|
Lamellipodia
The lamellipodium (plural lamellipodia) (from Latin ''lamella'', related to ', "thin sheet", and the Greek radical ''pod-'', "foot") is a cytoskeletal protein actin projection on the leading edge of the cell. It contains a quasi-two-dimensional actin mesh; the whole structure propels the cell across a substrate. Within the lamellipodia are ribs of actin called microspikes, which, when they spread beyond the lamellipodium frontier, are called filopodia. The lamellipodium is born of actin nucleation in the plasma membrane of the cell and is the primary area of actin incorporation or microfilament formation of the cell. Description Lamellipodia are found primarily in all mobile cells, such as the keratinocytes of fish and frogs, which are involved in the quick repair of wounds. The lamellipodia of these keratinocytes allow them to move at speeds of 10–20 μm / min over epithelial surfaces. When separated from the main part of a cell, a lamellipodium can still c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cortactin
Cortactin (from "''cortical actin'' binding protein") is a monomeric protein located in the cytoplasm of cells that can be activated by external stimuli to promote polymerization and rearrangement of the actin cytoskeleton, especially the actin cortex around the cellular periphery. It is present in all cell types. When activated, it will recruit Arp2/3 complex proteins to existing actin microfilaments, facilitating and stabilizing nucleation sites for actin branching. Cortactin is important in promoting lamellipodia formation, invadopodia formation, cell migration, and endocytosis. Gene In humans, cortactin is encoded by the ''CTTN'' gene on chromosome 11. Structure Cortactin is a thin, elongated monomer that consists of an amino-terminal acidic (NTA) region; 37-residue-long segments that are highly conserved among cortactin proteins of all species and repeated up to 6.5 times in tandem (“cortactin repeats”); a proline-rich region; and an SH3 domain. This basic structure ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
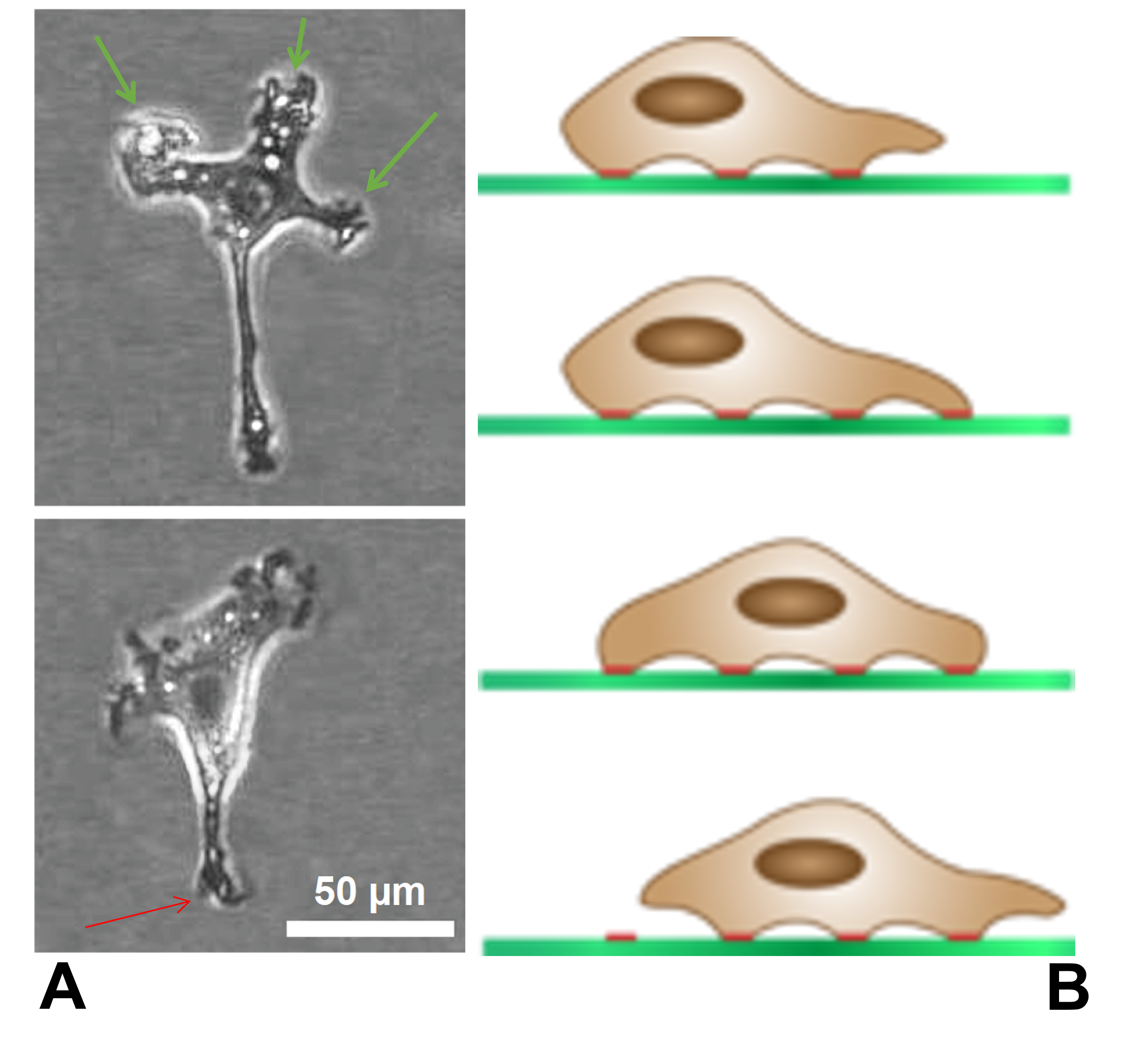
Microspikes
Filopodia (singular filopodium) are slender cytoplasmic projections that extend beyond the leading edge of lamellipodia in migrating cells. Within the lamellipodium, actin ribs are known as ''microspikes'', and when they extend beyond the lamellipodia, they're known as filopodia. They contain microfilaments (also called actin filaments) cross-linked into bundles by actin-bundling proteins, such as fascin and fimbrin. Filopodia form focal adhesions with the substratum, linking them to the cell surface. Many types of migrating cells display filopodia, which are thought to be involved in both sensation of chemotropic cues, and resulting changes in directed locomotion. Activation of the Rho family of GTPases, particularly cdc42 and their downstream intermediates, results in the polymerization of actin fibers by Ena/Vasp homology proteins. Growth factors bind to receptor tyrosine kinases resulting in the polymerization of actin filaments, which, when cross-linked, make up the sup ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Filopodia
Filopodia (singular filopodium) are slender cytoplasmic projections that extend beyond the leading edge of lamellipodia in migrating cells. Within the lamellipodium, actin ribs are known as ''microspikes'', and when they extend beyond the lamellipodia, they're known as filopodia. They contain microfilaments (also called actin filaments) cross-linked into bundles by actin-bundling proteins, such as fascin and fimbrin. Filopodia form focal adhesions with the substratum, linking them to the cell surface. Many types of migrating cells display filopodia, which are thought to be involved in both sensation of chemotropic cues, and resulting changes in directed locomotion. Activation of the Rho family of GTPases, particularly cdc42 and their downstream intermediates, results in the polymerization of actin fibers by Ena/Vasp homology proteins. Growth factors bind to receptor tyrosine kinases resulting in the polymerization of actin filaments, which, when cross-linked, make up the sup ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Wound Healing
Wound healing refers to a living organism's replacement of destroyed or damaged tissue by newly produced tissue. In undamaged skin, the epidermis (surface, epithelial layer) and dermis (deeper, connective layer) form a protective barrier against the external environment. When the barrier is broken, a regulated sequence of biochemical events is set into motion to repair the damage. This process is divided into predictable phases: blood clotting (hemostasis), inflammation, tissue growth ( cell proliferation), and tissue remodeling (maturation and cell differentiation). Blood clotting may be considered to be part of the inflammation stage instead of a separate stage. The wound healing process is not only complex but fragile, and it is susceptible to interruption or failure leading to the formation of non-healing chronic wounds. Factors that contribute to non-healing chronic wounds are diabetes, venous or arterial disease, infection, and metabolic deficiencies of old age.Enoch, S ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cytoskeleton
The cytoskeleton is a complex, dynamic network of interlinking protein filaments present in the cytoplasm of all cells, including those of bacteria and archaea. In eukaryotes, it extends from the cell nucleus to the cell membrane and is composed of similar proteins in the various organisms. It is composed of three main components, microfilaments, intermediate filaments and microtubules, and these are all capable of rapid growth or disassembly dependent on the cell's requirements. A multitude of functions can be performed by the cytoskeleton. Its primary function is to give the cell its shape and mechanical resistance to deformation, and through association with extracellular connective tissue and other cells it stabilizes entire tissues. The cytoskeleton can also contract, thereby deforming the cell and the cell's environment and allowing cells to migrate. Moreover, it is involved in many cell signaling pathways and in the uptake of extracellular material ( endocytosis), the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Microfilament
Microfilaments, also called actin filaments, are protein filaments in the cytoplasm of eukaryotic cells that form part of the cytoskeleton. They are primarily composed of polymers of actin, but are modified by and interact with numerous other proteins in the cell. Microfilaments are usually about 7 nm in diameter and made up of two strands of actin. Microfilament functions include cytokinesis, amoeboid movement, cell motility, changes in cell shape, endocytosis and exocytosis, cell contractility, and mechanical stability. Microfilaments are flexible and relatively strong, resisting buckling by multi-piconewton compressive forces and filament fracture by nanonewton tensile forces. In inducing cell motility, one end of the actin filament elongates while the other end contracts, presumably by myosin II molecular motors. Additionally, they function as part of actomyosin-driven contractile molecular motors, wherein the thin filaments serve as tensile platforms for myosin's ATP-de ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Actin
Actin is a family of globular multi-functional proteins that form microfilaments in the cytoskeleton, and the thin filaments in muscle fibrils. It is found in essentially all eukaryotic cells, where it may be present at a concentration of over 100 μM; its mass is roughly 42 kDa, with a diameter of 4 to 7 nm. An actin protein is the monomeric subunit of two types of filaments in cells: microfilaments, one of the three major components of the cytoskeleton, and thin filaments, part of the contractile apparatus in muscle cells. It can be present as either a free monomer called G-actin (globular) or as part of a linear polymer microfilament called F-actin (filamentous), both of which are essential for such important cellular functions as the mobility and contraction of cells during cell division. Actin participates in many important cellular processes, including muscle contraction, cell motility, cell division and cytokinesis, vesicle and organelle movement, cell sign ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cell (biology)
The cell is the basic structural and functional unit of life forms. Every cell consists of a cytoplasm enclosed within a membrane, and contains many biomolecules such as proteins, DNA and RNA, as well as many small molecules of nutrients and metabolites.Cell Movements and the Shaping of the Vertebrate Body in Chapter 21 of Molecular Biology of the Cell '' fourth edition, edited by Bruce Alberts (2002) published by Garland Science. The Alberts text discusses how the "cellular building blocks" move to shape developing embryos. It is also common to describe small molecules such as ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Arp2/3 Complex
Arp2/3 complex (Actin Related Protein 2/3 complex) is a seven-subunit protein complex that plays a major role in the regulation of the actin cytoskeleton. It is a major component of the actin cytoskeleton and is found in most actin cytoskeleton-containing eukaryotic cells. Two of its subunits, the ''A''ctin-''R''elated ''P''roteins ARP2 and ARP3, closely resemble the structure of monomeric actin and serve as nucleation sites for new actin filaments. The complex binds to the sides of existing ("mother") filaments and initiates growth of a new ("daughter") filament at a distinctive 70 degree angle from the mother. Branched actin networks are created as a result of this nucleation of new filaments. The regulation of rearrangements of the actin cytoskeleton is important for processes like cell locomotion, phagocytosis, and intracellular motility of lipid vesicles. The Arp2/3 complex was named after it was identified in 1994 by affinity chromatography from ''Acanthamoeba castellanii' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Adenosine Triphosphate
Adenosine triphosphate (ATP) is an organic compound that provides energy to drive many processes in living cells, such as muscle contraction, nerve impulse propagation, condensate dissolution, and chemical synthesis. Found in all known forms of life, ATP is often referred to as the "molecular unit of currency" of intracellular energy transfer. When consumed in metabolic processes, it converts either to adenosine diphosphate (ADP) or to adenosine monophosphate (AMP). Other processes regenerate ATP. The human body recycles its own body weight equivalent in ATP each day. It is also a precursor to DNA and RNA, and is used as a coenzyme. From the perspective of biochemistry, ATP is classified as a nucleoside triphosphate, which indicates that it consists of three components: a nitrogenous base (adenine), the sugar ribose, and the Polyphosphate, triphosphate. Structure ATP consists of an adenine attached by the 9-nitrogen atom to the 1′ carbon atom of a sugar (ribose), which i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Adenosine Diphosphate
Adenosine diphosphate (ADP), also known as adenosine pyrophosphate (APP), is an important organic compound in metabolism and is essential to the flow of energy in living cells. ADP consists of three important structural components: a sugar backbone attached to adenine and two phosphate groups bonded to the 5 carbon atom of ribose. The diphosphate group of ADP is attached to the 5’ carbon of the sugar backbone, while the adenine attaches to the 1’ carbon. ADP can be interconverted to adenosine triphosphate (ATP) and adenosine monophosphate (AMP). ATP contains one more phosphate group than does ADP. AMP contains one fewer phosphate group. Energy transfer used by all living things is a result of dephosphorylation of ATP by enzymes known as ATPases. The cleavage of a phosphate group from ATP results in the coupling of energy to metabolic reactions and a by-product of ADP. ATP is continually reformed from lower-energy species ADP and AMP. The biosynthesis of ATP is achieved through ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Frontiers In Bioscience
''Frontiers in Bioscience'' is a peer reviewed scientific journal. It was established in 1996 and covers all biological and medical sciences. The editor-in-chief is Graham Pawelec. The journal consists of three sections: ''Landmark Edition'', ''Scholar Edition'', and ''Elite Edition'' Abstracting and indexing The ''Landmark Edition'' of the journal is abstracted and indexed in: Reception According to the ''Journal Citation Reports'', the journal has a 2019 impact factor of 2.747. The journal has been placed on Jeffrey Beall Jeffrey Beall is an American librarian and library scientist, best known for drawing attention to "predatory open access publishing", a term he coined, and for creating what is now widely known as Beall's list, a list of potentially predatory op ...'s list of "potential, possible, or probable" predatory scholarly open-access publishers in 2012. References External links * {{DEFAULTSORT:Frontiers in Bioscience Biology journals Monthly journals Hybr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |