|
Meyer–Schuster Rearrangement
The Meyer–Schuster rearrangement is the chemical reaction described as an acid-catalysis, catalyzed rearrangement reaction, rearrangement of secondary and tertiary propargyl alcohols to α,β-unsaturated ketones if the alkyne group is internal and α,β-unsaturated aldehydes if the alkyne group is terminal. Reviews have been published by Swaminathan and Narayan,Swaminathan, S.; Narayan, K. V. "The Rupe and Meyer-Schuster Rearrangements" ''Chem. Rev.'' 1971, ''71'', 429–438.Review Vartanyan and Banbanyan, and Engel and Dudley, the last of which describes ways to promote the Meyer–Schuster rearrangement over other reactions available to propargyl alcohols. Mechanism The reaction mechanism begins with the protonation of the alcohol which leaves in an elimination reaction, E1 reaction to form the allene from the alkyne. Attack of a water molecule on the carbocation and deprotonation is followed by tautomerization to give the carbonyl#α,β-Unsaturated carbonyl compounds, α,β-u ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Reaction
A chemical reaction is a process that leads to the IUPAC nomenclature for organic transformations, chemical transformation of one set of chemical substances to another. Classically, chemical reactions encompass changes that only involve the positions of electrons in the forming and breaking of chemical bonds between atoms, with no change to the Atomic nucleus, nuclei (no change to the elements present), and can often be described by a chemical equation. Nuclear chemistry is a sub-discipline of chemistry that involves the chemical reactions of unstable and radioactive Chemical element, elements where both electronic and nuclear changes can occur. The substance (or substances) initially involved in a chemical reaction are called reagent, reactants or reagents. Chemical reactions are usually characterized by a chemical change, and they yield one or more Product (chemistry), products, which usually have properties different from the reactants. Reactions often consist of a sequence o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tautomerization
Tautomers () are structural isomers (constitutional isomers) of chemical compounds that readily interconvert. The chemical reaction interconverting the two is called tautomerization. This conversion commonly results from the relocation of a hydrogen atom within the compound. The phenomenon of tautomerization is called tautomerism, also called desmotropism. Tautomerism is for example relevant to the behavior of amino acids and nucleic acids, two of the fundamental building blocks of life. Care should be taken not to confuse tautomers with depictions of "contributing structures" in chemical resonance. Tautomers are distinct chemical species that can be distinguished by their differing atomic connectivities, molecular geometries, and physicochemical and spectroscopic properties, whereas resonance forms are merely alternative Lewis structure (valence bond theory) depictions of a single chemical species, whose true structure is best described as the "average" of the idealized, hypothe ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
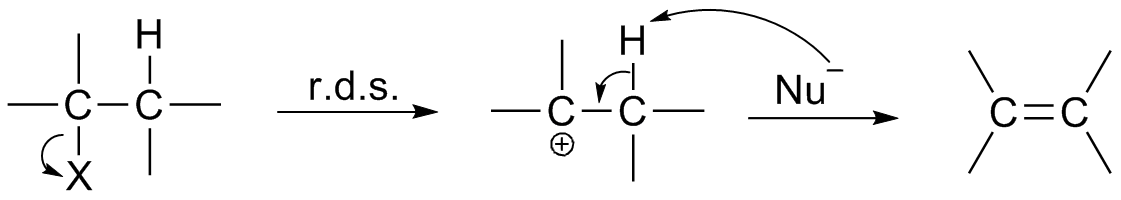
Reaction Intermediate
In chemistry, a reaction intermediate or an intermediate is a molecular entity that is formed from the reactants (or preceding intermediates) but is consumed in further reactions in stepwise chemical reactions that contain multiple elementary steps. Intermediates are the reaction product of one elementary step, but do not appear in the chemical equation for an overall chemical equation. For example, consider this hypothetical stepwise reaction: :A + B -> C + D The reaction includes two elementary steps: :A + B -> X :X -> C + D In this example, X is a reaction intermediate. IUPAC definition The IUPAC Gold Book defines an ''intermediate'' as a compound that has a lifetime greater than a molecular vibration that is formed (directly or indirectly) from the reactants and reacts further to give (either directly or indirectly) the products of a chemical reaction. The lifetime condition distinguishes true, chemically distinct intermediates from vibrational states or such transition st ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Enyne
In organic chemistry, an enyne is an organic compound containing a double bond (alkene) and a triple bond (alkyne). It is called a conjugated enyne when the double and triple bonds are conjugated. The term is a contraction of the terms alkene and alkyne. The simplest enyne is vinylacetylene. See also *Enyne metathesis *Enediyne In organic chemistry, enediynes are organic compounds containing two triple bonds and one double bond. Enediynes are most notable for their limited use as antitumor antibiotics (known as enediyne anticancer antibiotics). They are efficient at ... * Polyyne References Chemical nomenclature Alkene derivatives Alkyne derivatives Conjugated hydrocarbons {{organic-chemistry-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methyl
In organic chemistry, a methyl group is an alkyl derived from methane, containing one carbon atom bonded to three hydrogen atoms, having chemical formula . In formulas, the group is often abbreviated as Me. This hydrocarbon group occurs in many organic compounds. It is a very stable group in most molecules. While the methyl group is usually part of a larger molecule, bounded to the rest of the molecule by a single covalent bond (), it can be found on its own in any of three forms: methanide anion (), methylium cation () or methyl radical (). The anion has eight valence electrons, the radical seven and the cation six. All three forms are highly reactive and rarely observed. Methyl cation, anion, and radical Methyl cation The methylium cation () exists in the gas phase, but is otherwise not encountered. Some compounds are considered to be sources of the cation, and this simplification is used pervasively in organic chemistry. For example, protonation of methanol gives an elect ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acetylenic
In organic chemistry, the term acetylenic designates *A doubly unsaturated position (''sp''-hybridized) on a molecular framework, for instance in an alkyne such as acetylene; *An ethynyl fragment, HC\equivC–, or substituted homologue. See also * Allylic/Homoallylic * Benzylic In organic chemistry, benzyl is the substituent or molecular fragment possessing the structure . Benzyl features a benzene ring () attached to a methylene group () group. Nomenclature In IUPAC nomenclature, the prefix benzyl refers to a subst ... * Propargylic/Homopropargylic * Vinylic Organic chemistry {{organic-chemistry-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cage Effect (chemistry)
In chemistry, the cage effect (also known as geminate recombination) describes how the properties of a molecule are affected by its surroundings. First introduced by Franck and Rabinowitch in 1934, the cage effect suggests that instead of acting as an individual particle, molecules in solvent are more accurately described as an encapsulated particle. The encapsulated molecules or radicals are called cage pairs or geminate pairs. In order to interact with other molecules, the caged particle must diffuse from its solvent cage. The typical lifetime of a solvent cage is 10 seconds. Many manifestations of the cage effect exist. In free radical polymerization, radicals formed from the decomposition of an initiator molecule are surrounded by a cage consisting of solvent and/or monomer molecules. Within the cage, the free radicals undergo many collisions leading to their recombination or mutual deactivation. This can be described by the following reaction: : R\!-\!R \;\;\underse ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbonium Ion
In chemistry, a carbonium ion is any cation that has a pentavalent carbon atom. The name carbonium may also be used for the simplest member of the class, properly called methanium (), where the five valences are filled with hydrogen atoms. The next simplest carbonium ions after methanium have two carbon atoms. Ethynium, or protonated acetylene , and ethenium are usually classified in other families. The ethanium ion has been studied as an extremely rarefied gas by infrared spectroscopy. The isomers of octonium (protonated octane, ) have been studied. The carbonium ion has a planar geometry. In older literature, the name "carbonium ion" was used for what is today called carbenium. The current definitions were proposed by the chemist George Andrew Olah in 1972 and are now widely accepted. A stable carbonium ion is the complex pentakis(triphenylphosphinegold(I))methanium , produced by Schmidbauer and others. Preparation Carbonium ions can be obtained by treating alkanes with v ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Keto-enol Tautomerism
In organic chemistry, alkenols (shortened to enols) are a type of reactive structure or intermediate in organic chemistry that is represented as an alkene ( olefin) with a hydroxyl group attached to one end of the alkene double bond (). The terms ''enol'' and ''alkenol'' are portmanteaus deriving from "-ene"/"alkene" and the "-ol" suffix indicating the hydroxyl group of alcohols, dropping the terminal "-e" of the first term. Generation of enols often involves removal of a hydrogen adjacent (α-) to the carbonyl group—i.e., deprotonation, its removal as a proton, . When this proton is not returned at the end of the stepwise process, the result is an anion termed an enolate (see images at right). The enolate structures shown are schematic; a more modern representation considers the molecular orbitals that are formed and occupied by electrons in the enolate. Similarly, generation of the enol often is accompanied by "trapping" or masking of the hydroxy group as an ether, such as ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |