|
Lithium Borohydride
Lithium borohydride (LiBH4) is a borohydride and known in organic synthesis as a reducing agent for esters. Although less common than the related sodium borohydride, the lithium salt offers some advantages, being a stronger reducing agent and highly soluble in ethers, whilst remaining safer to handle than lithium aluminium hydride.Luca Banfi, Enrica Narisano, Renata Riva, Ellen W. Baxter, "Lithium Borohydride" e-EROS Encyclopedia of Reagents for Organic Synthesis, 2001, John Wiley & Sons. . Preparation Lithium borohydride may be prepared by the metathesis reaction, which occurs upon ball-milling the more commonly available sodium borohydride and lithium bromide: : NaBH4 + LiBr → NaBr + LiBH4 Alternatively, it may be synthesized by treating boron trifluoride with lithium hydride in diethyl ether: : BF3 + 4 LiH → LiBH4 + 3 LiF Reactions Lithium borohydride is a stronger reducing agent than sodium borohydride. In mixtures of methanol and diethyl ether, lithium borohydride is a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemoselectivity
Chemoselectivity is the preferential outcome of a chemical reaction over a set of possible alternative reactions. In another definition, chemoselectivity refers to the selective reactivity of one functional group in the presence of others; often this process in convoluted and protecting groups are on the molecular connectivity alone. Such predictions based on connectivity are generally considered plausible, but the physical outcome of the actual reaction is ultimately dependent on a number of factors that are practically impossible to predict to any useful accuracy (solvent, atomic orbitals, etc.). Chemoselectivity can be difficult to predict, but observing selective outcomes in cases where many reactions are plausible, is common. Examples include the selective organic Redox, reduction of the greater relative chemoselectivity of sodium borohydride Redox, reduction versus lithium aluminium hydride Redox, reduction. In another example, the compound 4-methoxyacetophenone is oxidized ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Megajoule
The joule ( , ; symbol: J) is the unit of energy in the International System of Units (SI). It is equal to the amount of work done when a force of 1 newton displaces a mass through a distance of 1 metre in the direction of the force applied. It is also the energy dissipated as heat when an electric current of one ampere passes through a resistance of one ohm for one second. It is named after the English physicist James Prescott Joule (1818–1889). Definition In terms of SI base units and in terms of SI derived units with special names, the joule is defined as One joule can also be defined by any of the following: * The work required to move an electric charge of one coulomb through an electrical potential difference of one volt, or one coulomb-volt (C⋅V). This relationship can be used to define the volt. * The work required to produce one watt of power for one second, or one watt-second (W⋅s) (compare kilowatt-hour, which is 3.6 megajoules). This relationship can be ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Energy Carrier
An energy carrier is a substance (fuel) or sometimes a phenomenon (energy system) that contains energy that can be later converted to other forms such as mechanical work or heat or to operate chemical or physical processes. Such carriers include springs, electrical batteries, capacitors, pressurized air, dammed water, hydrogen, petroleum, coal, wood, and natural gas. An energy carrier does not produce energy; it simply contains energy imbued by another system. Definition according to ISO 13600 According to ISO 13600, an energy carrier is either a substance or a phenomenon that can be used to produce mechanical work or heat or to operate chemical or physical processes. It is any system or substance that contains energy for conversion as usable energy later or somewhere else. This could be converted for use in, for example, an appliance or vehicle. Such carriers include springs, electrical batteries, capacitors, pressurized air, dammed water, hydrogen, petroleum, coal, wo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Energy Density
In physics, energy density is the amount of energy stored in a given system or region of space per unit volume. It is sometimes confused with energy per unit mass which is properly called specific energy or . Often only the ''useful'' or extractable energy is measured, which is to say that inaccessible energy (such as rest mass energy) is ignored. In cosmological and other general relativistic contexts, however, the energy densities considered are those that correspond to the elements of the stress–energy tensor and therefore do include mass energy as well as energy densities associated with pressure. Energy per unit volume has the same physical units as pressure and in many situations is synonymous. For example, the energy density of a magnetic field may be expressed as and behaves like a physical pressure. Likewise, the energy required to compress a gas to a certain volume may be determined by multiplying the difference between the gas pressure and the external pressure by ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
4002726 Schematics
4 (four) is a number, numeral and digit. It is the natural number following 3 and preceding 5. It is the smallest semiprime and composite number, and is considered unlucky in many East Asian cultures. In mathematics Four is the smallest composite number, its proper divisors being and . Four is the sum and product of two with itself: 2 + 2 = 4 = 2 x 2, the only number b such that a + a = b = a x a, which also makes four the smallest squared prime number p^. In Knuth's up-arrow notation, , and so forth, for any number of up arrows. By consequence, four is the only square one more than a prime number, specifically three. The sum of the first four prime numbers two + three + five + seven is the only sum of four consecutive prime numbers that yields an odd prime number, seventeen, which is the fourth super-prime. Four lies between the first proper pair of twin primes, three and five, which are the first two Fermat primes, like seventeen, which is the third. On the other ha ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tertiary (chemistry)
Tertiary is a term used in organic chemistry to classify various types of compounds (e. g. alcohols, alkyl halides, amines) or reactive intermediates (e. g. alkyl radicals, carbocations). See also * Primary (chemistry) * Secondary (chemistry) * Quaternary (chemistry) Quaternary is a term used in organic chemistry to classify various types of compounds (e. g. amines and ammonium salts).Paula Yurkanis Bruice: ''Organic Chemistry'', Pearson Education Inc., 2004, 4. Ed., p. 78, 104, 893, and 912, . {{clear See ... References {{Navbox stereochemistry Chemical nomenclature ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
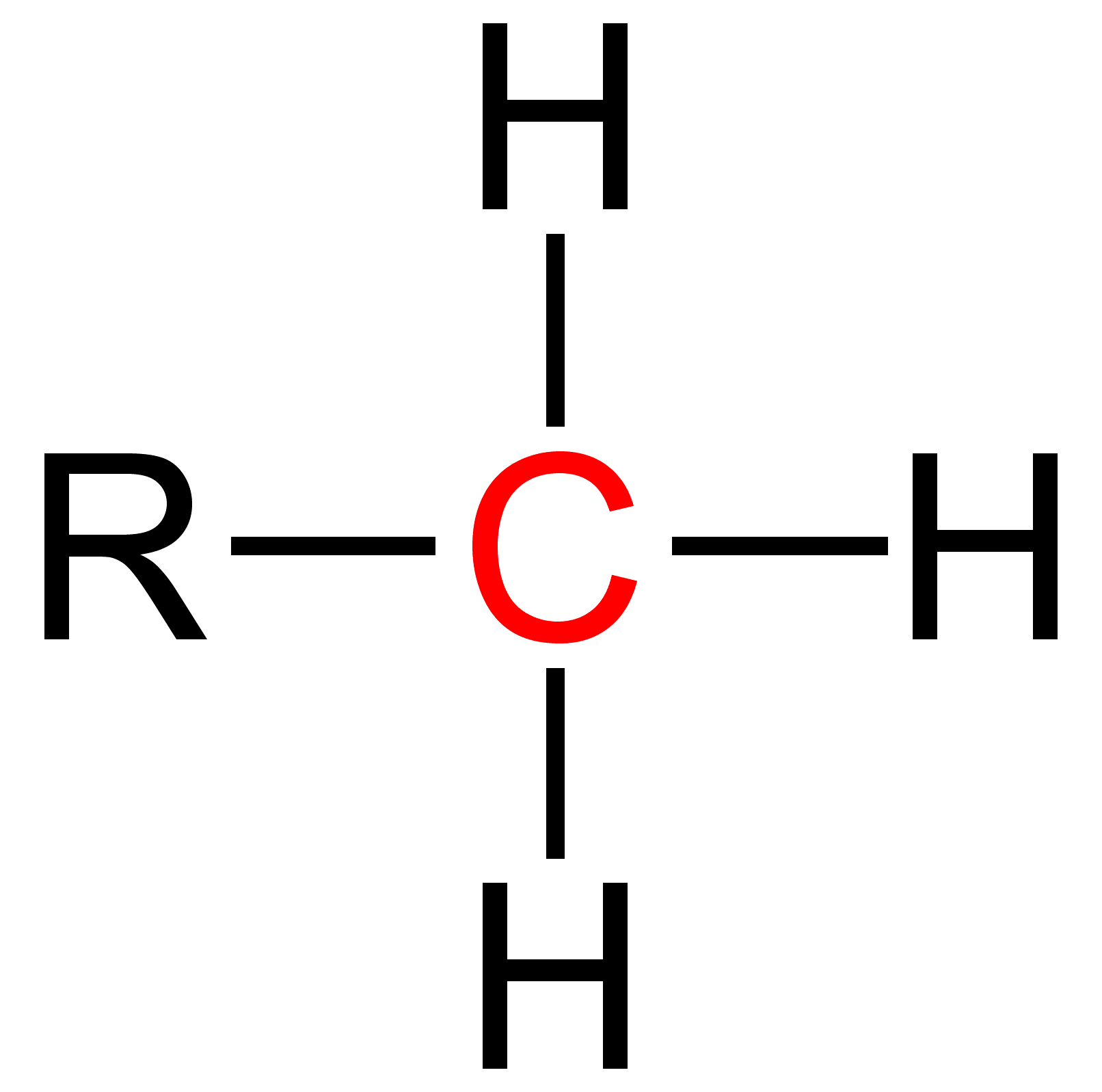
Secondary (chemistry)
Secondary is a term used in organic chemistry to classify various types of compounds (e. g. alcohols, alkyl halides, amines) or reactive intermediates (e. g. alkyl radicals, carbocations). An atom is considered secondary if it has two 'R' Groups attached to it. An 'R' group is a carbon containing group such as a methyl (CH3 ). A secondary compound is most often classified on an alpha carbon (middle carbon) or a nitrogen. The word secondary comes from the root word 'second' which means two. This nomenclature can be used in many cases and further used to explain relative reactivity. The reactivity of molecules varies with respect to the attached atoms. Thus, a primary, secondary, tertiary and quaternary molecule of the same function group will have different reactivities. Secondary Alcohols Secondary alcohols are identified by the groups attached to the central carbon that is bonding with the alcohol (-OH) group. A secondary alcohol is a carbon with four substituents includin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkyl Halides
The haloalkanes (also known as halogenoalkanes or alkyl halides) are alkanes containing one or more halogen substituents. They are a subset of the general class of halocarbons, although the distinction is not often made. Haloalkanes are widely used commercially. They are used as flame retardants, fire extinguishants, refrigerants, propellants, solvents, and pharmaceuticals. Subsequent to the widespread use in commerce, many halocarbons have also been shown to be serious pollutants and toxins. For example, the chlorofluorocarbons have been shown to lead to ozone depletion. Methyl bromide is a controversial fumigant. Only haloalkanes that contain chlorine, bromine, and iodine are a threat to the ozone layer, but fluorinated volatile haloalkanes in theory may have activity as greenhouse gases. Methyl iodide, a naturally occurring substance, however, does not have ozone-depleting properties and the United States Environmental Protection Agency has designated the compound a non-ozone l ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbamic Acid
Carbamic acid, which might also be called aminoformic acid or aminocarboxylic acid, is the chemical compound with the formula . It can be obtained by the reaction of ammonia and carbon dioxide at very low temperatures, which also yields an equal amount of ammonium carbamate . The compound is stable only up to about 250 K (−23 °C); at higher temperatures it decomposes into those two gases. The solid apparently consists of dimers, with the two molecules connected by hydrogen bonds between the two carboxyl groups –COOH.J. B. Bossa, P. Theulé, F. Duvernay, F. Borget and T. Chiavassa (2008): "Carbamic acid and carbamate formation in NH3:CO2 ices – UV irradiation versus thermal processes". ''Astronomy and Astrophysics'', volume 492, issue 3, pages 719-724. Carbamic acid could be seen as both an amine and carboxylic acid, and therefore an amino acid;R. K. Khanna and M. H. Moore (1999): "Carbamic acid: molecular structure and IR spectra". ''Spectrochimica Acta Part A: ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitro Group
In organic chemistry, nitro compounds are organic compounds that contain one or more nitro functional groups (). The nitro group is one of the most common explosophores (functional group that makes a compound explosive) used globally. The nitro group is also strongly electron-withdrawing. Because of this property, bonds alpha (adjacent) to the nitro group can be acidic. For similar reasons, the presence of nitro groups in aromatic compounds retards electrophilic aromatic substitution but facilitates nucleophilic aromatic substitution. Nitro groups are rarely found in nature. They are almost invariably produced by nitration reactions starting with nitric acid. Synthesis Preparation of aromatic nitro compounds Aromatic nitro compounds are typically synthesized by nitration. Nitration is achieved using a mixture of nitric acid and sulfuric acid, which produce the nitronium ion (), which is the electrophile: + The nitration product produced on the la ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
.png)