|
NK012
NK012 is a 'nanodevice' formulation of SN-38 (an irinotecan metabolite). NK012 is an SN-38-releasing polymeric micelle constructed by covalently attaching SN-38 to the block copolymer PEG-PGlu, followed by self-assembly of amphiphilic block copolymers in aqueous media. It has completed phase II clinical trials for triple-negative breast cancer and relapsed small cell lung cancer. In 2016, Nippon Kayaku received orphan drug designation for NK012 from the US FDA.Nippon Kayaku Announces FDA Orphan Drug Designation to New Polymeric micelle Anti-Cancer Drug NK012 for Small Cell Lung Cancer. (2016). Evaluategroup.com. Retrieved 24 November 2017, from http://www.evaluategroup.com/Universal/View.aspx?type=Story&id=644017 This means that if it is approved in the United States, Nippon Kayaku will be entitled to 7 years of market exclusivity. This is intended to incentivize future development, but does not mean the drug has been approved. See also * Nanomedicine * Targeted drug delivery Tar ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Triple-negative Breast Cancer
Triple-negative breast cancer (TNBC) is any breast cancer that lacks or show low levels of estrogen receptor (ER), progesterone receptor (PR) and human epidermal growth factor receptor 2 (HER2) overexpression and/or gene amplification (i.e. the tumor is negative on all three tests giving the name ''triple-negative''). Triple-negative is sometimes used as a surrogate term for basal-like. Triple-negative breast cancer comprises 15–20% of all breast cancer cases and affects more young women or women with a mutation in the BRCA1 gene than other breast cancers. Triple-negative breast cancers comprise a very heterogeneous group of cancers. TNBC is the most challenging breast cancer type to treat. Hormone therapy that is used for other breast cancers does not work for TNBC. In its early stages, the cancer is typically treated through surgery, radiation and chemotherapy. In later stages where surgery is not possible or the cancer has spread from the initial localised area, treatment i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
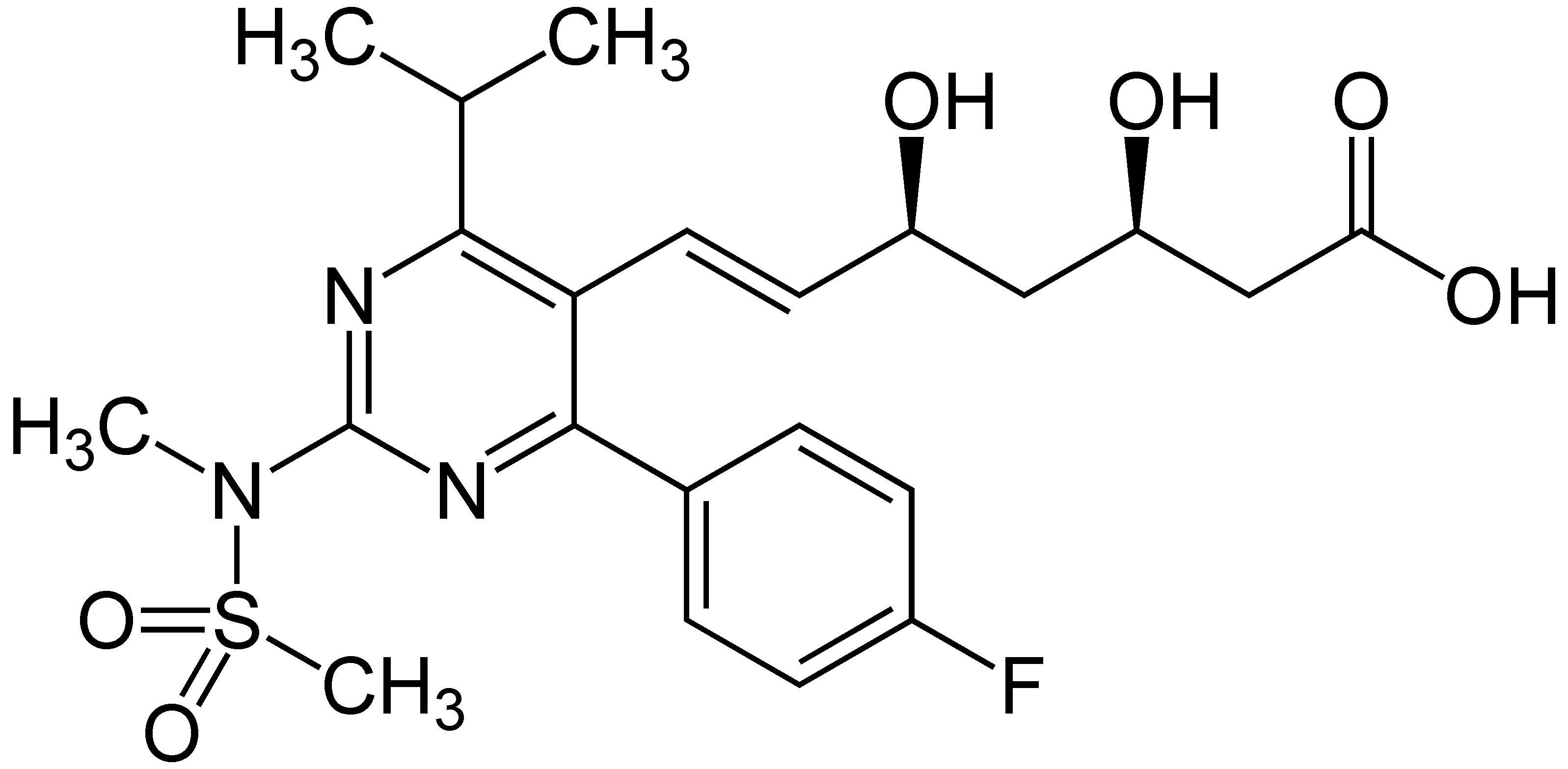
SN-38
SN-38 is an antineoplastic drug. It is the active metabolite of irinotecan (an analog of camptothecin - a topoisomerase I inhibitor) but has 1000 times more activity than irinotecan itself. In vitro cytotoxicity assays show that the potency of SN-38 relative to irinotecan varies from 2- to 2000-fold.http://labeling.pfizer.com/ShowLabeling.aspx?id=533 SN38 is formed via hydrolysis of irinotecan by carboxylesterases and metabolized via glucuronidation by UGT1A1. The variant of UGT1A1 in ~10% of Caucasians which leads to poor metabolism of SN-38 predicts irinotecan toxicity, as it is then less easily excreted from the body in its SN-38 glucuronide form. SN-38 and its glucuronide are lost into the bile and intestines. It can cause the symptoms of diarrhoea and myelosuppression experienced by ~25% of the patients administered irinotecan. Interactive pathway map See also * NK012, a nanodevice formulation of SN-38 *Sacituzumab govitecan Sacituzumab govitecan, sold under the brand ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Irinotecan
Irinotecan, sold under the brand name Camptosar among others, is a medication used to treat colon cancer, and small cell lung cancer. For colon cancer it is used either alone or with fluorouracil. For small cell lung cancer it is used with cisplatin. It is given intravenously. Common side effects include diarrhea, vomiting, bone marrow suppression, hair loss, shortness of breath, and fever. Other severe side effects include blood clots, colon inflammation, and allergic reactions. Those with two copies of the UGT1A1*28 gene variant are at higher risk for side effects. Use during pregnancy can result in harm to the baby. Irinotecan is a topoisomerase inhibitor—it blocks the topoisomerase I enzyme, resulting in DNA damage and cell death. Irinotecan was approved for medical use in the United States in 1996. It is on the World Health Organization's List of Essential Medicines. It is made from the natural compound camptothecin which is found in the Chinese ornamental tree ''Campt ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polymeric Micelle
Targeted drug delivery, sometimes called smart drug delivery, is a method of delivering medication to a patient in a manner that increases the concentration of the medication in some parts of the body relative to others. This means of delivery is largely founded on nanomedicine, which plans to employ nanoparticle-mediated drug delivery in order to combat the downfalls of conventional drug delivery. These nanoparticles would be loaded with drugs and targeted to specific parts of the body where there is solely diseased tissue, thereby avoiding interaction with healthy tissue. The goal of a targeted drug delivery system is to prolong, localize, target and have a protected drug interaction with the diseased tissue. The conventional drug delivery system is the absorption of the drug across a biological membrane, whereas the ''targeted release system'' releases the drug in a dosage form. The advantages to the targeted release system is the reduction in the frequency of the dosages take ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Block Copolymer
In polymer chemistry, a copolymer is a polymer derived from more than one species of monomer. The polymerization of monomers into copolymers is called copolymerization. Copolymers obtained from the copolymerization of two monomer species are sometimes called ''bipolymers''. Those obtained from three and four monomers are called ''terpolymers'' and ''quaterpolymers'', respectively. Copolymers can be characterized by a variety of techniques such as NMR spectroscopy and size-exclusion chromatography to determine the molecular size, weight, properties, and composition of the material. Commercial copolymers include acrylonitrile butadiene styrene (ABS), styrene/butadiene co-polymer (SBR), nitrile rubber, styrene-acrylonitrile, styrene-isoprene-styrene (SIS) and ethylene-vinyl acetate, all of which are formed by chain-growth polymerization. Another production mechanism is step-growth polymerization, which is used to produce the nylon-12/6/66 copolymer of nylon 12, nylon 6 and nylon 66 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Amphiphilic
An amphiphile (from the Greek αμφις amphis, both, and φιλíα philia, love, friendship), or amphipath, is a chemical compound possessing both hydrophilic (''water-loving'', polar) and lipophilic (''fat-loving'') properties. Such a compound is called amphiphilic or amphipathic. Common amphiphilic substances are soaps, detergents, and lipoproteins. The phospholipid amphiphiles are the major structural component of cell membranes. Amphiphiles are the basis for a number of areas of research in chemistry and biochemistry, notably that of lipid polymorphism. Organic compounds containing hydrophilic groups at both ends of the molecule are called bolaamphiphilic. The micelles they form in the aggregate are prolate. Structure The lipophilic group is typically a large hydrocarbon moiety, such as a long chain of the form CH3(CH2)n, with n > 4. The hydrophilic group falls into one of the following categories: # charged groups #* anionic. Examples, with the lipophilic part of the m ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Small Cell Lung Cancer
Small-cell carcinoma is a type of highly malignant cancer that most commonly arises within the lung, although it can occasionally arise in other body sites, such as the cervix, prostate, and gastrointestinal tract. Compared to non-small cell carcinoma, small cell carcinoma has a shorter doubling time, higher growth fraction, and earlier development of metastases. Extensive stage small cell lung cancer is classified as a rare disorder. Ten-year relative survival rate is 3.5%; however, women have a higher survival rate, 4.3%, and men lower, 2.8%. Survival can be higher or lower based on a combination of factors including stage, age, gender and race. Types of SCLC Small-cell lung carcinoma has long been divided into two clinicopathological stages, termed ''limited stage'' (LS) and ''extensive stage'' (ES). The stage is generally determined by the presence or absence of metastases, whether or not the tumor appears limited to the thorax, and whether or not the entire tumor burden wit ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nippon Kayaku
(Japan Pharmaceuticals) is a Japanese company that was founded in 1916 as the first industrial explosives manufacturer in Japan under the company name Nippon Kayaku Seizo Co., Ltd.. Its main business areas are functional chemicals, pharmaceuticals, safety systems and agrochemicals. It has 8 plants and 4 laboratories in Japan. It also has subsidiaries in different countries around the world. It is listed on the Nikkei 225. History The company was established in 1916 to produce explosives for the construction sector. The firm then diversified into chemical dyes and pharmaceuticals in the interwar period. By 1950, Nippon Kayaku had established itself as the ninth leading pharmaceutical firm in Japan. The launch of its anticancer drug, bleomycin, strengthened the firm's pharmaceutical business. Products Major products made by Nippon Kayaku are: epoxy resins, UV-curing type resins, functional films, colors for inkjet printers, catalysts, dyes pharmaceuticals, pharmaceutical API and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Orphan Drug
An orphan drug is a pharmaceutical agent developed to treat medical conditions which, because they are so rare, would not be profitable to produce without government assistance. The conditions are referred to as orphan diseases. The assignment of orphan status to a disease and to drugs developed to treat it is a matter of public policy in many countries and has yielded medical breakthroughs that might not otherwise have been achieved, due to the economics of drug research and development. In the U.S. and the EU, it is easier to gain marketing approval for an orphan drug. There may be other financial incentives, such as an extended period of exclusivity, during which the producer has sole rights to market the drug. All are intended to encourage development of drugs which would otherwise lack sufficient profit motive to attract corporate research budgets and personnel. Definition According to the US Food and Drug Administration (FDA), an orphan drug is defined as one "intended for ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
US FDA
The United States Food and Drug Administration (FDA or US FDA) is a federal agency of the Department of Health and Human Services. The FDA is responsible for protecting and promoting public health through the control and supervision of food safety, tobacco products, caffeine products, dietary supplements, prescription and over-the-counter pharmaceutical drugs (medications), vaccines, biopharmaceuticals, blood transfusions, medical devices, electromagnetic radiation emitting devices (ERED), cosmetics, animal foods & feed and veterinary products. The FDA's primary focus is enforcement of the Federal Food, Drug, and Cosmetic Act (FD&C), but the agency also enforces other laws, notably Section 361 of the Public Health Service Act, as well as associated regulations. Much of this regulatory-enforcement work is not directly related to food or drugs, but involves such things as regulating lasers, cellular phones, and condoms, as well as control of disease in contexts varying from h ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nanomedicine
Nanomedicine is the medical application of nanotechnology. Nanomedicine ranges from the medical applications of nanomaterials and BioBrick, biological devices, to Nanoelectronics, nanoelectronic biosensors, and even possible future applications of molecular nanotechnology such as Molecular machine#Biological, biological machines. Current problems for nanomedicine involve understanding the issues related to Nanotoxicology, toxicity and Implications of nanotechnology, environmental impact of Nanomaterials, nanoscale materials (materials whose structure is on the scale of nanometers, i.e. billionths of a meter). Functionalities can be added to nanomaterials by interfacing them with biological molecules or structures. The size of nanomaterials is similar to that of most biological molecules and structures; therefore, nanomaterials can be useful for both in vivo and in vitro biomedical research and applications. Thus far, the integration of nanomaterials with biology has led to the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |

.jpg)