|
Lysozyme PEGylation
Lysozyme PEGylation is the covalent attachment of Polyethylene glycol (PEG) to Lysozyme, which is one of the most widely investigated PEGylated proteins. The PEGylation of proteins has become a common practice of modern therapeutic drugs, as the process is capable of enhancing solubility, thermal stability, enzymatic degradation resistance, and serum half-life of the proteins of interest. Lysozyme, as a natural bactericidal enzyme, lyses the cell wall of various gram-positive bacteria and offers protection against microbial infections. Lysozyme has six lysine residues which are accessible for PEGylation reactions. Thus, the PEGylation of lysozyme, or lysozyme PEGylation, can be a good model system for the PEGylation of other proteins with enzymatic activities by showing the enhancement of its physical and thermal stability while retaining its activity. Previous works on lysozyme PEGylation showed various chromatographic schemes in order to purify PEGylated lysozyme, which inc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polyethylene Glycol
Polyethylene glycol (PEG; ) is a polyether compound derived from petroleum with many applications, from industrial manufacturing to medicine. PEG is also known as polyethylene oxide (PEO) or polyoxyethylene (POE), depending on its molecular weight. The structure of PEG is commonly expressed as H−(O−CH2−CH2)n−OH. Uses Medical uses * Pharmaceutical-grade PEG is used as an excipient in many pharmaceutical products, in oral, topical, and parenteral dosage forms. * PEG is the basis of a number of laxatives (as ''MiraLax''). Whole bowel irrigation with polyethylene glycol and added electrolytes is used for bowel preparation before surgery or colonoscopy. * PEG is used in medicines for treating disimpaction and maintenance therapy for children with constipation. * When attached to various protein medications or drug carriers, polyethylene glycol of suitable length slows down their clearance from the blood. * The possibility that PEG could be used to fuse axons is being ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electrostatic Interaction
Electrostatics is a branch of physics that studies electric charges at rest (static electricity). Since classical times, it has been known that some materials, such as amber, attract lightweight particles after rubbing. The Greek word for amber, (), was thus the source of the word 'electricity'. Electrostatic phenomena arise from the forces that electric charges exert on each other. Such forces are described by Coulomb's law. Even though electrostatically induced forces seem to be rather weak, some electrostatic forces are relatively large. The force between an electron and a proton, which together make up a hydrogen atom, is about 36 orders of magnitude stronger than the gravitational force acting between them. There are many examples of electrostatic phenomena, from those as simple as the attraction of plastic wrap to one's hand after it is removed from a package, to the apparently spontaneous explosion of grain silos, the damage of electronic components during manufactur ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Optical Density
Absorbance is defined as "the logarithm of the ratio of incident to transmitted radiant power through a sample (excluding the effects on cell walls)". Alternatively, for samples which scatter light, absorbance may be defined as "the negative logarithm of one minus absorptance, as measured on a uniform sample". The term is used in many technical areas to quantify the results of an experimental measurement. While the term has its origin in quantifying the absorption of light, it is often entangled with quantification of light which is “lost” to a detector system through other mechanisms. What these uses of the term tend to have in common is that they refer to a logarithm of the ratio of a quantity of light incident on a sample or material to that which is detected after the light has interacted with the sample. The term absorption refers to the physical process of absorbing light, while absorbance does not always measure only absorption; it may measure attenuation (of trans ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Enzymatic Activity
Enzyme assays are laboratory methods for measuring enzymatic activity. They are vital for the study of enzyme kinetics and enzyme inhibition. Enzyme units The quantity or concentration of an enzyme can be expressed in molar amounts, as with any other chemical, or in terms of activity in enzyme units. Enzyme activity Enzyme activity is a measure of the quantity of active enzyme present and is thus dependent on various physical conditions, ''which should be specified''. It is calculated using the following formula: :\operatorname=\operatorname=\operatorname\times\operatorname where :\operatorname= Enzyme activity :\operatorname= Moles of substrate converted per unit time :\operatorname= Rate of the reaction :\operatorname= Reaction volume The SI unit is the katal, 1 katal = 1 mol s−1 (mole per second), but this is an excessively large unit. A more practical and commonly used value is enzyme unit (U) = 1 μmol min−1 (micromole per minute). 1 U corresponds to 16.67 nano ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Potassium Ferricyanide
Potassium ferricyanide is the chemical compound with the formula K3 e(CN)6 This bright red salt contains the octahedrally coordinated 3−.html" ;"title="e(CN)6sup>3−">e(CN)6sup>3− ion. It is soluble in water and its solution shows some green-yellow fluorescence. It was discovered in 1822 by Leopold Gmelin. Preparation Potassium ferricyanide is manufactured by passing chlorine through a solution of potassium ferrocyanide. Potassium ferricyanide separates from the solution: :2 K4 e(CN)6+ Cl2 → 2 K3 e(CN)6+ 2 KCl Structure Like other metal cyanides, solid potassium ferricyanide has a complicated polymeric structure. The polymer consists of octahedral e(CN)6sup>3− centers crosslinked with K+ ions that are bound to the CN ligands. The K+---NCFe linkages break when the solid is dissolved in water. Applications The compound is also used to harden iron and steel, in electroplating, dyeing wool, as a laboratory reagent, and as a mild oxidizing agent in organic chemist ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Enzymatic Activity
Enzyme assays are laboratory methods for measuring enzymatic activity. They are vital for the study of enzyme kinetics and enzyme inhibition. Enzyme units The quantity or concentration of an enzyme can be expressed in molar amounts, as with any other chemical, or in terms of activity in enzyme units. Enzyme activity Enzyme activity is a measure of the quantity of active enzyme present and is thus dependent on various physical conditions, ''which should be specified''. It is calculated using the following formula: :\operatorname=\operatorname=\operatorname\times\operatorname where :\operatorname= Enzyme activity :\operatorname= Moles of substrate converted per unit time :\operatorname= Rate of the reaction :\operatorname= Reaction volume The SI unit is the katal, 1 katal = 1 mol s−1 (mole per second), but this is an excessively large unit. A more practical and commonly used value is enzyme unit (U) = 1 μmol min−1 (micromole per minute). 1 U corresponds to 16.67 nano ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Matrix-assisted Laser Desorption/ionization
In mass spectrometry, matrix-assisted laser desorption/ionization (MALDI) is an ionization technique that uses a laser energy absorbing matrix to create ions from large molecules with minimal fragmentation. It has been applied to the analysis of biomolecules ( biopolymers such as DNA, proteins, peptides and carbohydrates) and various organic molecules (such as polymers, dendrimers and other macromolecules), which tend to be fragile and fragment when ionized by more conventional ionization methods. It is similar in character to electrospray ionization (ESI) in that both techniques are relatively soft (low fragmentation) ways of obtaining ions of large molecules in the gas phase, though MALDI typically produces far fewer multi-charged ions. MALDI methodology is a three-step process. First, the sample is mixed with a suitable matrix material and applied to a metal plate. Second, a pulsed laser irradiates the sample, triggering ablation and desorption of the sample and matrix materi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Retention Time
In chemical analysis, chromatography is a laboratory technique for the separation of a mixture into its components. The mixture is dissolved in a fluid solvent (gas or liquid) called the ''mobile phase'', which carries it through a system (a column, a capillary tube, a plate, or a sheet) on which a material called the ''stationary phase'' is fixed. Because the different constituents of the mixture tend to have different affinities for the stationary phase and are retained for different lengths of time depending on their interactions with its surface sites, the constituents travel at different apparent velocities in the mobile fluid, causing them to separate. The separation is based on the differential partitioning between the mobile and the stationary phases. Subtle differences in a compound's partition coefficient result in differential retention on the stationary phase and thus affect the separation. Chromatography may be preparative or analytical. The purpose of preparativ ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Molecular-weight
The molecular mass (''m'') is the mass of a given molecule: it is measured in daltons (Da or u). Different molecules of the same compound may have different molecular masses because they contain different isotopes of an element. The related quantity relative molecular mass, as defined by IUPAC, is the ratio of the mass of a molecule to the unified atomic mass unit (also known as the dalton) and is unitless. The molecular mass and relative molecular mass are distinct from but related to the molar mass. The molar mass is defined as the mass of a given substance divided by the amount of a substance and is expressed in g/mol. That makes the molar mass an average of many particles or molecules, and the molecular mass the mass of one specific particle or molecule. The molar mass is usually the more appropriate figure when dealing with macroscopic (weigh-able) quantities of a substance. The definition of molecular weight is most authoritatively synonymous with relative molecular mass; ho ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
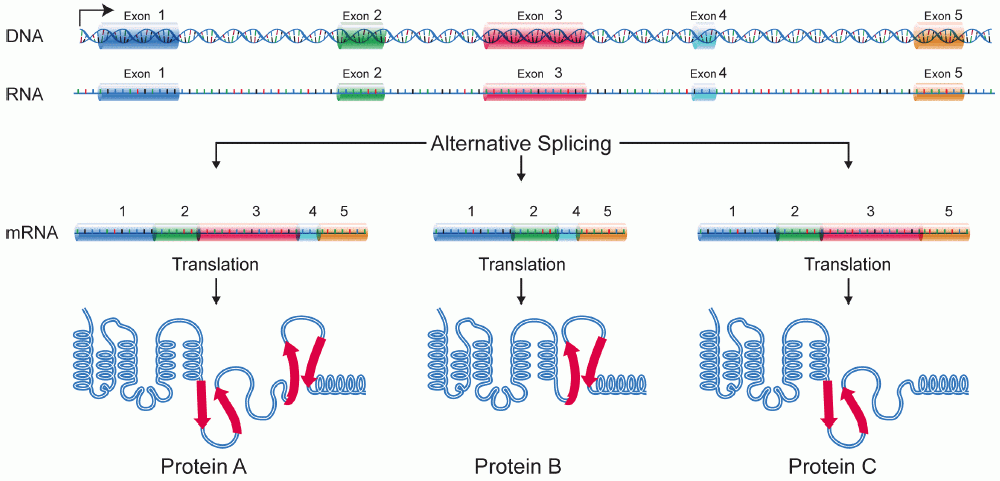
Protein Isoform
A protein isoform, or "protein variant", is a member of a set of highly similar proteins that originate from a single gene or gene family and are the result of genetic differences. While many perform the same or similar biological roles, some isoforms have unique functions. A set of protein isoforms may be formed from alternative splicings, variable promoter usage, or other post-transcriptional modifications of a single gene; post-translational modifications are generally not considered. (For that, see Proteoforms.) Through RNA splicing mechanisms, mRNA has the ability to select different protein-coding segments ( exons) of a gene, or even different parts of exons from RNA to form different mRNA sequences. Each unique sequence produces a specific form of a protein. The discovery of isoforms could explain the discrepancy between the small number of protein coding regions genes revealed by the human genome project and the large diversity of proteins seen in an organism: different ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrophobic Interaction Chromatography
In chemical analysis, chromatography is a laboratory technique for the separation of a mixture into its components. The mixture is dissolved in a fluid solvent (gas or liquid) called the ''mobile phase'', which carries it through a system (a column, a capillary tube, a plate, or a sheet) on which a material called the ''stationary phase'' is fixed. Because the different constituents of the mixture tend to have different affinities for the stationary phase and are retained for different lengths of time depending on their interactions with its surface sites, the constituents travel at different apparent velocities in the mobile fluid, causing them to separate. The separation is based on the differential partitioning between the mobile and the stationary phases. Subtle differences in a compound's partition coefficient result in differential retention on the stationary phase and thus affect the separation. Chromatography may be preparative or analytical. The purpose of preparativ ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |