|
Naphthalenetetracarboxylic Dianhydride
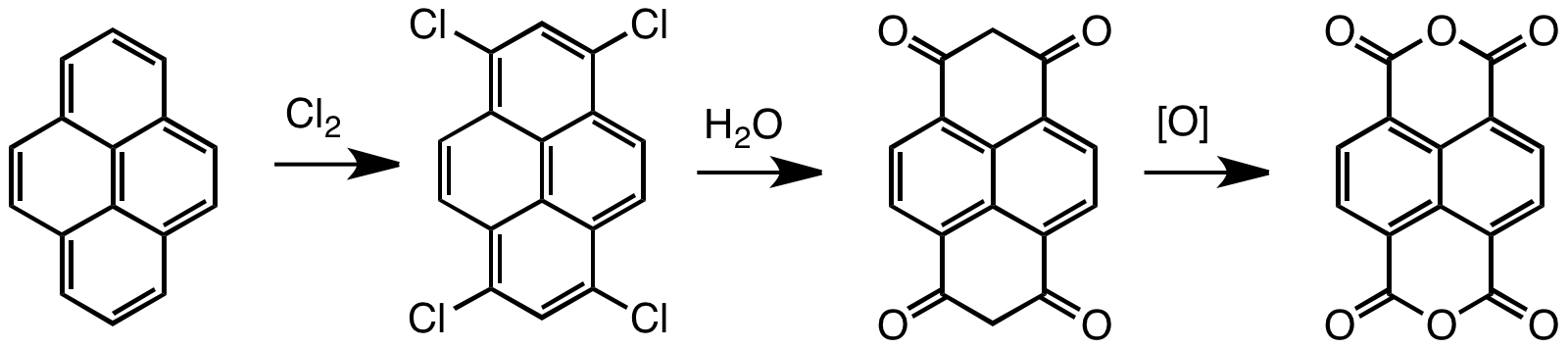
Naphthalenetetracarboxylic dianhydride (NTDA) is an organic compound related to naphthalene. The compound is a beige solid. NTDA is most commonly used as a precursor to naphthalenediimides (NDIs) (such as napthalenetetracarboxylic diimide), a family of compounds with many uses. Synthesis and structure Naphthalenetetracarboxylic dianhydride is prepared by oxidation of pyrene. Typical oxidants are chromic acid and chlorine. The unsaturated tetrachloride hydrolyzes to enols that tautomerize to the bis-dione, which in turn can be oxidized to the tetracarboxylic acid.F. Röhrscheid "Carboxylic Acids, Aromatic" in ''Ullmann's Encyclopedia of Industrial Chemistry'', Wiley-VCH, Weinheim, 2012. Naphthalene diimides Symmetrical naphthalene diimides are synthesized by the condensation reaction of primary amines and the dianhydride. Unsymmetrical derivatives, i.e. those derived from two different amines, are obtained by hydrolysis of one of the two anhydride groups prior to the conde ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organic Compound
In chemistry, organic compounds are generally any chemical compounds that contain carbon-hydrogen or carbon-carbon bonds. Due to carbon's ability to catenate (form chains with other carbon atoms), millions of organic compounds are known. The study of the properties, reactions, and syntheses of organic compounds comprise the discipline known as organic chemistry. For historical reasons, a few classes of carbon-containing compounds (e.g., carbonate salts and cyanide salts), along with a few other exceptions (e.g., carbon dioxide, hydrogen cyanide), are not classified as organic compounds and are considered inorganic. Other than those just named, little consensus exists among chemists on precisely which carbon-containing compounds are excluded, making any rigorous definition of an organic compound elusive. Although organic compounds make up only a small percentage of Earth's crust, they are of central importance because all known life is based on organic compounds. Living t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Radical Anion
In organic chemistry, a radical anion is a free radical species that carries a negative charge. Radical anions are encountered in organic chemistry as reduced derivatives of polycyclic aromatic compounds, e.g. sodium naphthenide. An example of a non-carbon radical anion is the superoxide anion, formed by transfer of one electron to an oxygen molecule. Radical anions are typically indicated by M^. Polycyclic radical anions Many aromatic compounds can undergo one-electron reduction by alkali metals. The electron is transferred from the alkali metal ion to an unoccupied antibonding p-p п* orbital of the aromatic molecule. This transfer is usually only energetically favorable if the aprotic solvent efficiently solvates the alkali metal ion. Effective solvents are those that bind to the alkali metal cation: diethyl ether < < [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ligand
In coordination chemistry, a ligand is an ion or molecule (functional group) that binds to a central metal atom to form a coordination complex. The bonding with the metal generally involves formal donation of one or more of the ligand's electron pairs, often through Lewis bases. The nature of metal–ligand bonding can range from covalent to ionic. Furthermore, the metal–ligand bond order can range from one to three. Ligands are viewed as Lewis bases, although rare cases are known to involve Lewis acidic "ligands". Metals and metalloids are bound to ligands in almost all circumstances, although gaseous "naked" metal ions can be generated in a high vacuum. Ligands in a complex dictate the reactivity of the central atom, including ligand substitution rates, the reactivity of the ligands themselves, and redox. Ligand selection requires critical consideration in many practical areas, including bioinorganic and medicinal chemistry, homogeneous catalysis, and environmental chemi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phenylenediamine
Phenylenediamine may refer to: * ''o''-phenylenediamine or OPD, a chemical compound C6H4(NH2)2 * ''m''-phenylenediamine or MPD, a chemical compound C6H4(NH2)2 * ''p''-phenylenediamine or PPD, a chemical compound C6H4(NH2)2 * ''N,N''-dimethyl-''p''-phenylenediamine or DMPD * ''N,N,N′,N′''-tetramethyl-''p''-phenylenediamine or TMPD, used in microbiology * ''N'',''N''-diethyl-p-phenylenediamine or DPD {{Chemistry index Diamines ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Perinone
Perinone is a class of organic compounds. The parent compound has two isomers, each of which are useful pigments. It is prepared from naphthalenetetracarboxylic dianhydride by condensation with ''o''-phenylenediamine. The two Isomers of perinone are useful pigments. The trans isomer is called Pigment Orange 43 ("PO43", ) and the cis isomer is called Pigment Red 194 ("PR194", ).K. Hunger. W. Herbst "Pigments, Organic" in ''Ullmann's Encyclopedia of Industrial Chemistry'', Wiley-VCH, Weinheim, 2012. Like some structurally related compounds perinone is also an organic semiconductor Organic semiconductors are solids whose building blocks are pi-bonded molecules or polymers made up by carbon and hydrogen atoms and – at times – heteroatoms such as nitrogen, sulfur and oxygen. They exist in the form of molecular crystals or ....{{cite journal , first1=W. James , last1=Feast , first2=Richard J. , last2=Peace , first3=Ian C. , last3=Sage , first4=Emma L. , last4=Wood , title=Po ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Intercalation (biochemistry)
In biochemistry, intercalation is the insertion of molecules between the planar bases of deoxyribonucleic acid (DNA). This process is used as a method for analyzing DNA and it is also the basis of certain kinds of poisoning. There are several ways molecules (in this case, also known as ''ligands'') can interact with DNA. Ligands may interact with DNA by covalently binding, electrostatically binding, or intercalating. Intercalation occurs when ligands of an appropriate size and chemical nature fit themselves in between base pairs of DNA. These ligands are mostly polycyclic, aromatic, and planar, and therefore often make good nucleic acid stains. Intensively studied DNA intercalators include berberine, ethidium bromide, proflavine, daunomycin, doxorubicin, and thalidomide. DNA intercalators are used in chemotherapeutic treatment to inhibit DNA replication in rapidly growing cancer cells. Examples include doxorubicin (adriamycin) and daunorubicin (both of which are used in treat ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Catenane
In macromolecular chemistry, a catenane () is a mechanically interlocked molecular architecture consisting of two or more interlocked macrocycles, i.e. a molecule containing two or more intertwined rings. The interlocked rings cannot be separated without breaking the covalent bonds of the macrocycles. They are conceptually related to other mechanically interlocked molecular architectures, such as rotaxanes, molecular knots or molecular Borromean rings. Recently the terminology "mechanical bond" has been coined that describes the connection between the macrocycles of a catenane. Catenanes have been synthesised in two different ways: statistical synthesis and template-directed synthesis. Synthesis There are two primary approaches to the organic synthesis of catenanes. The first is to simply perform a ring-closing reaction with the hope that some of the rings will form around other rings giving the desired catenane product. This so-called "statistical approach" led to the fi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Rotaxanes
In chemistry, a rotaxane () is a mechanically interlocked molecular architecture consisting of a dumbbell-shaped molecule which is threaded through a macrocycle (see graphical representation). The two components of a rotaxane are kinetically trapped since the ends of the dumbbell (often called ''stoppers'') are larger than the internal diameter of the ring and prevent dissociation (unthreading) of the components since this would require significant distortion of the covalent bonds. Much of the research concerning rotaxanes and other mechanically interlocked molecular architectures, such as catenanes, has been focused on their efficient synthesis or their utilization as artificial molecular machines. However, examples of rotaxane substructure have been found in naturally occurring peptides, including: cystine knot peptides, cyclotides or lasso-peptides such as microcin J25. Synthesis The earliest reported synthesis of a rotaxane in 1967 relied on the statistical probability t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Crown Ether
In organic chemistry, crown ethers are cyclic chemical compounds that consist of a ring containing several ether groups (). The most common crown ethers are cyclic oligomers of ethylene oxide, the repeating unit being ethyleneoxy, i.e., . Important members of this series are the tetramer (''n'' = 4), the pentamer (''n'' = 5), and the hexamer (''n'' = 6). The term "crown" refers to the resemblance between the structure of a crown ether bound to a cation, and a crown sitting on a person's head. The first number in a crown ether's name refers to the number of atoms in the cycle, and the second number refers to the number of those atoms that are oxygen. Crown ethers are much broader than the oligomers of ethylene oxide; an important group are derived from catechol. Crown ethers strongly bind certain cations, forming complexes. The oxygen atoms are well situated to coordinate with a cation located at the interior of the ring, whereas the exterior of t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Charge-transfer Complexes
In chemistry, a charge-transfer (CT) complex or electron-donor-acceptor complex describes a type of supramolecular assembly of two or more molecules or ions. The assembly consists of two molecules that self-attract through electrostatic forces, i.e., one has at least partial negative charge and the partner has partial positive charge, referred to respectively as the electron acceptor and electron donor. In some cases, the degree of charge transfer is "complete", such that the CT complex can be classified as a salt. In other cases, the charge-transfer association is weak, and the interaction can be disrupted easily by polar solvents. Examples Electron donor-acceptor complexes A number of organic compounds form charge-transfer complex, which are often described as electron-donor-acceptor complexes (EDA complexes). Typical acceptors are nitrobenzenes or tetracyanoethylene. The strength of their interaction with electron donors correlates with the ionization potentials of the c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Supramolecular Chemistry
Supramolecular chemistry refers to the branch of chemistry concerning chemical systems composed of a discrete number of molecules. The strength of the forces responsible for spatial organization of the system range from weak intermolecular forces, electrostatic charge, or hydrogen bonding to strong covalent bonding, provided that the electronic coupling strength remains small relative to the energy parameters of the component. While traditional chemistry concentrates on the covalent bond, supramolecular chemistry examines the weaker and reversible non-covalent interactions between molecules. These forces include hydrogen bonding, metal coordination, hydrophobic forces, van der Waals forces, pi–pi interactions and electrostatic effects. Important concepts advanced by supramolecular chemistry include molecular self-assembly, molecular folding, molecular recognition, host–guest chemistry, mechanically-interlocked molecular architectures, and dynamic covalent chemistry. The st ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electron Withdrawing Group
In chemistry, an electron-withdrawing group (EWG) is a substituent that has some of the following kinetic and thermodynamic implications: *with regards to electron transfer, electron-withdrawing groups enhance the oxidizing power tendency of the appended species. Tetracyanoethylene is an oxidant because the alkene is appended to four cyano substituents, which are electron-withdrawing. *with regards to acid-base reactions, acids with electron-withdrawing groups species have low acid dissociation constants. For EWG's attached to benzene, this effect is described by the Hammett equation, which allows EWGs to be discussed quantitatively. *with regards to nucleophilic substitution reactions, electron-withdrawing groups are susceptible to attack by weak nucleophiles. For example, compared to chlorobenzene, chlorodinitrobenzene is susceptible to reactions that displace chloride.{{cite journal , author=J. F. Bunnett, R. M. Conner, doi=10.15227/orgsyn.040.0034, title=2,4-Dinitroiodobe ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
4-3D-balls.png)



