|
Magnesium Gluconate
Magnesium gluconate is a compound with formula MgC12H22O14. It is the magnesium salt of gluconic acid. According to one study, magnesium gluconate showed the highest level of bioavailability of any magnesium salt which implies its viability as a supplement, although of the 10 salts studied, all increased magnesium levels significantly. This study did not include magnesium glycinate, which is one of the most bioavailable forms of magnesium. It has E number "E580". Use in medicine There are data on the pharmacological properties of magnesium gluconate. Gluconic acid is the initial substrate for the reactions of pentose phosphate path of oxidation of glucose, so it was suggested that it may affect the energy metabolism of mitochondria. In Ukraine, magnesium gluconate, together with potassium gluconate Potassium gluconate is the potassium salt of the conjugate base of gluconic acid. It is also referred to as 2,3,4,5,6-pentahydroxycaproic acid potassium salt, D-gluconic acid potas ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Compound (chemistry)
A chemical compound is a chemical substance composed of many identical molecules (or molecular entities) containing atoms from more than one chemical element held together by chemical bonds. A molecule consisting of atoms of only one element is therefore not a compound. A compound can be transformed into a different substance by a chemical reaction, which may involve interactions with other substances. In this process, bonds between atoms may be broken and/or new bonds formed. There are four major types of compounds, distinguished by how the constituent atoms are bonded together. Molecular compounds are held together by covalent bonds; ionic compounds are held together by ionic bonds; intermetallic compounds are held together by metallic bonds; coordination complexes are held together by coordinate covalent bonds. Non-stoichiometric compounds form a disputed marginal case. A chemical formula specifies the number of atoms of each element in a compound molecule, using the stan ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Magnesium
Magnesium is a chemical element with the symbol Mg and atomic number 12. It is a shiny gray metal having a low density, low melting point and high chemical reactivity. Like the other alkaline earth metals (group 2 of the periodic table) it occurs naturally only in combination with other elements and it almost always has an oxidation state of +2. It reacts readily with air to form a thin passivation coating of magnesium oxide that inhibits further corrosion of the metal. The free metal burns with a brilliant-white light. The metal is obtained mainly by electrolysis of magnesium salts obtained from brine. It is less dense than aluminium and is used primarily as a component in strong and lightweight alloys that contain aluminium. In the cosmos, magnesium is produced in large, aging stars by the sequential addition of three helium nuclei to a carbon nucleus. When such stars explode as supernovas, much of the magnesium is expelled into the interstellar medium where it ma ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Salt (chemistry)
In chemistry, a salt is a chemical compound consisting of an ionic assembly of positively charged cations and negatively charged anions, which results in a compound with no net electric charge. A common example is table salt, with positively charged sodium ions and negatively charged chloride ions. The component ions in a salt compound can be either inorganic, such as chloride (Cl−), or organic, such as acetate (). Each ion can be either monatomic, such as fluoride (F−), or polyatomic, such as sulfate (). Types of salt Salts can be classified in a variety of ways. Salts that produce hydroxide ions when dissolved in water are called ''alkali salts'' and salts that produce hydrogen ions when dissolved in water are called ''acid salts''. ''Neutral salts'' are those salts that are neither acidic nor basic. Zwitterions contain an anionic and a cationic centre in the same molecule, but are not considered salts. Examples of zwitterions are amino acids, many metabolites, peptid ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gluconic Acid
Gluconic acid is an organic compound with molecular formula C6H12O7 and condensed structural formula HOCH2(CHOH)4COOH. It is one of the 16 stereoisomers of 2,3,4,5,6-pentahydroxyhexanoic acid. In aqueous solution at neutral pH, gluconic acid forms the gluconate ion. The salts of gluconic acid are known as "gluconates". Gluconic acid, gluconate salts, and gluconate esters occur widely in nature because such species arise from the oxidation of glucose. Some drugs are injected in the form of gluconates. Chemical structure The chemical structure of gluconic acid consists of a six-carbon chain, with five hydroxyl groups positioned in the same way as in the open-chained form of glucose, terminating in a carboxylic acid group. In aqueous solution, gluconic acid exists in equilibrium with the cyclic ester glucono delta-lactone. Production Gluconic acid preparation was first reported by Hlasiwetz and Habermann in 1870 and involved the chemical oxidation of glucose. In 1880, Boutroux ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bioavailability
In pharmacology, bioavailability is a subcategory of absorption and is the fraction (%) of an administered drug that reaches the systemic circulation. By definition, when a medication is administered intravenously, its bioavailability is 100%. However, when a medication is administered via routes other than intravenous, its bioavailability is generally lower than that of intravenous due to intestinal endothelium absorption and first-pass metabolism. Thereby, mathematically, bioavailability equals the ratio of comparing the area under the plasma drug concentration curve versus time (AUC) for the extravascular formulation to the AUC for the intravascular formulation. AUC is used because AUC is proportional to the dose that has entered the systemic circulation. Bioavailability of a drug is an average value; to take population variability into account, deviation range is shown as ±. To ensure that the drug taker who has poor absorption is dosed appropriately, the bottom value o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Magnesium Glycinate
Magnesium glycinate, also known as magnesium diglycinate or magnesium bisglycinate, is the magnesium salt of glycine (one magnesium and two glycine molecules), and is sold as a dietary supplement. It contains 14.1% elemental magnesium by mass. Accordingly, 141 mg of elemental magnesium is contained in 1000 mg of magnesium glycinate. Uses Magnesium glycinate has been studied with applicability to patients with a bowel resection or pregnancy-induced leg cramps. See also * Magnesium (pharmaceutical preparation) * Magnesium deficiency (medicine) * Magnesium in biology Magnesium is an essential element in biological systems. Magnesium occurs typically as the Mg2+ ion. It is an essential mineral nutrient (i.e., element) for life and is present in every cell type in every organism. For example, adenosine triphosph ... References {{organic-compound-stub Magnesium compounds Glycinates Dietary supplements ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
E Number
E numbers ("E" stands for "Europe") are codes for substances used as food additives, including those found naturally in many foods such as vitamin C, for use within the European Union (EU) and European Free Trade Association (EFTA). Commonly found on food labels, their safety assessment and approval are the responsibility of the European Food Safety Authority (EFSA). The fact that an additive has an E number implies that its use was at one time permitted in products for sale in the European Single Market; some of these additives are no longer allowed today. Having a single unified list for food additives was first agreed upon in 1962 with food colouring. In 1964, the directives for preservatives were added, in 1970 antioxidants were added, in 1974 emulsifiers, stabilisers, thickeners and gelling agents were added as well. Numbering schemes The numbering scheme follows that of the International Numbering System (INS) as determined by the '' Codex Alimentarius'' committee, t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gluconic Acid
Gluconic acid is an organic compound with molecular formula C6H12O7 and condensed structural formula HOCH2(CHOH)4COOH. It is one of the 16 stereoisomers of 2,3,4,5,6-pentahydroxyhexanoic acid. In aqueous solution at neutral pH, gluconic acid forms the gluconate ion. The salts of gluconic acid are known as "gluconates". Gluconic acid, gluconate salts, and gluconate esters occur widely in nature because such species arise from the oxidation of glucose. Some drugs are injected in the form of gluconates. Chemical structure The chemical structure of gluconic acid consists of a six-carbon chain, with five hydroxyl groups positioned in the same way as in the open-chained form of glucose, terminating in a carboxylic acid group. In aqueous solution, gluconic acid exists in equilibrium with the cyclic ester glucono delta-lactone. Production Gluconic acid preparation was first reported by Hlasiwetz and Habermann in 1870 and involved the chemical oxidation of glucose. In 1880, Boutroux ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glucose
Glucose is a simple sugar with the molecular formula . Glucose is overall the most abundant monosaccharide, a subcategory of carbohydrates. Glucose is mainly made by plants and most algae during photosynthesis from water and carbon dioxide, using energy from sunlight, where it is used to make cellulose in cell walls, the most abundant carbohydrate in the world. In energy metabolism, glucose is the most important source of energy in all organisms. Glucose for metabolism is stored as a polymer, in plants mainly as starch and amylopectin, and in animals as glycogen. Glucose circulates in the blood of animals as blood sugar. The naturally occurring form of glucose is -glucose, while -glucose is produced synthetically in comparatively small amounts and is less biologically active. Glucose is a monosaccharide containing six carbon atoms and an aldehyde group, and is therefore an aldohexose. The glucose molecule can exist in an open-chain (acyclic) as well as ring (cyclic) form. Gluco ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
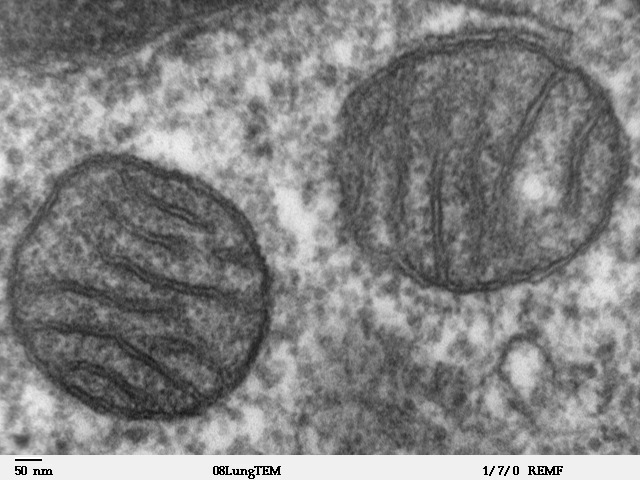
Mitochondria
A mitochondrion (; ) is an organelle found in the Cell (biology), cells of most Eukaryotes, such as animals, plants and Fungus, fungi. Mitochondria have a double lipid bilayer, membrane structure and use aerobic respiration to generate adenosine triphosphate (ATP), which is used throughout the cell as a source of chemical energy. They were discovered by Albert von Kölliker in 1857 in the voluntary muscles of insects. The term ''mitochondrion'' was coined by Carl Benda in 1898. The mitochondrion is popularly nicknamed the "powerhouse of the cell", a phrase coined by Philip Siekevitz in a 1957 article of the same name. Some cells in some multicellular organisms lack mitochondria (for example, mature mammalian red blood cells). A large number of unicellular organisms, such as microsporidia, parabasalids and diplomonads, have reduced or transformed their mitochondria into mitosome, other structures. One eukaryote, ''Monocercomonoides'', is known to have completely lost its mitocho ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Potassium Gluconate
Potassium gluconate is the potassium salt of the conjugate base of gluconic acid. It is also referred to as 2,3,4,5,6-pentahydroxycaproic acid potassium salt, D-gluconic acid potassium salt, or potassium D-gluconate. It contains 16.69% potassium by mass. Thus 5.99 g of potassium gluconate contains 1 g of potassium. It has a density of 1.73 g/cm3. Dietary uses Potassium gluconate is used as a mineral supplement and sequestrant. It is sold over-the-counter as tablets or capsules providing up to 593 mg of potassium gluconate, thereby containing 99 mg or 2.53 milliequivalents of elemental potassium. This is the permissible upper limit for each tablet or capsule of over-the-counter potassium supplements sold in the US. Potassium gluconate is also sold over-the-counter as bulk powder. As a food additive, potassium gluconate is used as an acidity regulator and yeast food. It is known as E number reference E577. Safety Its oral median lethal dose (LD50) in rats is 10.38 g/kg. This is ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gluconates
Gluconic acid is an organic compound with molecular formula C6H12O7 and condensed structural formula HOCH2(CHOH)4COOH. It is one of the 16 stereoisomers of 2,3,4,5,6-pentahydroxyhexanoic acid. In aqueous solution at neutral pH, gluconic acid forms the gluconate ion. The salt (chemistry), salts of gluconic acid are known as "gluconates". Gluconic acid, gluconate salts, and gluconate esters occur widely in nature because such species arise from the oxidation of glucose. Some drugs are injected in the form of gluconates. Chemical structure The chemical structure of gluconic acid consists of a six-carbon chain, with five hydroxyl groups positioned in the same way as in the glucose#Open-chain form, open-chained form of glucose, terminating in a carboxylic acid group. In aqueous solution, gluconic acid exists in equilibrium with the cyclic ester glucono delta-lactone. Production Gluconic acid preparation was first reported by Hlasiwetz and Habermann in 1870 and involved the chemica ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |