|
Mendeleev's Predicted Elements
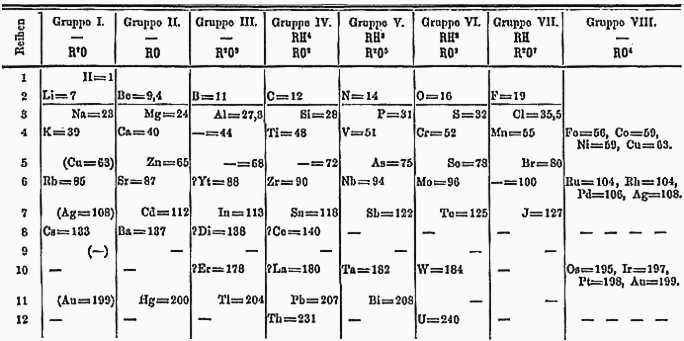
Dmitri Mendeleev published a periodic table of the chemical elements in 1869 based on properties that appeared with some regularity as he laid out the elements from lightest to heaviest. When Mendeleev proposed his periodic table, he noted gaps in the table and predicted that then-unknown elements existed with properties appropriate to fill those gaps. He named them eka-boron, eka-aluminium, eka-silicon, and eka-manganese, with respective atomic masses of 44, 68, 72, and 100. Prefixes To give provisional names to his predicted elements, Mendeleev used the prefixes ''eka''- , '' dvi''- or '' dwi-'', and ''tri''-, from the Sanskrit names of digits 1, 2, and 3, depending upon whether the predicted element was one, two, or three places down from the known element of the same group in his table. For example, germanium was called eka-silicon until its discovery in 1886, and rhenium was called dvi-manganese before its discovery in 1926. The ''eka-'' prefix was used by other theorist ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dmitri Mendeleev
Dmitri Ivanovich Mendeleev (sometimes transliterated as Mendeleyev or Mendeleef) ( ; russian: links=no, Дмитрий Иванович Менделеев, tr. , ; 8 February O.S. 27 January">Old_Style_and_New_Style_dates.html" ;"title="nowiki/>Old Style and New Style dates">O.S. 27 January18342 February [O.S. 20 January] 1907) was a Russian chemist and inventor. He is best known for formulating the Periodic Law and creating a version of the periodic table, periodic table of elements. He used the Periodic Law not only to correct the then-accepted properties of some known elements, such as the valence and atomic weight of uranium, but also to predict the properties of three elements that were yet to be discovered. Early life Mendeleev was born in the village of Verkhnie Aremzyani, near Tobolsk in Siberia, to (1783–1847) and Maria Dmitrievna Mendeleeva ( née Kornilieva) (1793–1850).''Maria Mendeleeva (1951)''. D. I. Mendeleev's Archive: Autobiographical Writings. Col ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Rare-earth Element
The rare-earth elements (REE), also called the rare-earth metals or (in context) rare-earth oxides or sometimes the lanthanides (yttrium and scandium are usually included as rare earths), are a set of 17 nearly-indistinguishable lustrous silvery-white soft heavy metals. Compounds containing rare earths have diverse applications in electrical and electronic components, lasers, glass, magnetic materials, and industrial processes. Scandium and yttrium are considered rare-earth elements because they tend to occur in the same ore deposits as the lanthanides and exhibit similar chemical properties, but have different electronic and magnetic properties. These metals tarnish slowly in air at room temperature and react slowly with cold water to form hydroxides, liberating hydrogen. They react with steam to form oxides, and at elevated temperature (400°C) ignite spontaneously. These elements and their compounds have no biological function other than in several specialized enzymes, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Scandium Oxide
Scandium(III) oxide or scandia is a inorganic compound with formula Sc2 O3. It is one of several oxides of rare earth elements with a high melting point. It is used in the preparation of other scandium compounds as well as in high-temperature systems (for its resistance to heat and thermal shock), electronic ceramics, and glass composition (as a helper material). Structure and physical properties Scandium(III) oxide adopts a cubic crystal structure (point group: tetrahedral (Th), space group: Ia) containing 6-coordinate metal centres. Powder diffraction analysis shows Sc−O bond distances of 2.159–2.071 Å. Scandium oxide is an insulator with a band gap of 6.0 eV. Production Scandium oxide is the primary form of refined scandium produced by the mining industry. Scandium-rich ores, such as thortveitite (Sc,Y)2(Si2O7) and kolbeckite ScPO4·2H2O are rare, however trace amounts of scandium are present in many other minerals. Scandium oxide is therefore predominant ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Unknown Element
Unknown or The Unknown may refer to: Film * ''The Unknown'' (1915 comedy film), a silent boxing film * ''The Unknown'' (1915 drama film) * ''The Unknown'' (1927 film), a silent horror film starring Lon Chaney * ''The Unknown'' (1936 film), a German drama film * ''The Unknown'' (1946 film), a mystery film * '' Anjaane: The Unknown'', a 2005 Bollywood horror movie * ''The Unknown'', a 2005 action/thriller starring Miles O'Keeffe * ''Unknown'' (2006 film), a thriller starring James Caviezel * ''Unknown'' (2011 film), a thriller starring Liam Neeson Literature * ''Unknown'' (magazine), an American pulp fantasy fiction magazine published from 1939 to 1943 * ''The Unknown'' (novel), a 1998 book by K. A. Applegate * ''The Unknown'', a comic book mini-series by Mark Waid Music * ''The Unknown'' (Madeline Juno album) (2014) * ''Unknown'' (Rasputina album) (2015) * ''The Unknown'' (The Vision Bleak album) (2016) * ''The Unknown'', a 2014 album by Dillon * "The Unknown" (song ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Transition Element
In chemistry, a transition metal (or transition element) is a chemical element in the d-block of the periodic table (groups 3 to 12), though the elements of group 12 (and less often group 3) are sometimes excluded. They are the elements that can use d orbitals as valence orbitals to form chemical bonds. The lanthanide and actinide elements (the f-block) are called inner transition metals and are sometimes considered to be transition metals as well. Since they are metals, they are lustrous and have good electrical and thermal conductivity. Most (with the exception of group 11 and group 12) are hard and strong, and have high melting and boiling temperatures. They form compounds in any of two or more different oxidation states and bind to a variety of ligands to form coordination complexes that are often coloured. They form many useful alloys and are often employed as catalysts in elemental form or in compounds such as coordination complexes and oxides. Most are strongly parama ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Rare Earth Element
The rare-earth elements (REE), also called the rare-earth metals or (in context) rare-earth oxides or sometimes the lanthanides ( yttrium and scandium are usually included as rare earths), are a set of 17 nearly-indistinguishable lustrous silvery-white soft heavy metals. Compounds containing rare earths have diverse applications in electrical and electronic components, lasers, glass, magnetic materials, and industrial processes. Scandium and yttrium are considered rare-earth elements because they tend to occur in the same ore deposits as the lanthanides and exhibit similar chemical properties, but have different electronic and magnetic properties. These metals tarnish slowly in air at room temperature and react slowly with cold water to form hydroxides, liberating hydrogen. They react with steam to form oxides, and at elevated temperature (400°C) ignite spontaneously. These elements and their compounds have no biological function other than in several specialized enzym ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Reforms Of Russian Orthography
The Russian orthography has been reformed officially and unofficially by changing the Russian alphabet over the course of the history of the Russian language. Several important reforms happened in the 18th–20th centuries. Early changes Old East Slavic adopted the Cyrillic script, approximately during the 10th century and at about the same time as the introduction of Eastern Christianity into the territories inhabited by the Eastern Slavs. No distinction was drawn between the vernacular language and the liturgical, though the latter was based on South Slavic rather than Eastern Slavic norms. As the language evolved, several letters, notably the ''yuses'' (Ѫ, Ѭ, Ѧ, Ѩ) were gradually and unsystematically discarded from both secular and church usage over the next centuries. The emergence of the centralized Russian state in the 15th and 16th centuries, the consequent rise of the state bureaucracy along with the development of the common economic, political and cultural sp ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dmitriy Mendeleev
Dmitri Ivanovich Mendeleev (sometimes transliterated as Mendeleyev or Mendeleef) ( ; russian: links=no, Дмитрий Иванович Менделеев, tr. , ; 8 February O.S. 27 January">Old_Style_and_New_Style_dates.html" ;"title="nowiki/>Old Style and New Style dates">O.S. 27 January18342 February [O.S. 20 January] 1907) was a Russian chemist and inventor. He is best known for formulating the Periodic Law and creating a version of the periodic table, periodic table of elements. He used the Periodic Law not only to correct the then-accepted properties of some known elements, such as the valence and atomic weight of uranium, but also to predict the properties of three elements that were yet to be discovered. Early life Mendeleev was born in the village of Verkhnie Aremzyani, near Tobolsk in Siberia, to (1783–1847) and Maria Dmitrievna Mendeleeva (née Kornilieva) (1793–1850).''Maria Mendeleeva (1951)''. D. I. Mendeleev's Archive: Autobiographical Writings. Collec ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Technetium
Technetium is a chemical element with the symbol Tc and atomic number 43. It is the lightest element whose isotopes are all radioactive. All available technetium is produced as a synthetic element. Naturally occurring technetium is a spontaneous fission product in uranium ore and thorium ore, the most common source, or the product of neutron capture in molybdenum ores. This silvery gray, crystalline transition metal lies between manganese and rhenium in group 7 of the periodic table, and its chemical properties are intermediate between those of both adjacent elements. The most common naturally occurring isotope is 99Tc, in traces only. Many of technetium's properties had been predicted by Dmitri Mendeleev before it was discovered. Mendeleev noted a gap in his periodic table and gave the undiscovered element the provisional name '' ekamanganese'' (''Em''). In 1937, technetium (specifically the technetium-97 isotope) became the first predominantly artificial element to be produced ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gallium
Gallium is a chemical element with the symbol Ga and atomic number 31. Discovered by French chemist Paul-Émile Lecoq de Boisbaudran in 1875, Gallium is in group 13 of the periodic table and is similar to the other metals of the group ( aluminium, indium, and thallium). Elemental gallium is a soft, silvery metal in standard temperature and pressure. In its liquid state, it becomes silvery white. If too much force is applied, the gallium may fracture conchoidally. Since its discovery in 1875, gallium has widely been used to make alloys with low melting points. It is also used in semiconductors, as a dopant in semiconductor substrates. The melting point of gallium is used as a temperature reference point. Gallium alloys are used in thermometers as a non-toxic and environmentally friendly alternative to mercury, and can withstand higher temperatures than mercury. An even lower melting point of , well below the freezing point of water, is claimed for the alloy galinstan ( ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Scandium
Scandium is a chemical element with the symbol Sc and atomic number 21. It is a silvery-white metallic d-block element. Historically, it has been classified as a rare-earth element, together with yttrium and the Lanthanides. It was discovered in 1879 by spectral analysis of the minerals euxenite and gadolinite from Scandinavia. Scandium is present in most of the deposits of rare-earth and uranium compounds, but it is extracted from these ores in only a few mines worldwide. Because of the low availability and difficulties in the preparation of metallic scandium, which was first done in 1937, applications for scandium were not developed until the 1970s, when the positive effects of scandium on aluminium alloys were discovered, and its use in such alloys remains its only major application. The global trade of scandium oxide is 15–20 tonnes per year. The properties of scandium compounds are intermediate between those of aluminium and yttrium. A diagonal relationship exi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |

%2C_page_5.gif)

