|
List Of Gases
This is a list of gases at standard conditions, which means substances that boil or sublime at or below 25 °C (77 °F) and 1 atm pressure and are reasonably stable. List This list is sorted by boiling point of gases in ascending order, but can be sorted on different values. "sub" and "triple" refer to the sublimation point and the triple point, which are given in the case of a substance that sublimes at 1 atm; "dec" refers to decomposition. "~" means approximately. Known as gas The following list has substances known to be gases, but with an unknown boiling point. *Fluoroamine * Trifluoromethyl trifluoroethyl trioxide CF3OOOCF2CF3 boils between 10 and 20° * Bis-trifluoromethyl carbonate boils between −10 and +10° possibly +12, freezing −60° * Difluorodioxirane boils between −80 and −90°. * Difluoroaminosulfinyl fluoride F2NS(O)F is a gas but decomposes over several hours * Trifluoromethylsulfinyl chloride CF3S(O)Cl *Nitrosyl cyanide ?−20° blu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Liquid Oxygen In A Beaker 4
A liquid is a nearly incompressible fluid that conforms to the shape of its container but retains a (nearly) constant volume independent of pressure. As such, it is one of the four fundamental states of matter (the others being solid, gas, and plasma), and is the only state with a definite volume but no fixed shape. A liquid is made up of tiny vibrating particles of matter, such as atoms, held together by intermolecular bonds. Like a gas, a liquid is able to flow and take the shape of a container. Most liquids resist compression, although others can be compressed. Unlike a gas, a liquid does not disperse to fill every space of a container, and maintains a fairly constant density. A distinctive property of the liquid state is surface tension, leading to wetting phenomena. Water is by far the most common liquid on Earth. The density of a liquid is usually close to that of a solid, and much higher than that of a gas. Therefore, liquid and solid are both termed condensed matter. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Krypton
Krypton (from grc, κρυπτός, translit=kryptos 'the hidden one') is a chemical element with the symbol Kr and atomic number 36. It is a colorless, odorless, tasteless noble gas that occurs in trace amounts in the atmosphere and is often used with other rare gases in fluorescent lamps. With rare exceptions, krypton is chemically inert. Krypton, like the other noble gases, is used in lighting and photography. Krypton light has many spectral lines, and krypton plasma is useful in bright, high-powered gas lasers (krypton ion and excimer lasers), each of which resonates and amplifies a single spectral line. Krypton fluoride also makes a useful laser medium. From 1960 to 1983, the official definition of meter was based on the wavelength of one spectral line of krypton-86, because of the high power and relative ease of operation of krypton discharge tubes. History Krypton was discovered in Britain in 1898 by William Ramsay, a Scottish chemist, and Morris Travers, an Engli ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Boron Trifluoride
Boron trifluoride is the inorganic compound with the formula BF3. This pungent, colourless, and toxic gas forms white fumes in moist air. It is a useful Lewis acid and a versatile building block for other boron compounds. Structure and bonding The geometry of a molecule of BF3 is trigonal planar. Its D3h symmetry conforms with the prediction of VSEPR theory. The molecule has no dipole moment by virtue of its high symmetry. The molecule is isoelectronic with the carbonate anion, . BF3 is commonly referred to as " electron deficient," a description that is reinforced by its exothermic reactivity toward Lewis bases. In the boron trihalides, BX3, the length of the B–X bonds (1.30 Å) is shorter than would be expected for single bonds, and this shortness may indicate stronger B–X π-bonding in the fluoride. A facile explanation invokes the symmetry-allowed overlap of a p orbital on the boron atom with the in-phase combination of the three similarly oriented p orbitals ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chlorine Fluoride
A chlorine fluoride is an interhalogen compound containing only chlorine and fluorine. {, class="wikitable" , - ! ! ClF ! ClF3 ! ClF5 , - , Systematic name , Chlorine monofluoride , Chlorine trifluoride , Chlorine pentafluoride , - , Molar mass , 54.45 g/mol , 92.45 g/mol , 130.45 g/mol , - , CAS number , , , {{CASREF, CAS=13637-63-3 , - , Melting point , −155.6 °C , −76.3 °C , −103 °C , - , Boiling point , −100 °C , 11.8 °C , −13.1 °C , - , Standard enthalpy of formation ΔfH°gas , −50.29 kJ/mol , −158.87 kJ/mol , −238.49 kJ/mol , - , Standard molar entropy S°gas , 217.91 J·K−1·mol−1 , 281.59 J·K−1·mol−1 , 310.73 J·K−1·mol−1 , - , Heat capacity Heat capacity or thermal capacity is a physical property of matter, defined as the amount of heat to be supplied to an object to produce a unit change in its temperature. The SI unit of heat capacity is joule per kelvin (J/K). Heat capacity ... Cp ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phosphorus(III) Fluoride
Phosphorus trifluoride (formula P F3), is a colorless and odorless gas. It is highly toxic and reacts slowly with water. Its main use is as a ligand in metal complexes. As a ligand, it parallels carbon monoxide in metal carbonyls, and indeed its toxicity is due to its binding with the iron in blood hemoglobin in a similar way to carbon monoxide. Physical properties Phosphorus trifluoride has an F−P−F bond angle of approximately 96.3°. Gaseous PF3 has a standard enthalpy of formation of −945 kJ/mol (−226 kcal/ mol). The phosphorus atom has a nuclear magnetic resonance chemical shift of 97 ppm (downfield of H3PO4). Properties Phosphorus trifluoride hydrolyzes especially at high pH, but it is less hydrolytically sensitive than phosphorus trichloride. It does not attack glass except at high temperatures, and anhydrous potassium hydroxide may be used to dry it with little loss. With hot metals, phosphides and fluorides are formed. With Lewis bases such as a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ethylene
Ethylene (IUPAC name: ethene) is a hydrocarbon which has the formula or . It is a colourless, flammable gas with a faint "sweet and musky" odour when pure. It is the simplest alkene (a hydrocarbon with carbon-carbon double bonds). Ethylene is widely used in the chemical industry, and its worldwide production (over 150 million tonnes in 2016) exceeds that of any other organic compound. Much of this production goes toward polyethylene, a widely used plastic containing polymer chains of ethylene units in various chain lengths. Ethylene is also an important natural plant hormone and is used in agriculture to force the ripening of fruits. The hydrate of ethylene is ethanol. Structure and properties This hydrocarbon has four hydrogen atoms bound to a pair of carbon atoms that are connected by a double bond. All six atoms that comprise ethylene are coplanar. The H-C-H angle is 117.4°, close to the 120° for ideal sp² hybridized carbon. The molecule is also relatively weak: rota ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cis-Difluorodiazine
Dinitrogen difluoride is a chemical compound with the formula N2F2. It is a gas at room temperature, and was first identified in 1952 as the thermal decomposition product of the azide N3F. It has the structure F−N=N−F and exists in both a ''cis''- and ''trans''-form. Isomers The ''cis'' configuration lies in a C2v symmetry and the ''trans''-form has a symmetry of C2h. These isomers are thermally interconvertible but can be separated by low temperature fractionation. The ''trans''-form is less thermodynamically stable but can be stored in glass vessels. The ''cis''-form attacks glass over a time scale of about 2 weeks to form silicon tetrafluoride and nitrous oxide: :2 N2F2 + SiO2 → SiF4 + 2 N2O Preparation Most preparations of dinitrogen difluoride give mixtures of the two isomers, but they can be prepared independently. An aqueous method involves ''N'',''N''-difluorourea with concentrated potassium hydroxide. This gives a 40% yield with three times more o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Xenon
Xenon is a chemical element with the symbol Xe and atomic number 54. It is a dense, colorless, odorless noble gas found in Earth's atmosphere in trace amounts. Although generally unreactive, it can undergo a few chemical reactions such as the formation of xenon hexafluoroplatinate, the first noble gas compound to be synthesized. Xenon is used in flash lamps and arc lamps, and as a general anesthetic. The first excimer laser design used a xenon dimer molecule (Xe2) as the lasing medium, and the earliest laser designs used xenon flash lamps as pumps. Xenon is also used to search for hypothetical weakly interacting massive particles and as a propellant for ion thrusters in spacecraft. Naturally occurring xenon consists of seven stable isotopes and two long-lived radioactive isotopes. More than 40 unstable xenon isotopes undergo radioactive decay, and the isotope ratios of xenon are an important tool for studying the early history of the Solar System. Radioactive xenon-135 is ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ozone
Ozone (), or trioxygen, is an inorganic molecule with the chemical formula . It is a pale blue gas with a distinctively pungent smell. It is an allotrope of oxygen that is much less stable than the diatomic allotrope , breaking down in the lower atmosphere to (dioxygen). Ozone is formed from dioxygen by the action of ultraviolet (UV) light and electrical discharges within the Earth's atmosphere. It is present in very low concentrations throughout the latter, with its highest concentration high in the ozone layer of the stratosphere, which absorbs most of the Sun's ultraviolet (UV) radiation. Ozone's odour is reminiscent of chlorine, and detectable by many people at concentrations of as little as in air. Ozone's O3 structure was determined in 1865. The molecule was later proven to have a bent structure and to be weakly diamagnetic. In standard conditions, ozone is a pale blue gas that condenses at cryogenic temperatures to a dark blue liquid and finally a violet-black soli ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Trans-Difluorodiazine
Dinitrogen difluoride is a chemical compound with the formula N2F2. It is a gas at room temperature, and was first identified in 1952 as the thermal decomposition product of the azide N3F. It has the structure F−N=N−F and exists in both a ''cis''- and ''trans''-form. Isomers The ''cis'' configuration lies in a C2v symmetry and the ''trans''-form has a symmetry of C2h. These isomers are thermally interconvertible but can be separated by low temperature fractionation. The ''trans''-form is less thermodynamically stable but can be stored in glass vessels. The ''cis''-form attacks glass over a time scale of about 2 weeks to form silicon tetrafluoride and nitrous oxide: :2 N2F2 + SiO2 → SiF4 + 2 N2O Preparation Most preparations of dinitrogen difluoride give mixtures of the two isomers, but they can be prepared independently. An aqueous method involves ''N'',''N''-difluorourea with concentrated potassium hydroxide. This gives a 40% yield with three times more o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Silane
Silane is an inorganic compound with chemical formula, . It is a colourless, pyrophoric, toxic gas with a sharp, repulsive smell, somewhat similar to that of acetic acid. Silane is of practical interest as a precursor to elemental silicon. Silane with alkyl groups are effective water repellents for mineral surfaces such as concrete and masonry. Silanes with both organic and inorganic attachments are used as coupling agents. Production Commercial-scale routes Silane can be produced by several routes. Typically, it arises from the reaction of hydrogen chloride with magnesium silicide: : Mg2Si + 4 HCl -> 2 MgCl2 + SiH4 It is also prepared from metallurgical-grade silicon in a two-step process. First, silicon is treated with hydrogen chloride at about 300 °C to produce trichlorosilane, HSiCl3, along with hydrogen gas, according to the chemical equation : Si + 3 HCl -> HSiCl3 + H2 The trichlorosilane is then converted to a mixture of silane and silicon tetrachloride: : 4 HS ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
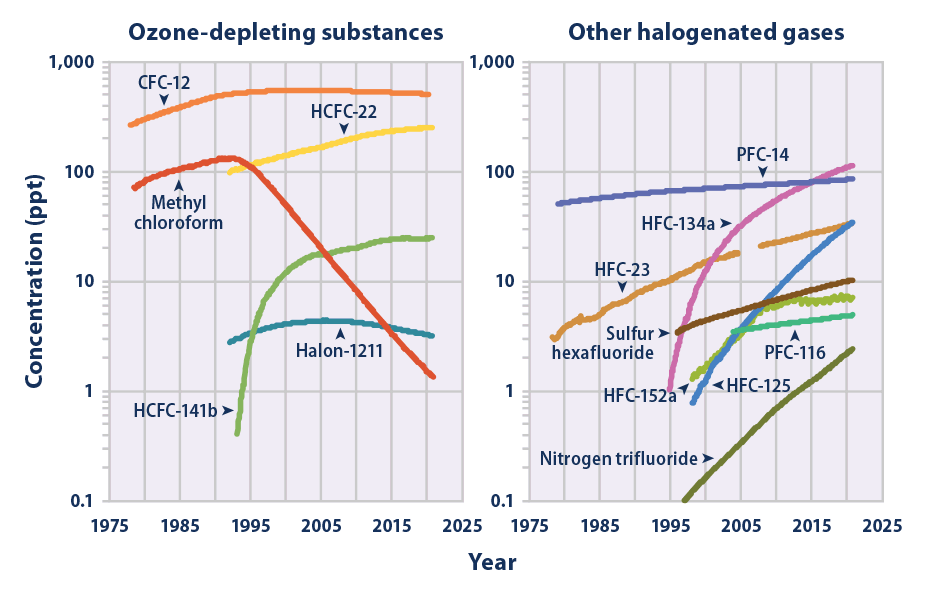
Nitrogen Trifluoride
Nitrogen trifluoride () is an inorganic, colorless, non-flammable, toxic gas with a slightly musty odor. It finds increasing use within the manufacturing of flat-panel displays, photovoltaics, LEDs and other microelectronics. Nitrogen trifluoride is also an extremely strong and long-lived greenhouse gas. Its atmospheric burden exceeded 2 parts per trillion during 2019 and has doubled every five years since the late 20th century. Synthesis and reactivity Nitrogen trifluoride did not exist in significant quantities on Earth prior to its synthesis by humans. It is a rare example of a binary fluoride that can be prepared directly from the elements only at very uncommon conditions, such as an electric discharge. After first attempting the synthesis in 1903, Otto Ruff prepared nitrogen trifluoride by the electrolysis of a molten mixture of ammonium fluoride and hydrogen fluoride. It proved to be far less reactive than the other nitrogen trihalides nitrogen trichloride, nitrogen trib ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |




platinum(0)-from-xtal-2008-3D-balls.png)