoamine oxidase inhibitor (MAOI) of the hydrazine class which is primarily used as an antidepressant and anxiolytic. Along with tranylcypromine and ... is produced. References Organobromides {{organohalide-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bromotrifluoromethane
Bromotrifluoromethane, commonly known as Halon 1301, R13B1, Halon 13B1 or BTM, is an organic halide with the chemical formula C Br F3. It is used for gaseous fire suppression as a far less toxic alternative to bromochloromethane. Table of physical properties Uses Halon 1301 was developed in a joint venture between the U.S. Army and DuPont in 1954, and introduced as an effective gaseous fire suppression fixed systems agent in the 1960s, and was used around valuable materials, such as aircraft, mainframe computers, and telecommunication switching centers, usually in total flooding systems . It was also widely used in the maritime industry to add a third level of protection should the main and emergency fire pumps become inoperable or ineffective. Halon 1301 was never widely used in portables outside military and spacecraft applications, due to its limited range, and invisible discharge. It does not produce the characteristic white cloud like CO2 and is difficult to direct ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bromoacetic Acid
Bromoacetic acid is the chemical compound with the formula CH2BrCO2H. This colorless solid is a relatively strong alkylating agent. Bromoacetic acid and its esters are widely used building blocks in organic synthesis, for example, in pharmaceutical chemistry. The compound is prepared by bromination of acetic acid, such as by a Hell–Volhard–Zelinsky reaction or using other reagents.. : CH3CO2H + Br2 → CH2BrCO2H + HBr See also * Acetic acid * Chloroacetic acid * Ethyl bromoacetate Ethyl bromoacetate is the chemical compound with the formula CH2BrCO2C2H5. It is the ethyl ester of bromoacetic acid and is prepared in two steps from acetic acid. It is a lachrymator and has a fruity, pungent odor. It is also a highly toxic alky ... References External links The microwave spectrum of bromoacetic acid {{DEFAULTSORT:Bromoacetic Acid Alkylating agents Acetic acids Organobromides ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Halogen
The halogens () are a group in the periodic table consisting of five or six chemically related elements: fluorine (F), chlorine (Cl), bromine (Br), iodine (I), astatine (At), and tennessine (Ts). In the modern IUPAC nomenclature, this group is known as group 17. The word "halogen" means "salt former" (or "salt maker"). When halogens react with metals, they produce a wide range of salts, including calcium fluoride, sodium chloride (common table salt), silver bromide and potassium iodide. The group of halogens is the only periodic table group that contains elements in three of the main states of matter at standard temperature and pressure. All of the halogens form acids when bonded to hydrogen. Most halogens are typically produced from minerals or salts. The middle halogens—chlorine, bromine, and iodine—are often used as disinfectants. Organobromides are the most important class of flame retardants, while elemental halogens are dangerous and can be toxic. History The fl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dehydrohalogenation
In chemistry, dehydrohalogenation is an elimination reaction which removes a hydrogen halide from a substrate. The reaction is usually associated with the synthesis of alkenes, but it has wider applications. Dehydrohalogenation from alkyl halides Traditionally, alkyl halides are substrates for dehydrohalogenations. The alkyl halide must be able to form an alkene, thus halides having no C–H bond on an adjacent carbon are not suitable substrates. Aryl halides are also unsuitable. Upon treatment with strong base, chlorobenzene dehydrohalogenates to give phenol via a benzyne intermediate. Base-promoted reactions to alkenes When treated with a strong base many alkyl chlorides convert to corresponding alkene. It is also called a β-elimination reaction and is a type of elimination reaction. Some prototypes are shown below: :\begin \ce\ &\ce \\ \ce\ &\ce \\ \ce\ &\ce \end Here ethyl chloride reacts with potassium hydroxide, typically in a solvent such as ethanol, giving ethylene. Li ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Markovnikov Rule
In organic chemistry, Markovnikov's rule or Markownikoff's rule describes the outcome of some addition reactions. The rule was formulated by Russian chemist Vladimir Markovnikov in 1870. Explanation The rule states that with the addition of a protic acid HX or other polar reagent to an asymmetric alkene, the acid hydrogen (H) or electropositive part gets attached to the carbon with more hydrogen substituents, and the halide (X) group or electronegative part gets attached to the carbon with more alkyl substituents. This is in contrast to Markovnikov's original definition, in which the rule is stated that the X component is added to the carbon with the fewest hydrogen atoms while the hydrogen atom is added to the carbon with the greatest number of hydrogen atoms. The same is true when an alkene reacts with water in an addition reaction to form an alcohol which involve formation of carbocations. The hydroxyl group (OH) bonds to the carbon that has the greater number of carbon– ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
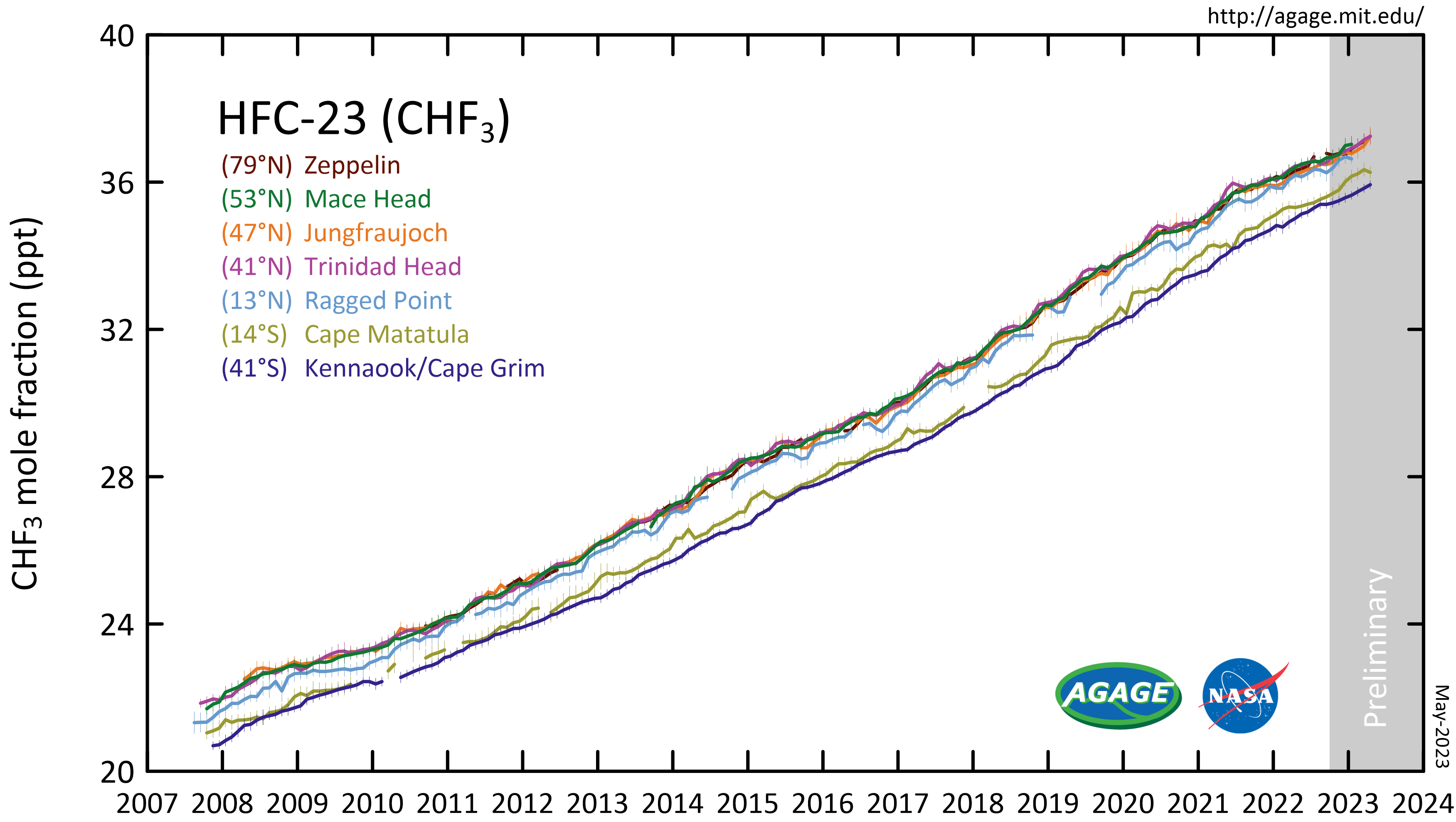
Fluoroform
Trifluoromethane or fluoroform is the chemical compound with the formula CHF3. It is one of the " haloforms", a class of compounds with the formula CHX3 (X = halogen) with C3v symmetry. Fluoroform is used in diverse applications in organic synthesis. It is not an ozone depleter but is a greenhouse gas. Synthesis About 20M kg/y are produced industrially as both a by-product of and precursor to the manufacture of Teflon. It is produced by reaction of chloroform with HF: :CHCl3 + 3 HF → CHF3 + 3 HCl It is also generated biologically in small amounts apparently by decarboxylation of trifluoroacetic acid. Historical Fluoroform was first obtained by Maurice Meslans in the violent reaction of iodoform with dry silver fluoride in 1894. The reaction was improved by Otto Ruff by substitution of silver fluoride by a mixture of mercury fluoride and calcium fluoride. The exchange reaction works with iodoform and bromoform, and the exchange of the first two halogen atoms by ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phosphorus Tribromide
Phosphorus tribromide is a colourless liquid with the formula P Br3. The liquid fumes in moist air due to hydrolysis and has a penetrating odour. It is used in the laboratory for the conversion of alcohols to alkyl bromides. Preparation PBr3 is prepared by treating red phosphorus with bromine. An excess of phosphorus is used in order to prevent formation of PBr5: :2 P + 3 Br2 → 2 PBr3 Because the reaction is highly exothermic, it is often conducted in the presence of a diluent such as PBr3. Reactions Phosphorus tribromide, like PCl3 and PF3, has both properties of a Lewis base and a Lewis acid. For example, with a Lewis acid such as boron tribromide it forms stable 1 :1 adducts such as Br3B · PBr3. At the same time PBr3 can react as an electrophile or Lewis acid in many of its reactions, for example with amines. The most important reaction of PBr3 is with alcohols, where it replaces an OH group with a bromine atom to produce an alkyl bromide. All three bromides ca ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bisphenol-A
Bisphenol A (BPA) is a chemical compound primarily used in the manufacturing of various plastics. It is a colourless solid which is soluble in most common organic solvents, but has very poor solubility in water. BPA is produced on an industrial scale by the condensation of phenol and acetone, and has a global production scale which is expected to reach 10 million tonnes in 2022. BPA's largest single application is as a co-monomer in the production of polycarbonates, which accounts for 65–70% of all BPA production. The manufacturing of epoxy resins and vinyl ester resins account for 25–30% of BPA use. The remaining 5% is used as a major component of several high-performance plastics, and as a minor additive in PVC, polyurethane, thermal paper, and several other materials. It is not a plasticizer, although it is often wrongly labelled as such. The health effects of BPA have been the subject of prolonged public and scientific debate. BPA is a xenoestrogen, exhibiting hormone- ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tetrabromobisphenol-A
Tetrabromobisphenol A (TBBPA) is a brominated flame retardant. The compound is a white solid (not colorless), although commercial samples appear yellow. It is one of the most common flame retardants. Production and use TBBPA is produced by the reaction of bromine with bisphenol A. Most commercial TBBPA products consist of a mixture that differ in the degree of bromination with the formula C15H16−xBrxO2 where x = 1 to 4. Its fire-retarding properties correlate with its bromine content. The annual consumption in Europe has been estimated as 6200 tons in 2004. TBBPA is mainly used as a reactive component of polymers, meaning that it is incorporated into the polymer backbone. It is used to prepare fire-resistant polycarbonates by replacing some bisphenol A. A lower grade of TBBPA is used to prepare epoxy resins, used in printed circuit boards. Toxicity A study was published by the European Food Safety Authority (EFSA) in December 2011 on the exposure of TBBPA and its deriv ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electrophilic Aromatic Substitution
Electrophilic aromatic substitution is an organic reaction in which an atom that is attached to an aromatic system (usually hydrogen) is replaced by an electrophile. Some of the most important electrophilic aromatic substitutions are aromatic nitration, aromatic halogenation, aromatic sulfonation, and alkylation and acylation Friedel–Crafts reaction. Illustrative reactions The most widely practised example of this reaction is the ethylation of benzene. :: Approximately 24,700,000 tons were produced in 1999. (After dehydrogenation and polymerization, the commodity plastic polystyrene is produced.) In this process, acids are used as catalyst to generate the incipient carbocation. Many other electrophilic reactions of benzene are conducted, although on a much smaller scale; they are valuable routes to key intermediates. The nitration of benzene is achieved via the action of the nitronium ion as the electrophile. The sulfonation with fuming sulfuric acid gives benzenesulfonic ac ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |