|
Membrane Scaling
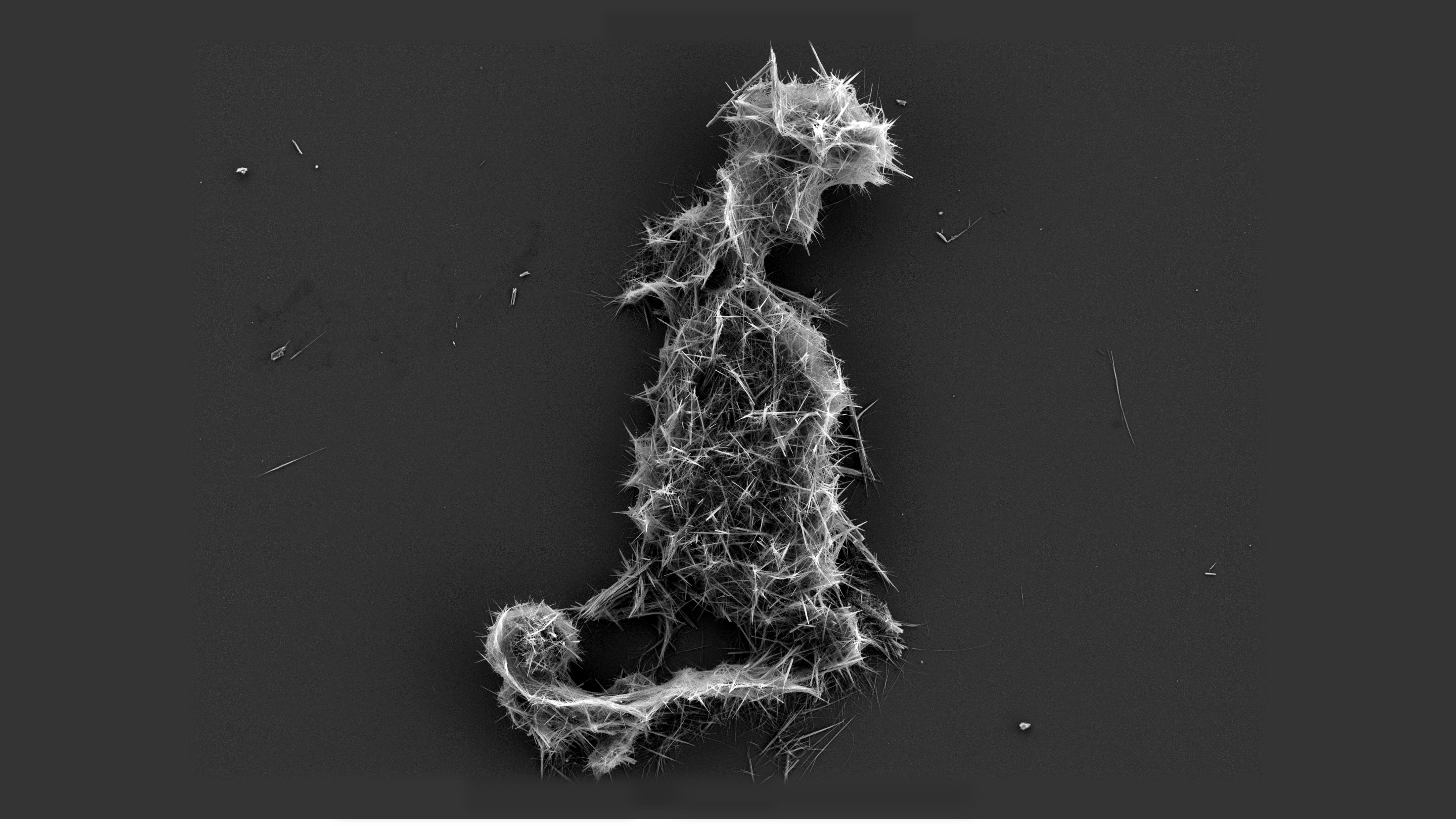
Membrane scaling is when one or more sparingly soluble salts (e.g., calcium carbonate, calcium phosphate, etc.) precipitate and form a dense layer on the membrane surface in reverse osmosis (RO) applications. Figures 1 and 2 show scanning electron microscopy (SEM) images of the RO membrane surface without and with scaling, respectively. Membrane scaling, like other types of membrane fouling, increases energy costs due to higher operating pressure, and reduces permeate water production. Furthermore, scaling may damage and shorten the lifetime of membranes due to frequent membrane cleanings and therefore it is a major operational challenge in RO applications. Membrane scaling can occur when sparingly soluble salts in RO concentrate become supersaturated, meaning their concentrations exceed their equilibrium (solubility) levels. In RO processes, the increased concentration of sparingly soluble salts in the concentrate is primarily caused by the withdrawal of permeate water from the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Virgin RO Membrane
Virginity is the state of a person who has never engaged in sexual intercourse. The term ''virgin'' originally only referred to sexually inexperienced women, but has evolved to encompass a range of definitions, as found in traditional, modern and ethical concepts. Heterosexual individuals may or may not consider loss of virginity to occur only through penile-vaginal penetration, while people of other sexual orientations often include oral sex, anal sex, or mutual masturbation in their definitions of losing one's virginity. There are cultural and religious traditions that place special value and significance on this state, predominantly towards unmarried females, associated with notions of personal purity, honor, and worth. Like chastity, the concept of virginity has traditionally involved sexual abstinence. The concept of virginity usually involves moral or religious issues and can have consequences in terms of social status and in interpersonal relationships.See her anpages 47 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Barium Sulfate
Barium sulfate (or sulphate) is the inorganic compound with the chemical formula Ba SO4. It is a white crystalline solid that is odorless and insoluble in water. It occurs as the mineral barite, which is the main commercial source of barium and materials prepared from it. The white opaque appearance and its high density are exploited in its main applications.Holleman, A. F. and Wiberg, E. (2001) ''Inorganic Chemistry'', San Diego, CA : Academic Press, . Uses Drilling fluids About 80% of the world's barium sulfate production, mostly purified mineral, is consumed as a component of oil well drilling fluid. It increases the density of the fluid, increasing the hydrostatic pressure in the well and reducing the chance of a blowout. Radiocontrast agent Barium sulfate in suspension is often used medically as a radiocontrast agent for X-ray imaging and other diagnostic procedures. It is most often used in imaging of the GI tract during what is colloquially known as a "barium meal". ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Water Treatment
Water treatment is any process that improves the Water quality, quality of water to make it appropriate for a specific end-use. The end use may be drinking water, drinking, industrial water supply, irrigation, river flow maintenance, water recreation or many other uses, including being safely returned to the environment. Water treatment removes contaminants and undesirable components, or reduces their concentration so that the water becomes fit for its desired end-use. This treatment is crucial to human health and allows humans to benefit from both drinking and irrigation use. Water is the most crucial compound for life on Earth, and having drinkable water is a key worldwide concern for the twenty-first century. All living things require clean, uncontaminated water as a basic requirement. Water covers more than 71 percent of the earth’s surface, but only around 1% of it is drinkable according to international standards due to various Contamination, contaminations . Waste water ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Functional Group
In organic chemistry, a functional group is a substituent or moiety in a molecule that causes the molecule's characteristic chemical reactions. The same functional group will undergo the same or similar chemical reactions regardless of the rest of the molecule's composition. This enables systematic prediction of chemical reactions and behavior of chemical compounds and the design of chemical synthesis. The reactivity of a functional group can be modified by other functional groups nearby. Functional group interconversion can be used in retrosynthetic analysis to plan organic synthesis. A functional group is a group of atoms in a molecule with distinctive chemical properties, regardless of the other atoms in the molecule. The atoms in a functional group are linked to each other and to the rest of the molecule by covalent bonds. For repeating units of polymers, functional groups attach to their nonpolar core of carbon atoms and thus add chemical character to carbon chains. Fun ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carboxylic Acid
In organic chemistry, a carboxylic acid is an organic acid that contains a carboxyl group () attached to an R-group. The general formula of a carboxylic acid is or , with R referring to the alkyl, alkenyl, aryl, or other group. Carboxylic acids occur widely. Important examples include the amino acids and fatty acids. Deprotonation of a carboxylic acid gives a carboxylate anion. Examples and nomenclature Carboxylic acids are commonly identified by their trivial names. They at oftentimes have the suffix ''-ic acid''. IUPAC-recommended names also exist; in this system, carboxylic acids have an ''-oic acid'' suffix. For example, butyric acid (C3H7CO2H) is butanoic acid by IUPAC guidelines. For nomenclature of complex molecules containing a carboxylic acid, the carboxyl can be considered position one of the parent chain even if there are other substituents, such as 3-chloropropanoic acid. Alternately, it can be named as a "carboxy" or "carboxylic acid" substituent on another ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phosphonate
In organic chemistry, phosphonates or phosphonic acids are organophosphorus compounds containing groups (where R = alkyl, aryl, or just hydrogen). Phosphonic acids, typically handled as salts, are generally nonvolatile solids that are poorly soluble in organic solvents, but soluble in water and common alcohols. Many commercially important compounds are phosphonates, including glyphosate (the active molecule of the herbicide Roundup), and ethephon, a widely used plant growth regulator. Bisphosphonates are popular drugs for treatment of osteoporosis.Svara, J.; Weferling, N.; Hofmann, T. "Phosphorus Compounds, Organic," in ''Ullmann's Encyclopedia of Industrial Chemistry'', Wiley-VCH, Weinheim, 2008. . In biochemistry and medicinal chemistry, phosphonate groups are used as stable bioisoteres for phosphate, such as in the antiviral nucleotide analog, Tenofovir, one of the cornerstones of anti-HIV therapy. And there is an indication that phosphonate derivatives are "promising ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfonate
In organosulfur chemistry, a sulfonate is a salt or ester of a sulfonic acid. It contains the functional group , where R is an organic group. Sulfonates are the conjugate bases of sulfonic acids. Sulfonates are generally stable in water, non-oxidizing, and colorless. Many useful compounds and even some biochemicals feature sulfonates. Sulfonate salts Anions with the general formula are called sulfonates. They are the conjugate bases of sulfonic acids with formula . As sulfonic acids tend to be strong acids, the corresponding sulfonates are weak bases. Due to the stability of sulfonate anions, the cations of sulfonate salts such as scandium triflate have application as Lewis acids. A classic preparation of sulfonates is the Strecker sulfite alkylation, in which an alkali sulfite salt displaces a halide, typically in the presence of an iodine catalyst: :RX + M2SO3 -> RSO3M + MX An alternative is the condensation of a sulfonyl halide with an alcohol in pyridine: :ROH + R' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Langelier Saturation Index
Hard water is water that has high mineral content (in contrast with "soft water"). Hard water is formed when water percolates through deposits of limestone, chalk or gypsum, which are largely made up of calcium and magnesium carbonates, bicarbonates and sulfates. Hard drinking water may have moderate health benefits. It can pose critical problems in industrial settings, where water hardness is monitored to avoid costly breakdowns in boilers, cooling towers, and other equipment that handles water. In domestic settings, hard water is often indicated by a lack of foam formation when soap is agitated in water, and by the formation of limescale in kettles and water heaters.World Health OrganizatioHardness in Drinking-Water 2003 Wherever water hardness is a concern, water softening is commonly used to reduce hard water's adverse effects. Origins Natural rainwater, snow and other forms of precipitation typically have low concentrations of multivalent cations such as calcium and magnes ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
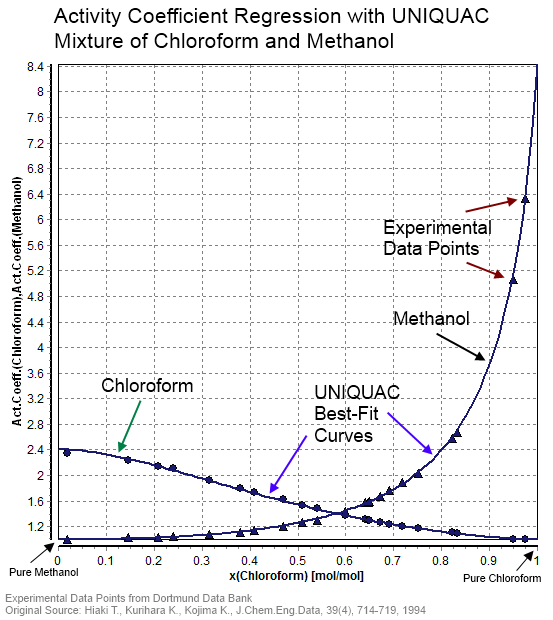
Activity Coefficient
In thermodynamics, an activity coefficient is a factor used to account for deviation of a mixture of chemical substances from ideal behaviour. In an ideal mixture, the microscopic interactions between each pair of chemical species are the same (or macroscopically equivalent, the enthalpy change of solution and volume variation in mixing is zero) and, as a result, properties of the mixtures can be expressed directly in terms of simple concentrations or partial pressures of the substances present e.g. Raoult's law. Deviations from ideality are accommodated by modifying the concentration by an ''activity coefficient''. Analogously, expressions involving gases can be adjusted for non-ideality by scaling partial pressures by a fugacity coefficient. The concept of activity coefficient is closely linked to that of activity in chemistry. Thermodynamic definition The chemical potential, \mu_\mathrm, of a substance B in an ideal mixture of liquids or an ideal solution is given by :\mu_ ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Solubility Equilibrium
Solubility equilibrium is a type of dynamic equilibrium that exists when a chemical compound in the solid state is in chemical equilibrium with a solution of that compound. The solid may dissolve unchanged, with dissociation, or with chemical reaction with another constituent of the solution, such as acid or alkali. Each solubility equilibrium is characterized by a temperature-dependent ''solubility product'' which functions like an equilibrium constant. Solubility equilibria are important in pharmaceutical, environmental and many other scenarios. Definitions A solubility equilibrium exists when a chemical compound in the solid state is in chemical equilibrium with a solution containing the compound. This type of equilibrium is an example of dynamic equilibrium in that some individual molecules migrate between the solid and solution phases such that the rates of dissolution and precipitation are equal to one another. When equilibrium is established and the solid has not all dissolv ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Undersaturated
Undersaturation is a state of a solution that contains less of a dissolved material than could be dissolved by that quantity of solvent under normal circumstances. It can also refer to a vapor of a compound that has a lower (partial) pressure than the compound's vapor pressure Vapor pressure (or vapour pressure in English-speaking countries other than the US; see spelling differences) or equilibrium vapor pressure is defined as the pressure exerted by a vapor in thermodynamic equilibrium with its condensed phas .... Undersaturation is often followed by ingassing of the solvate until saturation is reached.{{cite journal, title=Aragonite undersaturation in the Arctic Ocean: effects of ocean acidification and sea ice melt, pmid=19965425 , doi=10.1126/science.1174190 , volume=326, issue=5956 , journal=Science, pages=1098–100 , last1 = Yamamoto-Kawai , first1 = M , last2 = McLaughlin , first2 = FA , last3 = Carmack , first3 = EC , last4 = Nishino , first4 = S , ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Calcium Phosphate
The term calcium phosphate refers to a family of materials and minerals containing calcium ions (Ca2+) together with inorganic phosphate anions. Some so-called calcium phosphates contain oxide and hydroxide as well. Calcium phosphates are white solids of nutritious value and are found in many living organisms, e.g., bone mineral and tooth enamel. In milk, it exists in a colloidal form in micelles bound to casein protein with magnesium, zinc, and citrate–collectively referred to as colloidal calcium phosphate (CCP). Various calcium phosphate minerals are used in the production of phosphoric acid and fertilizers. Overuse of certain forms of calcium phosphate can lead to nutrient-containing surface runoff and subsequent adverse effects upon receiving waters such as algal blooms and eutrophication (over-enrichment with nutrients and minerals). Orthophosphates, di- and monohydrogen phosphates These materials contain Ca2+ combined with , , or : * Monocalcium phosphate, E341 (CAS# 775 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |

_-Youth_(1893).jpg)