|
NLRP1
NLRP1 encodes NACHT, LRR, FIIND, CARD domain and PYD domains-containing protein 1 in humans. NLRP1 was the first protein shown to form an inflammasome. Material was copied from this source, which is available under Creative Commons Attribution 4.0 International License NLRP1 is expressed by a variety of cell types, which are predominantly epithelial or hematopoietic. The expression is also seen within glandular epithelial structures including the lining of the small intestine, stomach, airway epithelia and in hairless or glabrous skin. NLRP1 polymorphisms are associated with skin extra-intestinal manifestations in CD. Its highest expression was detected in human skin, in psoriasis and in vitiligo. Polymorphisms of NLRP1 were found in lupus erythematosus and diabetes type 1. Variants of mouse NLRP1 were found to be activated upon N-terminal cleavage by the protease in anthrax lethal factor. Function This gene encodes a member of the Ced-4 family of apoptosis proteins. Ced-family ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
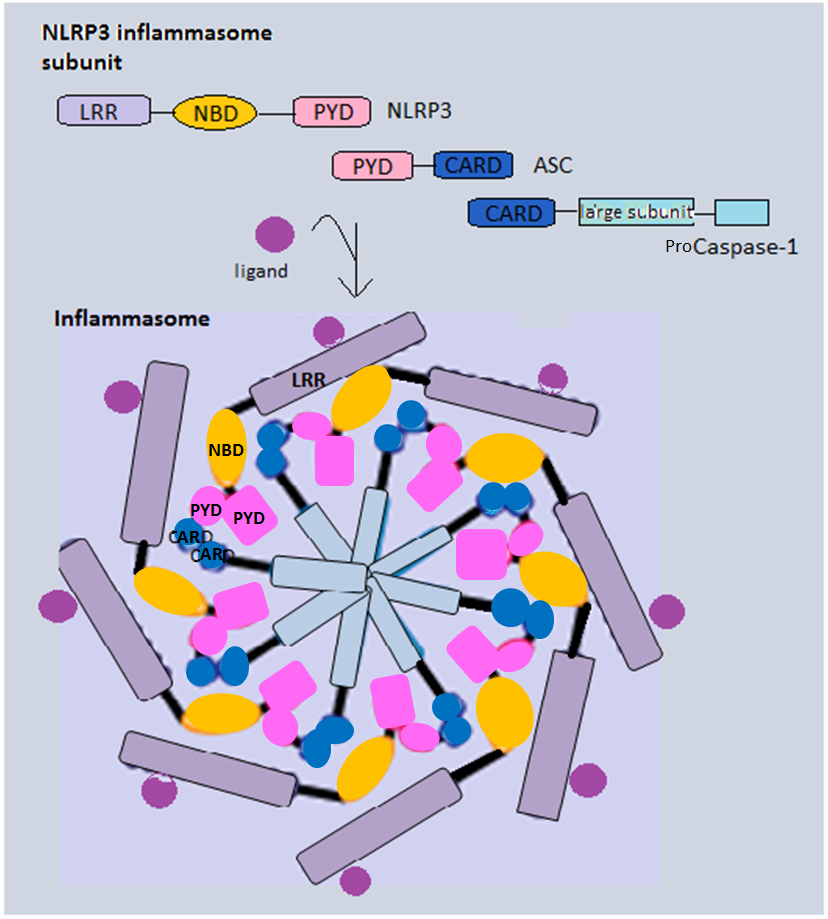
Inflammasome
Inflammasomes are cytosolic multiprotein oligomers of the innate immune system responsible for the activation of inflammatory responses. Activation and assembly of the inflammasome promotes proteolytic cleavage, maturation and secretion of pro-inflammatory cytokines interleukin 1β (IL-1β) and interleukin 18 (IL-18), as well as cleavage of Gasdermin-D. The N-terminal fragment resulting from this cleavage induces a pro-inflammatory form of programmed cell death distinct from apoptosis, referred to as pyroptosis, and is responsible for secretion of the mature cytokines, presumably through the formation of pores in the plasma membrane. Inflammasome activation is initiated by different kinds of cytosolic pattern recognition receptors (PRRs) that respond to either microbe-derived pathogen-associated molecular patterns (PAMPs) or danger-associated molecular patterns (DAMPs) generated by the host cell. Pattern recognition receptors involved in inflammasomes comprise NLRs (nucleoti ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
DPP9
Dipeptidyl peptidase 9 is an enzyme that in humans is encoded by the ''DPP9'' gene. This gene encodes a protein that is a member of the S9B family in clan SC of the serine proteases. The protein has been shown to have post-proline dipeptidyl aminopeptidase activity, cleaving Xaa-Pro dipeptides from the N-termini of proteins. Although the activity of this protein is similar to that of dipeptidyl peptidase 4 (DPP4), it does not appear to be membrane bound. In general, dipeptidyl peptidases appear to be involved in the regulation of the activity of their substrates and have been linked to a variety of diseases including type 2 diabetes, obesity and cancer. Several transcript variants of this gene have been described but not fully characterized. More specifically, DPP9 interacts with the NLRP1 protein and affects the level of activation of the NLPR1 i nflammasome. This function involves binding to a complex of full length NLRP1 and a proinflammatory fragment of NLRP1 after activation ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pyrin Domain
A pyrin domain (PYD, also known as PAAD/DAPIN) is a protein domain and a subclass of protein motif known as the death fold, the 4th and most recently discovered member of the death domain superfamily (DDF). It was originally discovered in the pyrin protein, or marenostrin, encoded by MEFV. The mutation of the MEFV gene is the cause of the disease known as Familial Mediterranean Fever. The domain is encoded in 23 human proteins and at least 31 mouse genes. Proteins containing a pyrin domain are frequently involved in programmed cell death processes including pyroptosis and apoptosis. Proteins that possess a pyrin domain interact with the pyrin domains in other proteins to form of multi-protein complexes called inflammasomes and to trigger downstream immune responses. Structure Pyrin domains are a ~90 amino acid motif present only at the N-terminus of proteins. The core is made of highly conserved hydrophobic residues surrounded by five or six alpha helices with α1→2 linkage ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bruno Reversade
Bruno Reversade (born 1978) is an American human geneticist and developmental biologist . He is a Director of the Institute of Molecular and Cellular Biology and the Genome Institute of Singapore at A*STAR (Singapore) and holds several faculty positions at other universities. Reversade is known for identifying mutated genes that cause Mendelian diseases, for his research on the genetics of identical twins and for the characterizations of novel hormones. Early life and education Bruno Reversade was born in 1974 into a French-American family. He was raised in Grenoble (France) and Washington, D.C. ( US). Bruno Reversade studied at the University Joseph Fourier, Pierre and Marie Curie University and UCLA. Scientific career Reversade became interested in developmental biology in 1997 when studying at the University of Western Ontario (Canada) under the tutelage of Greg Kelly. He earned his master's degree at the Pasteur Institute (Paris, France), where he studied head development ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
APAF1
Apoptotic protease activating factor 1, also known as APAF1, is a human homolog of ''C. elegans'' CED-4 gene. Function The protein was identified in the lab of Xiaodong Wang as an activator of caspase-3 in the presense of cytochromeC and dATP. This gene encodes a cytoplasmic protein that forms one of the central hubs in the apoptosis regulatory network. This protein contains (from the N terminal) a caspase recruitment domain (CARD), an ATPase domain (NB-ARC), few short helical domains and then several copies of the WD40 repeat domain. Upon binding cytochrome c and dATP, this protein forms an oligomeric apoptosome. The apoptosome binds and cleaves Procaspase-9 protein, releasing its mature, activated form. The precise mechanism for this reaction is still debated though work published by Guy Salvesen suggests that the apoptosome may induce caspase-9 dimerization and subsequent autocatalysis. Activated caspase-9 stimulates the subsequent caspase cascade that commits the cell to ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Caspase 9
Caspase-9 is an enzyme that in humans is encoded by the CASP9 gene. It is an initiator caspase, critical to the apoptotic pathway found in many tissues. Caspase-9 homologs have been identified in all mammals for which they are known to exist, such as ''Mus musculus'' and ''Pan troglodytes''. Caspase-9 belongs to a family of caspases, cysteine-aspartic proteases involved in apoptosis and cytokine signalling. Apoptotic signals cause the release of cytochrome c from mitochondria and activation of apaf-1 (apoptosome), which then cleaves the pro-enzyme of caspase-9 into the active dimer form. Regulation of this enzyme occurs through phosphorylation by an allosteric inhibitor, inhibiting dimerization and inducing a conformational change. Correct caspase-9 function is required for apoptosis, leading to the normal development of the central nervous system. Caspase-9 has multiple additional cellular functions that are independent of its role in apoptosis. Nonapoptotic roles of caspase ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Caspase Recruitment Domain
Caspase recruitment domains, or caspase activation and recruitment domains (CARDs), are interaction motifs found in a wide array of proteins, typically those involved in processes relating to inflammation and apoptosis. These domains mediate the formation of larger protein complexes via direct interactions between individual CARDs. CARD domains are found on a strikingly wide range of proteins, including helicases, kinases, mitochondrial proteins, caspases, and other cytoplasmic factors. Basic features CARD domains are a subclass of protein motif known as the death fold, which features an arrangement of six to seven antiparallel alpha helices with a hydrophobic core and an outer face composed of charged residues. Other motifs in this class include the pyrin domain (PYD), death domain (DD), and death effector domain (DED), all of which also function primarily in regulation of apoptosis and inflammatory responses. In apoptosis CARD domains were originally characterized based on ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Immune System
The immune system is a network of biological processes that protects an organism from diseases. It detects and responds to a wide variety of pathogens, from viruses to parasitic worms, as well as cancer cells and objects such as wood splinters, distinguishing them from the organism's own healthy tissue. Many species have two major subsystems of the immune system. The innate immune system provides a preconfigured response to broad groups of situations and stimuli. The adaptive immune system provides a tailored response to each stimulus by learning to recognize molecules it has previously encountered. Both use molecules and cells to perform their functions. Nearly all organisms have some kind of immune system. Bacteria have a rudimentary immune system in the form of enzymes that protect against virus infections. Other basic immune mechanisms evolved in ancient plants and animals and remain in their modern descendants. These mechanisms include phagocytosis, antimicrobial pe ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
3C-like Protease
The 3C-like protease (3CLpro) or main protease (Mpro), formally known as C30 endopeptidase or 3-chymotrypsin-like protease, is the main protease found in coronaviruses. It cleaves the coronavirus polyprotein at eleven conserved sites. It is a cysteine protease and a member of the PA clan of proteases. It has a cysteine-histidine catalytic dyad at its active site and cleaves a Gln–(Ser/Ala/Gly) peptide bond. The Enzyme Commission refers to this family as SARS coronavirus main proteinase (Mpro; ). The 3CL protease corresponds to coronavirus nonstructural protein 5 (nsp5). The "3C" in the common name refers to the 3C protease (3Cpro) which is a homologous protease found in picornaviruses. Function The 3C-like protease is able to catalytically cleave a peptide bond between a glutamine at position P1 and a small amino acid (serine, alanine, or glycine) at position P1'. The SARS coronavirus 3CLpro can for instance self-cleave the following peptides: The protease is impor ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Map Kinase
A mitogen-activated protein kinase (MAPK or MAP kinase) is a type of protein kinase that is specific to the amino acids serine and threonine (i.e., a serine/threonine-specific protein kinase). MAPKs are involved in directing cellular responses to a diverse array of stimuli, such as mitogens, osmotic stress, heat shock and proinflammatory cytokines. They regulate cell functions including proliferation, gene expression, differentiation, mitosis, cell survival, and apoptosis. MAP kinases are found in eukaryotes only, but they are fairly diverse and encountered in all animals, fungi and plants, and even in an array of unicellular eukaryotes. MAPKs belong to the CMGC (CDK/MAPK/GSK3/CLK) kinase group. The closest relatives of MAPKs are the cyclin-dependent kinases (CDKs). Discovery The first mitogen-activated protein kinase to be discovered was ERK1 (MAPK3) in mammals. Since ERK1 and its close relative ERK2 (MAPK1) are both involved in growth factor signaling, the family ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Immune Response
An immune response is a reaction which occurs within an organism for the purpose of defending against foreign invaders. These invaders include a wide variety of different microorganisms including viruses, bacteria, parasites, and fungi which could cause serious problems to the health of the host organism if not cleared from the body. There are two distinct aspects of the immune response, the innate and the adaptive, which work together to protect against pathogens. The innate branch—the body's first reaction to an invader—is known to be a non-specific and quick response to any sort of pathogen. Components of the innate immune response include physical barriers like the skin and mucous membranes, immune cells such as neutrophils, macrophages, and monocytes, and soluble factors including cytokines and complement. On the other hand, the adaptive branch is the body's immune response which is catered against specific antigens and thus, it takes longer to activate the components involv ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |