|
Liebeskind–Srogl Coupling
The Liebeskind–Srogl coupling reaction is an organic reaction forming a new carbon–carbon bond from a thioester and a boronic acid using a metal catalyst. It is a cross-coupling reaction. This reaction was invented by and named after Jiri Srogl from the Academy of Sciences, Czech Republic, and Lanny S. Liebeskind from Emory University, Atlanta, Georgia, USA. There are three generations of this reaction, with the first generation shown below. The original transformation used catalytic Pd(0), TFP = tris(2-furyl)phosphine as an additional ligand and stoichiometric Copper(I)-thiophene-2-carboxylate, CuTC = copper(I) thiophene-2-carboxylate as a co-metal catalyst. The overall reaction scheme is shown below. Liebeskind-Srogl reaction is most commonly seen with sulfide or thioester electrophiles and boronic acid or stannane nucleophiles but many other coupling partners are viable. In addition to alkyl and aryl thioesters; (hetero)aryl sulfides, thioamides, sulfanyl alkynes, and thi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organic Reaction
Organic reactions are chemical reactions involving organic compounds. The basic organic chemistry reaction types are addition reactions, elimination reactions, substitution reactions, pericyclic reactions, rearrangement reactions, Mechanistic Organic Photochemistry, photochemical reactions and organic redox reaction, redox reactions. In organic synthesis, organic reactions are used in the construction of new organic molecules. The production of many man-made chemicals such as drugs, plastics, food additives, fabrics depend on organic reactions. The oldest organic reactions are combustion of organic fuels and saponification of fats to make soap. Modern organic chemistry starts with the Wöhler synthesis in 1828. In the history of the Nobel Prize in Chemistry awards have been given for the invention of specific organic reactions such as the Grignard reaction in 1912, the Diels-Alder reaction in 1950, the Wittig reaction in 1979 and olefin metathesis in 2005. Classifications Organic c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxidative Insertion
Oxidative addition and reductive elimination are two important and related classes of reactions in organometallic chemistry. Oxidative addition is a process that increases both the oxidation state and coordination number of a metal centre. Oxidative addition is often a step in catalytic cycles, in conjunction with its reverse reaction, reductive elimination. Role in transition metal chemistry For transition metals, oxidative reaction results in the decrease in the d''n'' to a configuration with fewer electrons, often 2e fewer. Oxidative addition is favored for metals that are (i) basic and/or (ii) easily oxidized. Metals with a relatively low oxidation state often satisfy one of these requirements, but even high oxidation state metals undergo oxidative addition, as illustrated by the oxidation of Pt(II) with chlorine: : tCl4sup>2− + Cl2 → tCl6sup>2− In classical organometallic chemistry, the formal oxidation state of the metal and the electron count of the complex both in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Terpene
Terpenes () are a class of natural products consisting of compounds with the formula (C5H8)n for n > 1. Comprising more than 30,000 compounds, these unsaturated hydrocarbons are produced predominantly by plants, particularly conifers. Terpenes are further classified by the number of carbons: monoterpenes (C10), sesquiterpenes (C15), diterpenes (C20), as examples. The terpene alpha-pinene, is a major component of the common solvent, turpentine. History and terminology The term ''terpene'' was coined in 1866 by the German chemist August Kekulé to denote all hydrocarbons having the empirical formula C10H16, of which camphene was one. Previously, many hydrocarbons having the empirical formula C10H16 had been called "camphene", but many other hydrocarbons of the same composition had had different names. Kekulé coined the term "terpene" in order to reduce the confusion. The name "terpene" is a shortened form of "terpentine", an obsolete spelling of "turpentine". Although sometimes ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Viridin
Viridin is an antifungal metabolite of ''Gliocladium virens'' that was first reported in 1945. Belonging to a class of molecules known as furanosteroids, it has a characteristic highly strained electrophilic furan ring fused between C-4 and C-6 of the steroid framework. Members of this family, including wortmannin, are known to be potent, irreversible covalent inhibitors of phosphoinositide 3-kinases (PI3Ks). Biosynthesis The complete biosynthesis of viridin is not known. Through -14Cevalonic acid labeling studies, it has been shown that viridin is biosynthesized from a triterpenoid pathway, which starts from squalene, rather than a diterpenoid pathway. Incorporation of lanosterol into viridin has been described. Although the mechanisms and order of the remaining steps from lanosterol to viridin are not certain, these include the formation of the furan ring, the loss of C-13 and C-14 methyl groups, aromatization of the C-ring, removal of the side chain, and oxidation of the A- ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Viridin
Viridin is an antifungal metabolite of ''Gliocladium virens'' that was first reported in 1945. Belonging to a class of molecules known as furanosteroids, it has a characteristic highly strained electrophilic furan ring fused between C-4 and C-6 of the steroid framework. Members of this family, including wortmannin, are known to be potent, irreversible covalent inhibitors of phosphoinositide 3-kinases (PI3Ks). Biosynthesis The complete biosynthesis of viridin is not known. Through -14Cevalonic acid labeling studies, it has been shown that viridin is biosynthesized from a triterpenoid pathway, which starts from squalene, rather than a diterpenoid pathway. Incorporation of lanosterol into viridin has been described. Although the mechanisms and order of the remaining steps from lanosterol to viridin are not certain, these include the formation of the furan ring, the loss of C-13 and C-14 methyl groups, aromatization of the C-ring, removal of the side chain, and oxidation of the A- ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Total Synthesis
Total synthesis is the complete chemical synthesis of a complex molecule, often a natural product, from simple, commercially-available precursors. It usually refers to a process not involving the aid of biological processes, which distinguishes it from semisynthesis. Syntheses may sometimes conclude at a precursor with further known synthetic pathways to a target molecule, in which case it is known as a formal synthesis. Total synthesis target molecules can be natural products, medicinally-important active ingredients, known intermediates, or molecules of theoretical interest. Total synthesis targets can also be organometallic or inorganic, though these are rarely encountered. Total synthesis projects often require a wide diversity of reactions and reagents, and subsequently requires broad chemical knowledge and training to be successful. Often, the aim is to discover a new route of synthesis for a target molecule for which there already exist known routes. Sometimes, however, no ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Metallothionein
Metallothionein (MT) is a family of cysteine-rich, low molecular weight (MW ranging from 500 to 14000 Da) proteins. They are localized to the membrane of the Golgi apparatus. MTs have the capacity to bind both physiological (such as zinc, copper, selenium) and xenobiotic (such as cadmium, mercury, silver, arsenic, lead) heavy metals through the thiol group of its cysteine residues, which represent nearly 30% of its constituent amino acid residues. MT was discovered in 1957 by Vallee and Margoshe from purification of a Cd-binding protein from horse (equine) renal cortex. MT plays a role in the protection against metal toxicity and oxidative stress, and is involved in zinc and copper regulation. There are four main isoforms expressed in humans (family 1, see chart below): MT1 (subtypes A, B, E, F, G, H, L, M, X), MT2, MT3, and MT4. In the human body, large quantities are synthesised primarily in the liver and kidneys. Their production is dependent on availability of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gen2bitch
Gen may refer to: * ''Gen'' (film), 2006 Turkish horror film directed by Togan Gökbakar * Gen (Street Fighter), a video game character from the ''Street Fighter'' series * Gen Fu, a video game character from the ''Dead or Alive'' series * Gen language, the language of Togo * Gen-san, a character in the anime series '' Sky Girls'' *, Japanese Nordic combined skier *, Japanese singer-songwriter, musician, actor, and writer *, Japanese footballer *, Japanese novelist, visual novel writer and anime screenwriter *, Japanese engineer and businessman *, Japanese politician *, Japanese ballet dancer and choreographer * Generation as in GenX, GenZ, etc. Gen. or GEN may refer to: * General officer, a high senior rank in the military * GEN Corporation, of Japan * Gen Digital, a computer security software company in United States * GEN Energija, a state-owned power company in Slovenia * GEN, a website published by Medium * Global Ecovillage Network * Global Editors Network * '' Gew ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Reductive Elimination
Reductive elimination is an elementary step in organometallic chemistry in which the oxidation state of the metal center decreases while forming a new covalent bond between two ligands. It is the microscopic reverse of oxidative addition, and is often the product-forming step in many catalytic processes. Since oxidative addition and reductive elimination are reverse reactions, the same mechanisms apply for both processes, and the product equilibrium depends on the thermodynamics of both directions. General information Reductive elimination is often seen in higher oxidation states, and can involve a two-electron change at a single metal center (mononuclear) or a one-electron change at each of two metal centers (binuclear, dinuclear, or bimetallic). For mononuclear reductive elimination, the oxidation state of the metal decreases by two, while the d-electron count of the metal increases by two. This pathway is common for d8 metals Ni(II), Pd(II), and Au(III) and d6 metals Pt(I ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
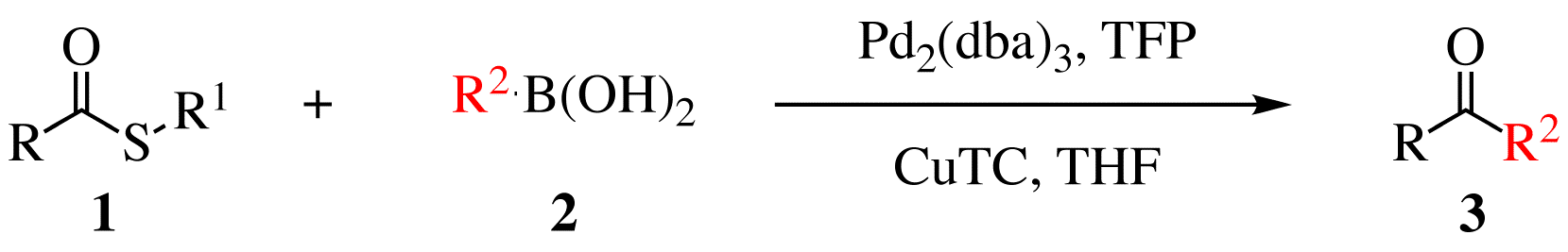




_with_Cd-Cd_bond.jpg)
