|
Methyl Red Indicator
In organic chemistry, a methyl group is an alkyl derived from methane, containing one carbon atom bonded to three hydrogen atoms, having chemical formula . In formulas, the group is often abbreviated as Me. This hydrocarbon group occurs in many organic compounds. It is a very stable group in most molecules. While the methyl group is usually part of a larger molecule, bounded to the rest of the molecule by a single covalent bond (), it can be found on its own in any of three forms: methanide anion (), methylium cation () or methyl radical (). The anion has eight valence electrons, the radical seven and the cation six. All three forms are highly reactive and rarely observed. Methyl cation, anion, and radical Methyl cation The methylium cation () exists in the gas phase, but is otherwise not encountered. Some compounds are considered to be sources of the cation, and this simplification is used pervasively in organic chemistry. For example, protonation of methanol gives an electr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methyl Group General Formulae V
In organic chemistry, a methyl group is an alkyl derived from methane, containing one carbon atom bonded to three hydrogen atoms, having chemical formula . In formulas, the group is often abbreviated as Me. This hydrocarbon group occurs in many organic compounds. It is a very stable group in most molecules. While the methyl group is usually part of a larger molecule, bounded to the rest of the molecule by a single covalent bond (), it can be found on its own in any of three forms: methanide anion (), methylium cation () or methyl radical (). The anion has eight valence electrons, the radical seven and the cation six. All three forms are highly reactive and rarely observed. Methyl cation, anion, and radical Methyl cation The methylium cation () exists in the gas phase, but is otherwise not encountered. Some compounds are considered to be sources of the cation, and this simplification is used pervasively in organic chemistry. For example, protonation of methanol gives ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Protonation
In chemistry, protonation (or hydronation) is the adding of a proton (or hydron, or hydrogen cation), (H+) to an atom, molecule, or ion, forming a conjugate acid. (The complementary process, when a proton is removed from a Brønsted–Lowry acid, is deprotonation.) Some examples include *The protonation of water by sulfuric acid: *:H2SO4 + H2O H3O+ + *The protonation of isobutene in the formation of a carbocation: *:(CH3)2C=CH2 + HBF4 (CH3)3C+ + *The protonation of ammonia in the formation of ammonium chloride from ammonia and hydrogen chloride: *:NH3( g) + HCl( g) → NH4Cl( s) Protonation is a fundamental chemical reaction and is a step in many stoichiometric and catalytic processes. Some ions and molecules can undergo more than one protonation and are labeled polybasic, which is true of many biological macromolecules. Protonation and deprotonation (removal of a proton) occur in most acid–base reactions; they are the core of most acid–base reaction theories. A B ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
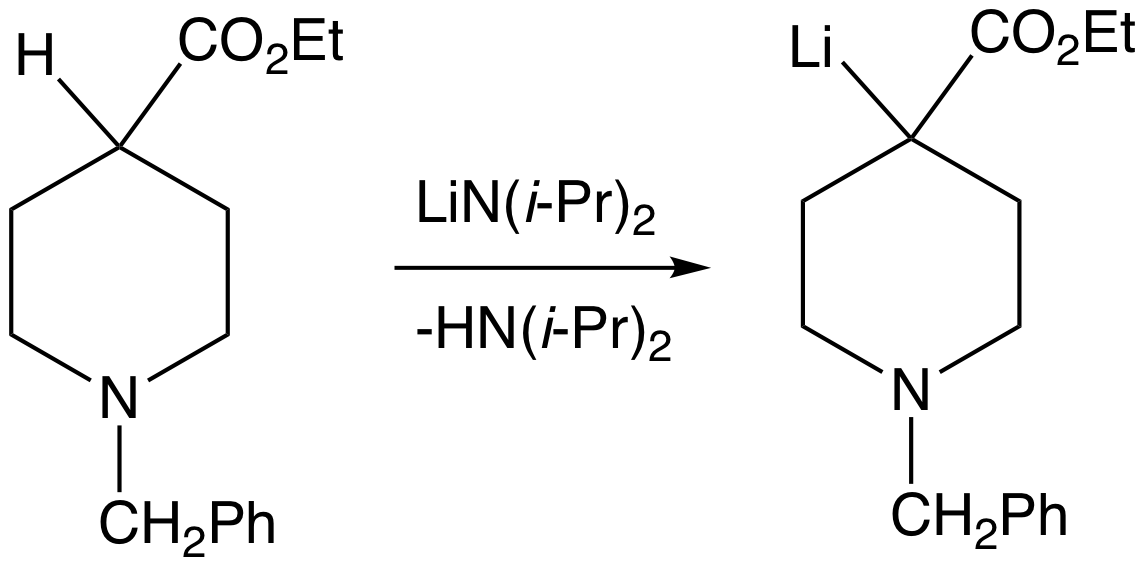
Grignard Reagents
A Grignard reagent or Grignard compound is a chemical compound with the general formula , where X is a halogen and R is an organic group, normally an alkyl or aryl. Two typical examples are methylmagnesium chloride and phenylmagnesium bromide . They are a subclass of the organomagnesium compounds. Grignard compounds are popular reagents in organic synthesis for creating new carbon-carbon bonds. For example, when reacted with another halogenated compound in the presence of a suitable catalyst, they typically yield and the magnesium halide as a byproduct; and the latter is insoluble in the solvents normally used. In this aspect, they are similar to organolithium reagents. Pure Grignard reagents are extremely reactive solids. They are normally handled as solutions in solvents such as diethyl ether or tetrahydrofuran; which are relatively stable as long as water is excluded. In such a medium, a Grignard reagent is invariably present as a complex with the magnesium atom ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methyl Lithium
Methyllithium is the simplest organolithium reagent with the empirical formula CH3Li. This s-block organometallic compound adopts an oligomeric structure both in solution and in the solid state. This highly reactive compound, invariably used in solution with an ether as the solvent, is a reagent in organic synthesis as well as organometallic chemistry. Operations involving methyllithium require anhydrous conditions, because the compound is highly reactive toward water. Oxygen and carbon dioxide are also incompatible with MeLi. Methyllithium is usually not prepared, but purchased as a solution in various ethers. Synthesis In the direct synthesis, methyl bromide is treated with a suspension of lithium in diethyl ether. :2 Li + MeBr → LiMe + LiBr The lithium bromide forms a complex with the methyllithium. Most commercially available methyllithium consists of this complex. "Halide-free" methyllithium is prepared from methyl chloride. Lithium chloride precipitates fro ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diethynylbenzene Dianion
In organic chemistry, a diethynylbenzene dianion is an anion consisting of two ethynyl anions as substituents on a benzene ring. With the chemical formula , three positional isomers are possible, differing in the relative positions of the two substituents around the ring: *''ortho''-diethynylbenzene dianion *''meta''-diethynylbenzene dianion *''para''-diethynylbenzene dianion The gaseous state of all three anions are of theoretical interest. They have been generated by decarboxylation of benzene dipropynoic acids, using the technique of mass spectrometry. The three isomers of the dianion are the three strongest known superbases ever, with the ''ortho'' isomer being the strongest, with a proton affinity of . The ''meta'' isomer is the second-strongest, and the ''para'' isomer is the third-strongest. Observation These dianions were generated in a linear quadrupole ion-trap mass spectrometer. Electrospray ionization (ESI) of the diacid precursor results in the dicarboxylate dia ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lithium Monoxide Anion
Lithium monoxide anion (LiO−) is a superbase existing in the gas phase. It was the strongest known base until 2008, when the isomeric diethynylbenzene dianions were determined to have a higher proton affinity. The methanide ion CH3− was the strongest known base before lithium monoxide anion was discovered. LiO− has a proton affinity of ~1782 kJ mol−1. Synthesis of the lithium monoxide anion The anion is prepared in a mass spectrometer by successive decarboxylation and decarbonylation of lithium oxalate anion under collision induced dissociation (CID) conditions: LiO(C=O)(CO) → LiOCO− + CO2, LiOCO− → LiO− + CO. The above method to synthesize the lithium monoxide anion is inefficient and difficult to carry out. The required ion rapidly reacts with traces of moisture and molecular oxygen present in the air. The reaction is further intensified by the high pressure argon that is introduced into the instrument to carry out the CID step. References See also ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Superbase
A superbase is a compound that has a particularly high affinity for protons. Superbases are of theoretical interest and potentially valuable in organic synthesis. Superbases have been described and used since the 1850s.''Superbases for Organic Synthesis'' Ed. Ishikawa, T., John Wiley and Sons, Ltd.: West Sussex, UK. 2009. Definitions Generically IUPAC defines a superbase as a "compound having a very high basicity, such as lithium diisopropylamide." Superbases are often defined in two broad categories, organic and organometallic. Organic superbases are charge-neutral compounds with basicities greater than that of proton sponge (pKBH+ = 18.6 in MeCN)." In a related definition: any species with a higher absolute proton affinity (APA = 245.3 kcal/mol) and intrinsic gas phase basicity (GB = 239 kcal/mol) than proton sponge. Common superbases of this variety feature amidine, guanidine, and phosphazene functional groups. Strong superbases can be designed by utilizing multiple int ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mole (unit)
The mole, symbol mol, is the unit of amount of substance in the International System of Units (SI). The quantity amount of substance is a measure of how many elementary entities of a given substance are in an object or sample. The mole is defined as containing exactly elementary entities. Depending on what the substance is, an elementary entity may be an atom, a molecule, an ion, an ion pair, or a subatomic particle such as an electron. For example, 10 moles of water (a chemical compound) and 10 moles of mercury (a chemical element), contain equal amounts of substance and the mercury contains exactly one atom for each molecule of the water, despite the two having different volumes and different masses. The number of elementary entities in one mole is known as the Avogadro number, which is the approximate number of nucleons ( protons or neutrons) in one gram of ordinary matter. The previous definition of a mole was simply the number of elementary entities equal to that of 1 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Kilojoule
The joule ( , ; symbol: J) is the unit of energy in the International System of Units (SI). It is equal to the amount of work done when a force of 1 newton displaces a mass through a distance of 1 metre in the direction of the force applied. It is also the energy dissipated as heat when an electric current of one ampere passes through a resistance of one ohm for one second. It is named after the English physicist James Prescott Joule (1818–1889). Definition In terms of SI base units and in terms of SI derived units with special names, the joule is defined as One joule can also be defined by any of the following: * The work required to move an electric charge of one coulomb through an electrical potential difference of one volt, or one coulomb-volt (C⋅V). This relationship can be used to define the volt. * The work required to produce one watt of power for one second, or one watt-second (W⋅s) (compare kilowatt-hour, which is 3.6 megajoules). This relationship c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Enthalpy Of Reaction
The standard enthalpy of reaction (denoted \Delta_ H^\ominus or \Delta H_^\ominus) for a chemical reaction is the difference between total reactant and total product molar enthalpies, calculated for substances in their standard states. This can in turn be used to predict the total chemical bond energy liberated or bound during reaction, as long as the enthalpy of mixing is also accounted for. For a generic chemical reaction :\nu_ \text + \nu_ \text ~+ ~... \rightarrow \nu_ \text + \nu_ \text ~+ ~... the standard enthalpy of reaction \Delta_ H^\ominus is related to the standard enthalpy of formation \Delta_ H^\ominus values of the reactants and products by the following equation: : \Delta_ H^\ominus = \sum_ \nu_p\Delta_ H_^ - \sum_ \nu_r\Delta_ H_^ In this equation, \nu_p and \nu_r are the stoichiometric coefficients of each product p and reactant r. The standard enthalpy of formation, which has been determined for a vast number of substances, is the change of enthalpy ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Torr
The torr (symbol: Torr) is a unit of pressure based on an absolute scale, defined as exactly of a standard atmosphere (). Thus one torr is exactly (≈ ). Historically, one torr was intended to be the same as one "millimeter of mercury", but subsequent redefinitions of the two units made them slightly different (by less than ). The torr is not part of the International System of Units (SI). It is often combined with the metric prefix milli to name one millitorr (mTorr) or 0.001 Torr. The unit was named after Evangelista Torricelli, an Italian physicist and mathematician who discovered the principle of the barometer in 1644. Nomenclature and common errors The unit name ''torr'' is written in lower case, while its symbol ("Torr") is always written with upper-case initial; including in combinations with prefixes and other unit symbols, as in "mTorr" (millitorr) or "Torr⋅L/s" (torr-litres per second). The symbol (uppercase) should be used with prefix symbols ( ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ketene
In organic chemistry, a ketene is an organic compound of the form , where R and R' are two arbitrary monovalent chemical groups (or two separate substitution sites in the same molecule). The name may also refer to the specific compound ethenone , the simplest ketene. Although they are highly useful, most ketenes are unstable. When used as reagents in a chemical procedure, they are typically generated when needed, and consumed as soon as (or while) they are produced. History Ketenes were first studied as a class by Hermann Staudinger before 1905. Ketenes were systematically investigated by Hermann Staudinger in 1905 in the form of diphenylketene (conversion of \alpha-chlorodiphenyl acetyl chloride with zinc). Staudinger was inspired by the first examples of reactive organic intermediates and stable radicals discovered by Moses Gomberg in 1900 (compounds with triphenylmethyl group). Properties Ketenes are highly electrophilic at the carbon atom bonded with the heteroat ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |