|
Marsh Test
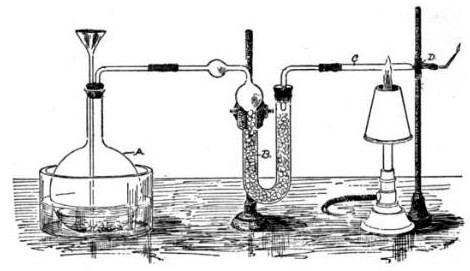
The Marsh test is a highly sensitive method in the detection of arsenic, especially useful in the field of forensic toxicology when arsenic was used as a poison. It was developed by the chemist James Marsh and first published in 1836. The method continued to be used, with improvements, in forensic toxicology until the 1970s. Arsenic, in the form of white arsenic trioxide , was a highly favored poison, being odourless, easily incorporated into food and drink, and before the advent of the Marsh test, untraceable in the body. In France, it came to be known as ' ("inheritance powder"). For the untrained, arsenic poisoning will have symptoms similar to cholera. Precursor methods The first breakthrough in the detection of arsenic poisoning was in 1775 when Carl Wilhelm Scheele discovered a way to change arsenic trioxide to garlic-smelling arsine gas (AsH3), by treating it with nitric acid (HNO3) and combining it with zinc. :As2O3 + 6 Zn + 12 HNO3 → 2 AsH3 + 6 Zn(NO3)2 + 3 H2O ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Marsh Test Apparatus
A marsh is a wetland that is dominated by Herbaceous plant, herbaceous rather than woody plant species.Keddy, P.A. 2010. Wetland Ecology: Principles and Conservation (2nd edition). Cambridge University Press, Cambridge, UK. 497 p Marshes can often be found at the edges of lakes and streams, where they form a transition between the aquatic and terrestrial ecosystems. They are often dominated by Poaceae, grasses, Juncaceae, rushes or reeds. If woody plants are present they tend to be low-growing shrubs, and the marsh is sometimes called a Carr (landform), carr. This form of vegetation is what differentiates marshes from other types of wetland such as swamps, which are dominated by trees, and mires, which are wetlands that have accumulated deposits of acidic peat. Marshes provide habitats for many kinds of Invertebrate, invertebrates, fish, Amphibian, amphibians, Anseriformes, waterfowl and Aquatic mammal, aquatic mammals. This Productivity (ecology), biological productivity mea ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen Sulfide
Hydrogen sulfide is a chemical compound with the formula . It is a colorless chalcogen-hydride gas, and is poisonous, corrosive, and flammable, with trace amounts in ambient atmosphere having a characteristic foul odor of rotten eggs. The underground mine gas term for foul-smelling hydrogen sulfide-rich gas mixtures is ''stinkdamp''. Swedish chemist Carl Wilhelm Scheele is credited with having discovered the chemical composition of purified hydrogen sulfide in 1777. The British English spelling of this compound is hydrogen sulphide, a spelling no longer recommended by the Royal Society of Chemistry or the International Union of Pure and Applied Chemistry. Hydrogen sulfide is toxic to humans and most other animals by inhibiting cellular respiration in a manner similar to hydrogen cyanide. When it is inhaled or it or its salts are ingested in high amounts, damage to organs occurs rapidly with symptoms ranging from breathing difficulties to convulsions and death. Despite this, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tulle
Tulle (; ) is a commune in central France. It is the third-largest town in the former region of Limousin and is the capital of the department of Corrèze, in the region of Nouvelle-Aquitaine. Tulle is also the episcopal see of the Roman Catholic Diocese of Tulle. Stretching over more than three kilometres in the narrow and tortuous Corrèze valley, Tulle spreads its old quarters on the hillside overlooking the river, while the Notre-Dame cathedral emerges from the heart of the town. Known sometimes as "the town on seven hills", Tulle rose to prominence through the development of its manufacturing sector. Geography Tulle is the third largest town in Limousin, behind Limoges and Brive-la-Gaillarde. It is situated in a very deep part of the river Corrèze valley, at its confluence with several of its tributaries, the Solane and the Céronne on the right bank, and the Saint-Bonnette and the Montane on the left bank. It stretches along a very narrow strip several kilometre ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Trivalent
In chemistry, the valence (US spelling) or valency (British spelling) of an element is the measure of its combining capacity with other atoms when it forms chemical compounds or molecules. Description The combining capacity, or affinity of an atom of a given element is determined by the number of hydrogen atoms that it combines with. In methane, carbon has a valence of 4; in ammonia, nitrogen has a valence of 3; in water, oxygen has a valence of 2; and in hydrogen chloride, chlorine has a valence of 1. Chlorine, as it has a valence of one, can be substituted for hydrogen. Phosphorus has a valence of 5 in phosphorus pentachloride, . Valence diagrams of a compound represent the connectivity of the elements, with lines drawn between two elements, sometimes called bonds, representing a saturated valency for each element. The two tables below show some examples of different compounds, their valence diagrams, and the valences for each element of the compound. Modern definitio ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polysulfide
Polysulfides are a class of chemical compounds containing chains of sulfur atoms. There are two main classes of polysulfides: inorganic and organic. Among the inorganic polysulfides, there are ones which contain anions, which have the general formula . These anions are the conjugate bases of the hydrogen polysulfides . Organic polysulfides generally have the formulae , where R = alkyl or aryl. Polysulfide salts and complexes The alkali metal polysulfides arise by treatment of a solution of sulfide, e.g. sodium sulfide, with elemental sulfur: : In some cases, these anions have been obtained as organic salts, which are soluble in organic solvents. The energy released in the reaction of sodium and elemental sulfur is the basis of battery technology. The sodium–sulfur battery and the lithium–sulfur battery require high temperatures to maintain liquid polysulfide and -conductive membranes that are unreactive toward sodium, sulfur, and sodium sulfide. Polysulfides are ligands ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bismuthine
Bismuthine (IUPAC name: bismuthane) is the chemical compound with the formula BiH3. As the heaviest analogue of ammonia (a pnictogen hydride), BiH3 is unstable, decomposing to bismuth metal well below 0 °C. This compound adopts the expected pyramidal structure with H–Bi–H angles of around 90°. The term ''bismuthine'' may also refer to a member of the family of organobismuth(III) species having the general formula , where R is an organic substituent. For example, Bi(CH3)3 is ''trimethylbismuthine''. Preparation and properties BiH3 is prepared by the redistribution of methylbismuthine (BiH2Me):Holleman, A. F.; Wiberg, E. "Inorganic Chemistry" Academic Press: San Diego, 2001.. :3 BiH2Me → 2 BiH3 + BiMe3 The required BiH2Me, which is also thermally unstable, is generated by reduction of methylbismuth dichloride, BiCl2Me with LiAlH4. As suggested by the behavior of SbH3, BiH3 is unstable and decomposes to its constituent elements according to the following equa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bismuth
Bismuth is a chemical element with the symbol Bi and atomic number 83. It is a post-transition metal and one of the pnictogens, with chemical properties resembling its lighter group 15 siblings arsenic and antimony. Elemental bismuth occurs naturally, and its sulfide and oxide forms are important commercial ores. The free element is 86% as dense as lead. It is a brittle metal with a silvery-white color when freshly produced. Surface oxidation generally gives samples of the metal a somewhat rosy cast. Further oxidation under heat can give bismuth a vividly iridescent appearance due to thin-film interference. Bismuth is both the most diamagnetic element and one of the least thermally conductive metals known. Bismuth was long considered the element with the highest atomic mass whose nuclei do not spontaneously decay. However, in 2003 it was discovered to be extremely weakly radioactive. The metal's only primordial isotope, bismuth-209, experiences alpha decay at such a m ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Hypochlorite
Sodium hypochlorite (commonly known in a dilute solution as bleach) is an inorganic chemical compound with the formula NaOCl (or NaClO), comprising a sodium cation () and a hypochlorite anion (or ). It may also be viewed as the sodium salt of hypochlorous acid. The anhydrous compound is unstable and may decompose explosively. It can be crystallized as a pentahydrate ·5, a pale greenish-yellow solid which is not explosive and is stable if kept refrigerated. Sodium hypochlorite is most often encountered as a pale greenish-yellow dilute solution referred to as liquid bleach, which is a household chemical widely used (since the 18th century) as a disinfectant or a bleaching agent. In solution, the compound is unstable and easily decomposes, liberating chlorine, which is the active principle of such products. Sodium hypochlorite is the oldest and still most important chlorine-based bleach. Its corrosive properties, common availability, and reaction products make it a signif ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Stibine
Stibine (IUPAC name: stibane) is a chemical compound with the formula SbH3. A pnictogen hydride, this colourless, highly toxic gas is the principal covalent hydride of antimony, and a heavy analogue of ammonia. The molecule is pyramidal with H–Sb–H angles of 91.7° and Sb–H distances of 170.7 pm (1.707 Å). This gas has an offensive smell like hydrogen sulfide (rotten eggs). Preparation SbH3 is generally prepared by the reaction of Sb3+ sources with H− equivalents: :2 Sb2O3 + 3 LiAlH4 → 4 SbH3 + 1.5 Li2O + 1.5 Al2O3 :4 SbCl3 + 3 NaBH4 → 4 SbH3 + 3 NaCl + 3 BCl3 Alternatively, sources of Sb3− react with protonic reagents (even water) to also produce this unstable gas: :Na3Sb + 3 H2O → SbH3 + 3 NaOH Properties The chemical properties of SbH3 resemble those for AsH3. Typical for a heavy hydride (e.g. AsH3, H2Te, SnH4), SbH3 is unstable with respect to its elements. The gas decomposes slowly at room temperature but rapidly at 200 °C: ::2 SbH3 → 3 H2 + ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Antimony
Antimony is a chemical element with the symbol Sb (from la, stibium) and atomic number 51. A lustrous gray metalloid, it is found in nature mainly as the sulfide mineral stibnite (Sb2S3). Antimony compounds have been known since ancient times and were powdered for use as medicine and cosmetics, often known by the Arabic name kohl. The earliest known description of the metal in the West was written in 1540 by Vannoccio Biringuccio. China is the largest producer of antimony and its compounds, with most production coming from the Xikuangshan Mine in Hunan. The industrial methods for refining antimony from stibnite are roasting followed by reduction with carbon, or direct reduction of stibnite with iron. The largest applications for metallic antimony are in alloys with lead and tin, which have improved properties for solders, bullets, and plain bearings. It improves the rigidity of lead-alloy plates in lead–acid batteries. Antimony trioxide is a prominent additive ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Woolwich
Woolwich () is a district in southeast London, England, within the Royal Borough of Greenwich. The district's location on the River Thames led to its status as an important naval, military and industrial area; a role that was maintained throughout the 16th to 20th centuries. After several decades of economic hardship and social deprivation, the area now has several large-scale urban renewal projects. Geography Woolwich is situated from Charing Cross. It has a long frontage to the south bank of the Thames river. From the riverside it rises up quickly along the northern slopes of Shooter's Hill towards the common, at and the ancient London–Dover Road, at . The ancient parish of Woolwich, more or less the present-day wards Woolwich Riverside and Woolwich Common, comprises . This included North Woolwich, which is now part of the London Borough of Newham. The ancient parishes of Plumstead and Eltham became part of the civil parish of Woolwich in 1930. Parts of th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Royal Arsenal
The Royal Arsenal, Woolwich is an establishment on the south bank of the River Thames in Woolwich in south-east London, England, that was used for the manufacture of armaments and ammunition, proofing, and explosives research for the British armed forces. It was originally known as the Woolwich Warren, having begun on land previously used as a domestic warren in the grounds of a Tudor house, Tower Place. Much of the initial history of the site is linked with that of the Office of Ordnance, which purchased the Warren in the late 17th century in order to expand an earlier base at Gun Wharf in Woolwich Dockyard. Over the next two centuries, as operations grew and innovations were pursued, the site expanded massively. At the time of the First World War the Arsenal covered and employed close to 80,000 people. Thereafter its operations were scaled down. It finally closed as a factory in 1967 and the Ministry of Defence moved out in 1994. Today the area, so long a secret enc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |