|
Mineral Hydration
In chemistry, mineral hydration is an inorganic chemical reaction which adds water to the crystal structure of a mineral, usually creating a new mineral, usually called a ''hydrate''. In geological terms, the process of mineral hydration is known as ''retrograde alteration'' and is a process occurring in retrograde metamorphism. It commonly accompanies metasomatism and is often a feature of wall rock alteration around ore bodies. Hydration of minerals occurs generally in concert with hydrothermal circulation which may be driven by tectonic or igneous activity. Processes There are two main ways in which minerals hydrate. One is conversion of an oxide to a double hydroxide, as with the hydration of calcium oxide—CaO—to calcium hydroxide—Ca(OH)2, the other is with the incorporation of water molecules directly into the crystalline structure of a new mineral. The later process is exhibited in the hydration of feldspars to clay minerals, garnet to chlorite, or kyanite to m ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemistry
Chemistry is the scientific study of the properties and behavior of matter. It is a natural science that covers the elements that make up matter to the compounds made of atoms, molecules and ions: their composition, structure, properties, behavior and the changes they undergo during a reaction with other substances. Chemistry also addresses the nature of chemical bonds in chemical compounds. In the scope of its subject, chemistry occupies an intermediate position between physics and biology. It is sometimes called the central science because it provides a foundation for understanding both basic and applied scientific disciplines at a fundamental level. For example, chemistry explains aspects of plant growth ( botany), the formation of igneous rocks ( geology), how atmospheric ozone is formed and how environmental pollutants are degraded ( ecology), the properties of the soil on the moon ( cosmochemistry), how medications work (pharmacology), and how to collect DNA ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Muscovite
Muscovite (also known as common mica, isinglass, or potash mica) is a hydrated phyllosilicate mineral of aluminium and potassium with formula K Al2(Al Si3 O10)( F,O H)2, or ( KF)2( Al2O3)3( SiO2)6( H2O). It has a highly perfect basal cleavage yielding remarkably thin laminae (sheets) which are often highly elastic. Sheets of muscovite 5 meters × 3 meters (16.5 feet × 10 feet) have been found in Nellore, India. Muscovite has a Mohs hardness of 2–2.25 parallel to the 01face, 4 perpendicular to the 01and a specific gravity of 2.76–3. It can be colorless or tinted through grays, browns, greens, yellows, or (rarely) violet or red, and can be transparent or translucent. It is anisotropic and has high birefringence. Its crystal system is monoclinic. The green, chromium-rich variety is called fuchsite; mariposite is also a chromium-rich type of muscovite. Muscovite is the most common mica, found in granites, pegmatites, gneisses, and schists, and as a contact metamorphic roc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydromagnesite
Hydromagnesite is a hydrated magnesium carbonate mineral with the formula Mg5(CO3)4(OH)2·4H2O. It generally occurs associated with the weathering products of magnesium containing minerals such as serpentine or brucite. It occurs as incrustations and vein or fracture fillings in ultramafic rocks and serpentinites, and occurs in hydrothermally altered dolomite and marble. Hydromagnesite commonly appears in caves as speleothems and " moonmilk", deposited from water that has seeped through magnesium rich rocks. It is the most common cave carbonate after calcite and aragonite. The mineral thermally decomposes, over a temperature range of approximately 220 °C to 550 °C, releasing water and carbon dioxide leaving a magnesium oxide residue. Hydromagnesite was first described in 1836 for an occurrence in Hoboken, New Jersey. Stromatolites in an alkaline ( pH greater than 9) freshwater lake ( Salda Gölü) in southern Turkey are made of hydromagnesite precipitated by diatom ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbonate
A carbonate is a salt of carbonic acid (H2CO3), characterized by the presence of the carbonate ion, a polyatomic ion with the formula . The word ''carbonate'' may also refer to a carbonate ester, an organic compound containing the carbonate group C(=O)(O–)2. The term is also used as a verb, to describe carbonation: the process of raising the concentrations of carbonate and bicarbonate ions in water to produce carbonated water and other carbonated beverageseither by the addition of carbon dioxide gas under pressure or by dissolving carbonate or bicarbonate salts into the water. In geology and mineralogy, the term "carbonate" can refer both to carbonate minerals and carbonate rock (which is made of chiefly carbonate minerals), and both are dominated by the carbonate ion, . Carbonate minerals are extremely varied and ubiquitous in chemically precipitated sedimentary rock. The most common are calcite or calcium carbonate, CaCO3, the chief constituent of limestone (as ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Goethite
Goethite (, ) is a mineral of the diaspore group, consisting of iron(III) oxide-hydroxide, specifically the "α" polymorph. It is found in soil and other low-temperature environments such as sediment. Goethite has been well known since ancient times for its use as a pigment (brown ochre). Evidence has been found of its use in paint pigment samples taken from the caves of Lascaux in France. It was first described in 1806 based on samples found in the Hollertszug Mine in Herdorf, Germany. The mineral was named after the German polymath and poet Johann Wolfgang von Goethe (1749–1832). Composition Goethite is an iron oxyhydroxide containing ferric iron. It is the main component of rust and bog iron ore. Goethite's hardness ranges from 5.0 to 5.5 on the Mohs Scale, and its specific gravity varies from 3.3 to 4.3. The mineral forms prismatic needle-like crystals ("needle ironstone") but is more typically massive. Feroxyhyte and lepidocrocite are both polymorphs of the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Brucite
Brucite is the mineral form of magnesium hydroxide, with the chemical formula Mg( OH)2. It is a common alteration product of periclase in marble; a low-temperature hydrothermal vein mineral in metamorphosed limestones and chlorite schists; and formed during serpentinization of dunites. Brucite is often found in association with serpentine, calcite, aragonite, dolomite, magnesite, hydromagnesite, artinite, talc and chrysotile. It adopts a layered CdI2-like structure with hydrogen-bonds between the layers. Discovery Brucite was first described in 1824 by François Sulpice Beudant and named for the discoverer, American mineralogist, Archibald Bruce (1777–1818). A fibrous variety of brucite is called nemalite. It occurs in fibers or laths, usually elongated along 010 but sometimes 120 crystalline directions. Occurrence A notable location in the U.S. is Wood's Chrome Mine, Cedar Hill Quarry, Lancaster County, Pennsylvania. Yellow, white and blue Brucite with a botryoidal habi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrothermal
Hydrothermal circulation in its most general sense is the circulation of hot water (Ancient Greek ὕδωρ, ''water'',Liddell, H.G. & Scott, R. (1940). ''A Greek-English Lexicon. revised and augmented throughout by Sir Henry Stuart Jones. with the assistance of. Roderick McKenzie.'' Oxford: Clarendon Press. and θέρμη, ''heat'' ). Hydrothermal circulation occurs most often in the vicinity of sources of heat within the Earth's crust. In general, this occurs near volcanic activity, but can occur in the shallow to mid crust along deeply penetrating fault irregularities or in the deep crust related to the intrusion of granite, or as the result of orogeny or metamorphism. Seafloor hydrothermal circulation Hydrothermal circulation in the oceans is the passage of the water through mid-oceanic ridge systems. The term includes both the circulation of the well-known, high-temperature vent waters near the ridge crests, and the much-lower-temperature, diffuse flow of water through sed ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phyllosilicate
Silicate minerals are rock-forming minerals made up of silicate groups. They are the largest and most important class of minerals and make up approximately 90 percent of Earth's crust. In mineralogy, silica (silicon dioxide, ) is usually considered a silicate mineral. Silica is found in nature as the mineral quartz, and its polymorphs. On Earth, a wide variety of silicate minerals occur in an even wider range of combinations as a result of the processes that have been forming and re-working the crust for billions of years. These processes include partial melting, crystallization, fractionation, metamorphism, weathering, and diagenesis. Living organisms also contribute to this geologic cycle. For example, a type of plankton known as diatoms construct their exoskeletons ("frustules") from silica extracted from seawater. The frustules of dead diatoms are a major constituent of deep ocean sediment, and of diatomaceous earth. General structure A silicate mineral is generally an ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
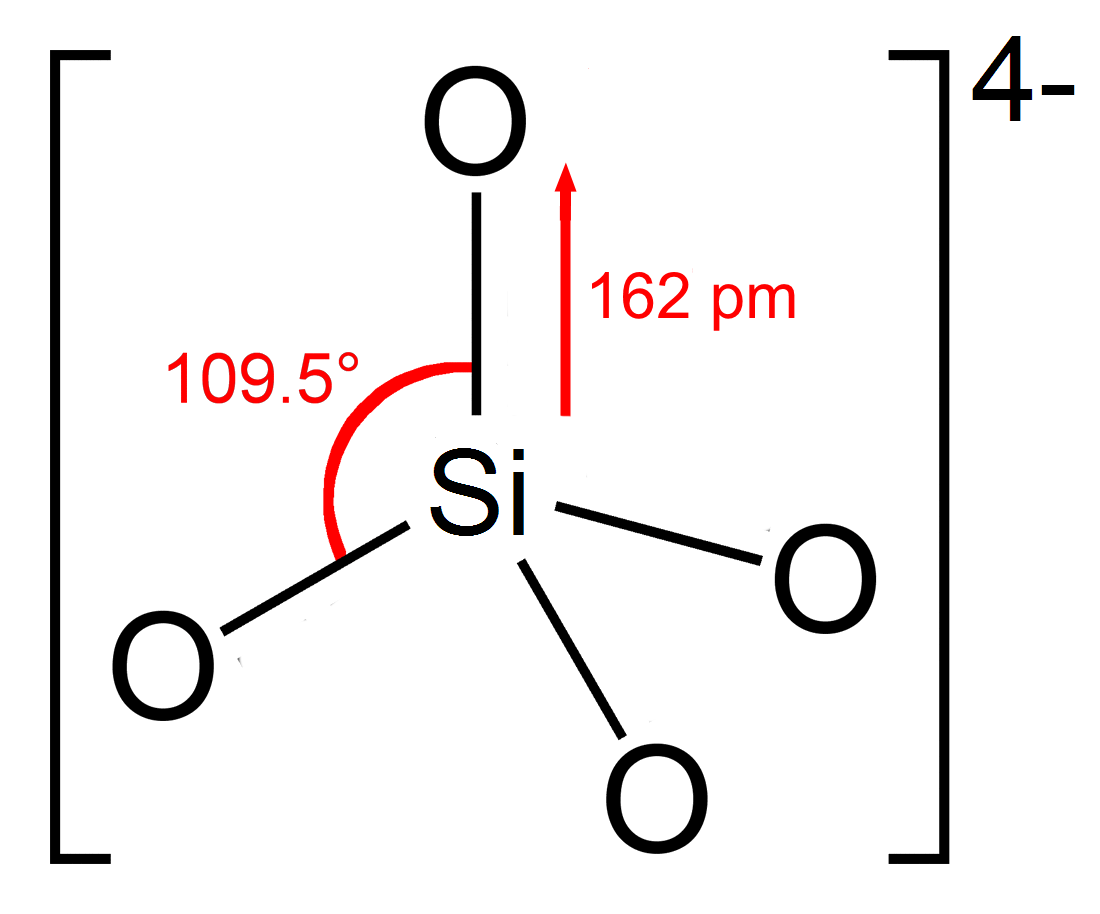
Silicate
In chemistry, a silicate is any member of a family of polyatomic anions consisting of silicon and oxygen, usually with the general formula , where . The family includes orthosilicate (), metasilicate (), and pyrosilicate (, ). The name is also used for any salt of such anions, such as sodium metasilicate; or any ester containing the corresponding chemical group, such as tetramethyl orthosilicate. The name "silicate" is sometimes extended to any anions containing silicon, even if they do not fit the general formula or contain other atoms besides oxygen; such as hexafluorosilicate .Most commonly, silicates are encountered as silicate minerals. For diverse manufacturing, technological, and artistic needs, silicates are versatile materials, both natural (such as granite, gravel, and garnet) and artificial (such as Portland cement, ceramics, glass, and waterglass). Structural principles In all silicates, silicon atom occupies the center of an idealized tetrahedro ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Affinity
In chemical physics and physical chemistry, chemical affinity is the electronic property by which dissimilar chemical species are capable of forming chemical compounds. Chemical affinity can also refer to the tendency of an atom or compound to combine by chemical reaction with atoms or compounds of unlike composition. History Early theories The idea of ''affinity'' is extremely old. Many attempts have been made at identifying its origins. The majority of such attempts, however, except in a general manner, end in futility since "affinities" lie at the basis of all magic, thereby pre-dating science. Physical chemistry, however, was one of the first branches of science to study and formulate a "theory of affinity". The name ''affinitas'' was first used in the sense of chemical relation by German philosopher Albertus Magnus near the year 1250. Later, those as Robert Boyle, John Mayow, Johann Glauber, Isaac Newton, and Georg Stahl put forward ideas on elective affinity in attem ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Portland Cement
Portland cement is the most common type of cement in general use around the world as a basic ingredient of concrete, mortar, stucco, and non-specialty grout. It was developed from other types of hydraulic lime in England in the early 19th century by Joseph Aspdin, and is usually made from limestone. It is a fine powder, produced by heating limestone and clay minerals in a kiln to form clinker, grinding the clinker, and adding 2 to 3 percent of gypsum. Several types of portland cement are available. The most common, called ordinary portland cement (OPC), is grey, but white Portland cement is also available. Its name is derived from its resemblance to Portland stone which was quarried on the Isle of Portland in Dorset, England. It was named by Joseph Aspdin who obtained a patent for it in 1824. His son William Aspdin is regarded as the inventor of "modern" portland cement due to his developments in the 1840s. The low cost and widespread availability of the limestone, sh ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cement
A cement is a binder, a chemical substance used for construction that sets, hardens, and adheres to other materials to bind them together. Cement is seldom used on its own, but rather to bind sand and gravel (aggregate) together. Cement mixed with fine aggregate produces mortar for masonry, or with sand and gravel, produces concrete. Concrete is the most widely used material in existence and is behind only water as the planet's most-consumed resource. Cements used in construction are usually inorganic, often lime or calcium silicate based, which can be characterized as hydraulic or the less common non-hydraulic, depending on the ability of the cement to set in the presence of water (see hydraulic and non-hydraulic lime plaster). Hydraulic cements (e.g., Portland cement) set and become adhesive through a chemical reaction between the dry ingredients and water. The chemical reaction results in mineral hydrates that are not very water-soluble and so are quite durable ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |





.jpg)