|
Mozingo Reduction
The Mozingo reduction, also known as Mozingo reaction or thioketal reduction, is a chemical reaction capable of fully reducing a ketone or aldehyde to the corresponding alkane via a dithioacetal. The reaction scheme is as follows: The ketone or aldehyde is activated by conversion to cyclic dithioacetal by reaction with a dithiol (nucleophilic substitution) in presence of a H+ donating acid. The cyclic dithioacetal structure is then hydrogenolyzed using Raney nickel. Raney nickel is converted irreversibly to nickel sulfide. This method is milder than either the Clemmensen or Wolff-Kishner reductions, which employ strongly acidic or basic conditions, respectively, that might interfere with other functional groups. History The reaction is named after Ralph Mozingo, who reported the cleavage of thioether In organic chemistry, an organic sulfide (British English sulphide) or thioether is an organosulfur functional group with the connectivity as shown on right. Like many other ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Reaction
A chemical reaction is a process that leads to the IUPAC nomenclature for organic transformations, chemical transformation of one set of chemical substances to another. Classically, chemical reactions encompass changes that only involve the positions of electrons in the forming and breaking of chemical bonds between atoms, with no change to the Atomic nucleus, nuclei (no change to the elements present), and can often be described by a chemical equation. Nuclear chemistry is a sub-discipline of chemistry that involves the chemical reactions of unstable and radioactive Chemical element, elements where both electronic and nuclear changes can occur. The substance (or substances) initially involved in a chemical reaction are called reagent, reactants or reagents. Chemical reactions are usually characterized by a chemical change, and they yield one or more Product (chemistry), products, which usually have properties different from the reactants. Reactions often consist of a sequence o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogenolysis
Hydrogenolysis is a chemical reaction whereby a carbon–carbon or carbon–heteroatom single bond is cleaved or undergoes lysis (breakdown) by hydrogen.Ralph Connor, Homer Adkins. Hydrogenolysis Of Oxygenated Organic Compounds. J. Am. Chem. Soc.; 1932; 54(12); 4678–4690. The heteroatom may vary, but it usually is oxygen, nitrogen, or sulfur. A related reaction is hydrogenation, where hydrogen is added to the molecule, without cleaving bonds. Usually hydrogenolysis is conducted catalytically using hydrogen gas. History The term "hydrogenolysis" was coined by Carleton Ellis in reference to hydrogenolysis of carbon–carbon bonds. Earlier, Paul Sabatier had already observed the hydrogenolysis of benzyl alcohol to toluene, and as early as 1906, Padoa and Ponti had observed the hydrogenolysis of furfuryl alcohol. Homer Burton Adkins and Ralph Connor were the first to call the carbon–oxygen bond cleavage "hydrogenolysis". In the petrochemical industry In petroleum refineries, c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organic Reduction Reactions
Organic may refer to: * Organic, of or relating to an organism, a living entity * Organic, of or relating to an anatomical organ Chemistry * Organic matter, matter that has come from a once-living organism, is capable of decay or is the product of decay, or is composed of organic compounds * Organic compound, a compound that contains carbon ** Organic chemistry, chemistry involving organic compounds Farming, certification and products * Organic farming, agriculture conducted according to certain standards, especially the use of stated methods of fertilization and pest control * Organic certification, accreditation process for producers of organically-farmed products * Organic horticulture, the science and art of growing fruits, vegetables, flowers, or ornamental plants by following the essential principles of organic agriculture * Organic products, "organics": ** Organic food, food produced from organic farming methods and often certified organic according to organic farming stand ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Melville Wolfrom
Melville Lawrence Wolfrom (April 2, 1900 – June 20, 1969) was an American chemist. Early life, education, and career Melville Wolfrom's grandfather Johann Lorenz Wolfrum immigrated to the United States from Aš in 1854, and was of Sudeten Germans, Sudeten German descent. His son Friedrich (Frederick) Wolfrum married Maria Louisa Sutter. Melville Wolfrom was born on April 2, 1900, the youngest of nine children. His father died when Melville was seven years old. Three of his brothers acquired a patent for a horse harness snap, and as a teen, Melville helped manufacture them out of the family home. He graduated from Ohio's Bellevue High School (Ohio), Bellevue High School in 1917 as salutatorian and began working for the National Carbon Company. A year later, Wolfrom enrolled at Western Reserve University. He dropped out of the university and the Students' Army Training Corps after November 1918. In 1919, Wolfrom returned to school at Washington Square College, only to drop out a s ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thioether
In organic chemistry, an organic sulfide (British English sulphide) or thioether is an organosulfur functional group with the connectivity as shown on right. Like many other sulfur-containing compounds, volatile sulfides have foul odors. A sulfide is similar to an ether except that it contains a sulfur atom in place of the oxygen. The grouping of oxygen and sulfur in the periodic table suggests that the chemical properties of ethers and sulfides are somewhat similar, though the extent to which this is true in practice varies depending on the application. Nomenclature Sulfides are sometimes called thioethers, especially in the old literature. The two organic substituents are indicated by the prefixes. (CH3)2S is called dimethylsulfide. Some sulfides are named by modifying the common name for the corresponding ether. For example, C6H5SCH3 is methyl phenyl sulfide, but is more commonly called thioanisole, since its structure is related to that for anisole, C6H5OCH3. The modern sy ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Functional Group
In organic chemistry, a functional group is a substituent or moiety in a molecule that causes the molecule's characteristic chemical reactions. The same functional group will undergo the same or similar chemical reactions regardless of the rest of the molecule's composition. This enables systematic prediction of chemical reactions and behavior of chemical compounds and the design of chemical synthesis. The reactivity of a functional group can be modified by other functional groups nearby. Functional group interconversion can be used in retrosynthetic analysis to plan organic synthesis. A functional group is a group of atoms in a molecule with distinctive chemical properties, regardless of the other atoms in the molecule. The atoms in a functional group are linked to each other and to the rest of the molecule by covalent bonds. For repeating units of polymers, functional groups attach to their nonpolar core of carbon atoms and thus add chemical character to carbon chains. Fun ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Wolff–Kishner Reduction
The Wolff–Kishner reduction is a reaction used in organic chemistry to convert carbonyl functionalities into methylene groups. In the context of complex molecule synthesis, it is most frequently employed to remove a carbonyl group after it has served its synthetic purpose of activating an intermediate in a preceding step. As such, there is no obvious retron for this reaction. The reaction was reported by Nikolai Kischner in 1911 and Ludwig Wolff in 1912, In general, the reaction mechanism first involves the ''in situ'' generation of a hydrazone by condensation of hydrazine with the ketone or aldehyde substrate. Sometimes it is however advantageous to use a pre-formed hydrazone as substrate (see modifications). The rate determining step of the reaction is de-protonation of the hydrazone by an alkoxide base to form a diimide anion by a concerted, solvent mediated protonation/de-protonation step. Collapse of this alkyldiimide with loss of N2 leads to formation of an alkylanion whic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Clemmensen Reduction
Clemmensen reduction is a chemical reaction described as a reduction of ketones (or aldehydes) to alkanes using zinc amalgam and concentrated hydrochloric acid. This reaction is named after Erik Christian Clemmensen, a Danish chemist. The original Clemmensen reduction conditions are particularly effective at reducing aryl-alkyl ketones, such as those formed in a Friedel-Crafts acylation. The two-step sequence of Friedel-Crafts acylation followed by Clemmensen reduction constitutes a classical strategy for the primary alkylation of arenes. With aliphatic or cyclic ketones, modified Clemmensen conditions using activated zinc dust in an anhydrous solution of hydrogen chloride in diethyl ether or acetic anhydride is much more effective. The substrate must be tolerant of the strongly acidic conditions of the Clemmensen reduction (37% HCl). Several alternatives are available. Acid-sensitive substrates that are stable to strong base can be reduced using the Wolff-Kishner reductio ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nickel Sulfide
Nickel sulfide is any inorganic compound with the formula NiSx. They range in color from bronze (Ni3S2) to black (NiS2). The nickel sulfide with simplest stoichiometry is NiS, also known as the mineral millerite. From the economic perspective, Ni9S8, the mineral pentlandite, is the chief source of mined nickel. Other minerals include heazlewoodite (Ni3S2) and polydymite (Ni3S4). The mineral Vaesite is NiS2. Some nickel sulfides are used commercially as catalysts. Structure Like many related materials, nickel sulfide adopts the nickel arsenide motif. In this structure, nickel is octahedral and the sulfide centers are in trigonal prismatic sites. NiS has two polymorphs. The α-phase has a hexagonal unit cell, while the β-phase has a rhombohedral cell. The α-phase is stable at temperatures above , and converts into the β-phase at lower temperatures. That phase transition causes an increase in volume by 2-4%. Synthesis and reactions The precipitation of solid black nickel ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
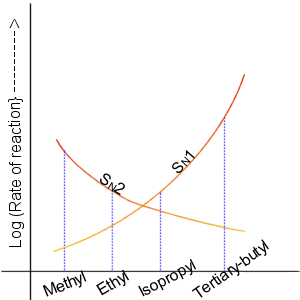
Nucleophilic Substitution
In chemistry, a nucleophilic substitution is a class of chemical reactions in which an electron-rich chemical species (known as a nucleophile) replaces a functional group within another electron-deficient molecule (known as the electrophile). The molecule that contains the electrophile and the leaving functional group is called the substrate. The most general form of the reaction may be given as the following: :\text\mathbf + \ce + \text\mathbf The electron pair (:) from the nucleophile (Nuc) attacks the substrate () and bonds with it. Simultaneously, the leaving group (LG) departs with an electron pair. The principal product in this case is . The nucleophile may be electrically neutral or negatively charged, whereas the substrate is typically neutral or positively charged. An example of nucleophilic substitution is the hydrolysis of an alkyl bromide, R-Br under basic conditions, where the attacking nucleophile is hydroxyl () and the leaving group is bromide (). :R-Br + OH- ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Redox
Redox (reduction–oxidation, , ) is a type of chemical reaction in which the oxidation states of substrate (chemistry), substrate change. Oxidation is the loss of Electron, electrons or an increase in the oxidation state, while reduction is the gain of electrons or a decrease in the oxidation state. There are two classes of redox reactions: * ''Electron-transfer'' – Only one (usually) electron flows from the reducing agent to the oxidant. This type of redox reaction is often discussed in terms of redox couples and electrode potentials. * ''Atom transfer'' – An atom transfers from one substrate to another. For example, in the rusting of iron, the oxidation state of iron atoms increases as the iron converts to an oxide, and simultaneously the oxidation state of oxygen decreases as it accepts electrons released by the iron. Although oxidation reactions are commonly associated with the formation of oxides, other chemical species can serve the same function. In hydrogen ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |