|
Nierenstein Reaction
The Nierenstein reaction is an organic reaction describing the conversion of an acid chloride into a haloketone with diazomethane. It is an insertion reaction in that the methylene group from the diazomethane is inserted into the carbon-chlorine bond of the acid chloride. Reaction mechanism The reaction proceeds through a diazonium salt intermediate formed by nucleophilic acyl substitution of the chloride with diazomethyl anion. The chloride then displaces the diazo group in an SN2 reaction, with N2 as the leaving group. If excess diazomethane is present during the reaction, it can act as a base, abstracting a hydrogen from the diazonium-salt intermediate. The result is a neutral diazoketone, which does not react with the chloride. Instead, the byproduct, diazonium-methyl from the other diazomethane molecule, can be attacked by the chloride to produce chloromethane. The unreactive diazoketone can be re-activated and reacted by treatment with hydrogen chloride to give the normal ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Maximilian Nierenstein
Maximilian Nierenstein (also known as Moses Max Nierenstein or Max Nierenstein; 1877–1946) was a professor of biochemistry at the University of Bristol. He is known for the Nierenstein reaction, an organic reaction describing the conversion of an acid chloride into an haloketone with diazomethane. In 1912, Polish biochemist Casimir Funk isolated a complex of micronutrients and proposed the complex be named "vitamine" (a portmanteau of "vital amine"), a name reportedly suggested by friend Max Nierenstein. He also studied natural phenols and tannins found in different plant species. He showed in 1945 that luteic acid, a molecule present in the myrobalanitannin, a tannin found in the fruit of ''Terminalia chebula'', is an intermediary compound in the synthesis of ellagic acid. Working with Arthur George Perkin, he prepared ellagic acid from algarobilla and certain other fruits in 1905. He suggested its formation from galloyl-glycine by ''Penicillium'' in 1915. Tannase is an enz ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chloromethane
Chloromethane, also called methyl chloride, Refrigerant-40, R-40 or HCC 40, is an organic compound with the chemical formula . One of the haloalkanes, it is a colorless, odorless, flammable gas. Methyl chloride is a crucial reagent in industrial chemistry, although it is rarely present in consumer products, and was formerly utilized as a refrigerant. Occurrence Chloromethane is an abundant organohalogen, anthropogenic or natural, in the atmosphere. Marine Laboratory cultures of marine phytoplankton (''Phaeodactylum tricornutum'', ''Phaeocystis'' sp., ''Thalassiosira weissflogii'', ''Chaetoceros calcitrans'', ''Isochrysis'' sp., ''Porphyridium'' sp., ''Synechococcus'' sp., ''Tetraselmis'' sp., ''Prorocentrum'' sp., and ''Emiliana huxleyi'') produce CH3Cl, but in relatively insignificant amounts. An extensive study of 30 species of polar macroalgae revealed the release of significant amounts of CH3Cl in only ''Gigartina skottsbergii'' and ''Gymnogongrus antarcticus''. Biogenesis ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Arndt–Eistert Reaction
In organic chemistry, the Arndt–Eistert reaction is the conversion of a carboxylic acid to its homologue. Named for the German chemists Fritz Arndt (1885–1969) and Bernd Eistert (1902–1978), the method entails treating an acid chlorides with diazomethane. It is a popular method of producing β-amino acids from α-amino acids. Conditions Aside from the acid chloride substrate, three reagents are required: diazomethane, water, and a metal catalyst. Each has been well investigated. The diazomethane is required in excess so as to react with the HCl formed previously. Not taking diazomethane in excess results in HCl reacting with the diazoketone to form chloromethyl ketone and N2. Mild conditions allow this reaction to take place while not affecting complex or reducible groups in the reactant-acid. The reaction requires the presence of a nucleophile (water). A metal catalyst is required. Usually Ag2O is chosen but other metals and even light effect the reac ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
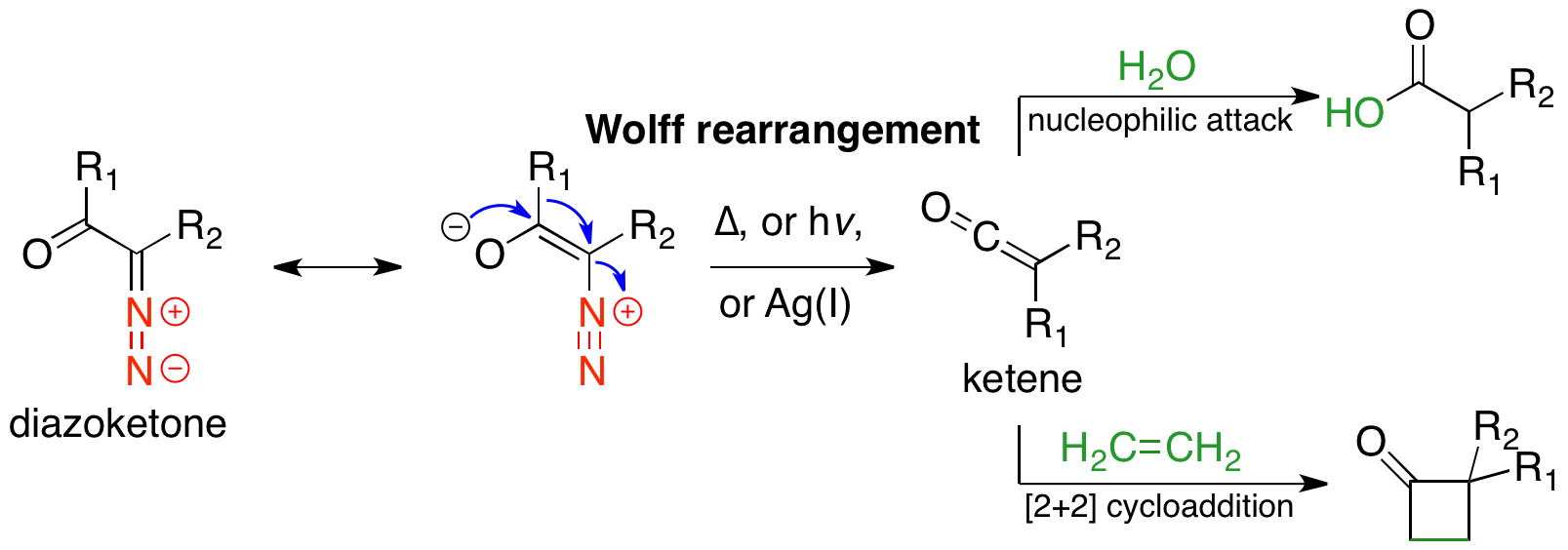
Wolff Rearrangement
The Wolff rearrangement is a reaction in organic chemistry in which an α-diazocarbonyl compound is converted into a ketene by loss of dinitrogen with accompanying 1,2-rearrangement. The Wolff rearrangement yields a ketene as an intermediate product, which can undergo nucleophilic attack with weakly acidic nucleophiles such as water, alcohols, and amines, to generate carboxylic acid derivatives or undergo +2cycloaddition reactions to form four-membered rings. The mechanism of the Wolff rearrangement has been the subject of debate since its first use. No single mechanism sufficiently describes the reaction, and there are often competing concerted and carbene-mediated pathways; for simplicity, only the textbook, concerted mechanism is shown below. The reaction was discovered by Ludwig Wolff in 1902. The Wolff rearrangement has great synthetic utility due to the accessibility of α-diazocarbonyl compounds, variety of reactions from the ketene intermediate, and stereochemical ret ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Curtius Rearrangement
The Curtius rearrangement (or Curtius reaction or Curtius degradation), first defined by Theodor Curtius in 1885, is the thermal decomposition of an acyl azide to an isocyanate with loss of nitrogen gas. The isocyanate then undergoes attack by a variety of nucleophiles such as water, alcohols and amines, to yield a primary amine, carbamate or urea derivative respectively. Several reviews have been published. Preparation of acyl azide The acyl azide is usually made from the reaction of acid chlorides or anydrides with sodium azide or trimethylsilyl azide. Acyl azides are also obtained from treating acylhydrazines with nitrous acid. Alternatively, the acyl azide can be formed by the direct reaction of a carboxylic acid with diphenylphosphoryl azide (DPPA). Reaction mechanism It was believed that the Curtius rearrangement was a two-step processes, with the loss of nitrogen gas forming an acyl nitrene, followed by migration of the R-group to give the isocyanate. However, recent ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dioxane
1,4-Dioxane () is a heterocyclic organic compound, classified as an ether. It is a colorless liquid with a faint sweet odor similar to that of diethyl ether. The compound is often called simply dioxane because the other dioxane isomers ( 1,2- and 1,3-) are rarely encountered. Dioxane is used as a solvent for a variety of practical applications as well as in the laboratory, and also as a stabilizer for the transport of chlorinated hydrocarbons in aluminum containers.Wisconsin Department of Health Services (20131,4-Dioxane Fact Sheet Publication 00514. Accessed 2016-11-12. Synthesis Dioxane is produced by the acid-catalysed dehydration of diethylene glycol, which in turn is obtained from the hydrolysis of ethylene oxide. In 1985, the global production capacity for dioxane was between 11,000 and 14,000 tons. In 1990, the total U.S. production volume of dioxane was between 5,250 and 9,150 tons. Structure The dioxane molecule is centrosymmetric, meaning that it adopts a chai ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Benzoyl
In organic chemistry, benzoyl (, ) is the functional group with the formula C6H5CO-. It can be viewed as benzaldehyde missing one hydrogen. The term "benzoyl" should not be confused with benzyl, which has the formula C6H5CH2. The benzoyl group is given the symbol "Bz". Benzyl is commonly abbreviated "Bn". Sources Benzoyl chloride is a favored source of benzoyl groups, being used to prepare benzoyl ketones, benzamides (benzoyl amides), and benzoate esters. The source of many naturally occurring benzoyl compounds is the thioester benzoyl-CoA. Irradiation of benzil generates benzoyl radicals, which have the formula PhCO. Benzoyl compounds Many ketones contain the benzoyl group. They have the formula C6H5CO–R, an important example being benzophenone. Benzoyl esters and amides are common in organic chemistry. The esters are used as a protecting groups in organic synthesis, which can be easily removed by hydrolysis in dilute basic solution. Benzoyl-β-D-glucoside is a natural su ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organic Syntheses
''Organic Syntheses'' is a peer-reviewed scientific journal that was established in 1921. It publishes detailed and checked procedures for the synthesis of organic compounds. A unique feature of the review process is that all of the data and experiments reported in an article must be successfully repeated in the laboratory of a member of the editorial board as a check for reproducibility prior to publication. The journal is published by Organic Syntheses, Inc., a non-profit corporation. An annual print version is published by John Wiley & Sons on behalf of Organic Syntheses, Inc. History Prior to World War I, work on synthetic organic chemistry in the United States had been quite limited, and most of the reagents used in laboratories had to be imported from Europe. When export stoppages and trade embargoes cut off this source, Clarence Derick, a professor of chemistry at University of Illinois at Urbana-Champaign, began an effort to synthesize these needed chemicals in industr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nierenstein Reaction Mechanism2
Maximilian Nierenstein (also known as Moses Max Nierenstein or Max Nierenstein; 1877–1946) was a professor of biochemistry at the University of Bristol. He is known for the Nierenstein reaction, an organic reaction describing the conversion of an acid chloride into an haloketone with diazomethane. In 1912, Polish biochemist Casimir Funk isolated a complex of micronutrients and proposed the complex be named "vitamine" (a portmanteau of "vital amine"), a name reportedly suggested by friend Max Nierenstein. He also studied natural phenols and tannins found in different plant species. He showed in 1945 that luteic acid, a molecule present in the myrobalanitannin, a tannin found in the fruit of ''Terminalia chebula'', is an intermediary compound in the synthesis of ellagic acid. Working with Arthur George Perkin, he prepared ellagic acid from algarobilla and certain other fruits in 1905. He suggested its formation from galloyl-glycine by ''Penicillium'' in 1915. Tannase is an enzy ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen Chloride
The compound hydrogen chloride has the chemical formula and as such is a hydrogen halide. At room temperature, it is a colourless gas, which forms white fumes of hydrochloric acid upon contact with atmospheric water vapor. Hydrogen chloride gas and hydrochloric acid are important in technology and industry. Hydrochloric acid, the aqueous solution of hydrogen chloride, is also commonly given the formula HCl. Reactions Hydrogen chloride is a diatomic molecule, consisting of a hydrogen atom H and a chlorine atom Cl connected by a polar covalent bond. The chlorine atom is much more electronegative than the hydrogen atom, which makes this bond polar. Consequently, the molecule has a large dipole moment with a negative partial charge (δ−) at the chlorine atom and a positive partial charge (δ+) at the hydrogen atom. In part because of its high polarity, HCl is very soluble in water (and in other polar solvents). Upon contact, and HCl combine to form hydronium cations and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |