|
L-quisqualic Acid
Quisqualic acid is an agonist of the AMPA, kainate, and group I metabotropic glutamate receptors. It is one of the most potent AMPA receptor agonists known. It causes excitotoxicity and is used in neuroscience to selectively destroy neurons in the brain or spinal cord. Quisqualic acid occurs naturally in the seeds of ''Quisqualis'' species. Research conducted by the USDA Agricultural Research Service, has demonstrated quisqualic acid is also present within the flower petals of zonal geranium (''Pelargonium x hortorum'') and is responsible for causing rigid paralysis of the Japanese beetle. Quisqualic acid is thought to mimic L-glutamic acid, which is a neurotransmitter in the insect neuromuscular junction and mammalian central nervous system. See also * Quisqualamine * Non-proteinogenic amino acids In biochemistry, non-coded or non-proteinogenic amino acids are distinct from the 22 proteinogenic amino acids (21 in eukaryotesplus formylmethionine in eukaryotes with prokaryote ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Agonist
An agonist is a chemical that activates a receptor to produce a biological response. Receptors are cellular proteins whose activation causes the cell to modify what it is currently doing. In contrast, an antagonist blocks the action of the agonist, while an inverse agonist causes an action opposite to that of the agonist. Etymology From the Greek αγωνιστής (agōnistēs), contestant; champion; rival < αγων (agōn), contest, combat; exertion, struggle < αγω (agō), I lead, lead towards, conduct; drive Types of agonists can be activated by either endogenous agonists (such as |
Non-proteinogenic Amino Acids
In biochemistry, non-coded or non-proteinogenic amino acids are distinct from the 22 proteinogenic amino acids (21 in eukaryotesplus formylmethionine in eukaryotes with prokaryote organelles like mitochondria) which are naturally encoded in the genome of organisms for the assembly of proteins. However, over 140 non-proteinogenic amino acids occur naturally in proteins and thousands more may occur in nature or be synthesized in the laboratory. Chemically synthesized amino acids can be called unnatural amino acids. Unnatural amino acids can be synthetically prepared from their native analogs via modifications such as amine alkylation, side chain substitution, structural bond extension cyclization, and isosteric replacements within the amino acid backbone. Many non-proteinogenic amino acids are important: * intermediates in biosynthesis, * in post-translational formation of proteins, * in a physiological role (e.g. components of bacterial cell walls, neurotransmitters and toxins), * ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Toxic Amino Acids
Toxicity is the degree to which a chemical substance or a particular mixture of substances can damage an organism. Toxicity can refer to the effect on a whole organism, such as an animal, bacterium, or plant, as well as the effect on a substructure of the organism, such as a cell ( cytotoxicity) or an organ such as the liver (hepatotoxicity). By extension, the word may be metaphorically used to describe toxic effects on larger and more complex groups, such as the family unit or society at large. Sometimes the word is more or less synonymous with poisoning in everyday usage. A central concept of toxicology is that the effects of a toxicant are dose-dependent; even water can lead to water intoxication when taken in too high a dose, whereas for even a very toxic substance such as snake venom there is a dose below which there is no detectable toxic effect. Toxicity is species-specific, making cross-species analysis problematic. Newer paradigms and metrics are evolving to bypa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Kainate Receptor Agonists
Kainic acid, or kainate, is an acid that naturally occurs in some seaweed. Kainic acid is a potent neuroexcitatory amino acid agonist that acts by activating receptors for glutamate, the principal excitatory neurotransmitter in the central nervous system. Glutamate is produced by the cell's metabolic processes and there are four major classifications of glutamate receptors: NMDA receptors, AMPA receptors, kainate receptors, and the metabotropic glutamate receptors. Kainic acid is an agonist for kainate receptors, a type of ionotropic glutamate receptor. Kainate receptors likely control a sodium channel that produces excitatory postsynaptic potentials (EPSPs) when glutamate binds. Kainic acid is commonly injected into laboratory animal models to study the effects of experimental ablation. Kainic acid is a direct agonist of the glutamic kainate receptors and large doses of concentrated solutions produce immediate neuronal death by overstimulating neurons to death. Such damage and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
AMPA Receptor Agonists
α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid, better known as AMPA, is a compound that is a specific agonist for the AMPA receptor, where it mimics the effects of the neurotransmitter glutamate. There are several types of glutamatergic ion channels in the central nervous system including AMPA, kainic acid and ''N''-methyl-D-aspartic acid (NMDA) channels. In the synapse, these receptors serve very different purposes. AMPA can be used experimentally to distinguish the activity of one receptor from the other in order to understand their differing functions. AMPA generates fast excitatory postsynaptic potentials (EPSP). AMPA activates AMPA receptors that are non-selective cationic channels allowing the passage of Na+ and K+ and therefore have an equilibrium potential near 0 mV. AMPA was first synthesized, along with several other ibotenic acid Ibotenic acid or (''S'')-2-amino-2-(3-hydroxyisoxazol-5-yl)acetic acid, also referred to as ibotenate, is a chemical compo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Imides
In organic chemistry, an imide is a functional group consisting of two acyl groups bound to nitrogen. The compounds are structurally related to acid anhydrides, although imides are more resistant to hydrolysis. In terms of commercial applications, imides are best known as components of high-strength polymers, called polyimides. Inorganic imides are also known as solid state or gaseous compounds, and the imido group (=NH) can also act as a ligand. Nomenclature Most imides are cyclic compounds derived from dicarboxylic acids, and their names reflect the parent acid. Examples are succinimide, derived from succinic acid, and phthalimide, derived from phthalic acid. For imides derived from amines (as opposed to ammonia), the ''N''-substituent is indicated by a prefix. For example, N-ethylsuccinimide is derived from succinic acid and ethylamine. Isoimides are isomeric with normal imides and have the formula RC(O)OC(NR′)R″. They are often intermediates that convert to the more symmet ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lactams
A lactam is a cyclic amide, formally derived from an amino alkanoic acid. The term is a portmanteau of the words ''lactone'' + ''amide''. Nomenclature Greek prefixes in alphabetical order indicate ring size: * α-Lactam (3-atom rings) * β-Lactam (4-atom rings) * γ-Lactam (5-atom rings) * δ-Lactam (6-atom rings) * ε-Lactam (7-atom rings) This ring-size nomenclature stems from the fact that a hydrolyzed α-Lactam leads to an α-amino acid and a β-Lactam to a β-amino acid, ''etc''. Synthesis General synthetic methods exist for the organic synthesis of lactams. Beckmann rearrangement Lactams form by the acid-catalyzed rearrangement of oximes in the Beckmann rearrangement. Schmidt reaction Lactams form from cyclic ketones and hydrazoic acid in the Schmidt reaction. Cyclization of amino acids Lactams can be formed from cyclisation of amino acids via the coupling between an amine and a carboxylic acid within the same molecule. Lactamization is most efficient in this wa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
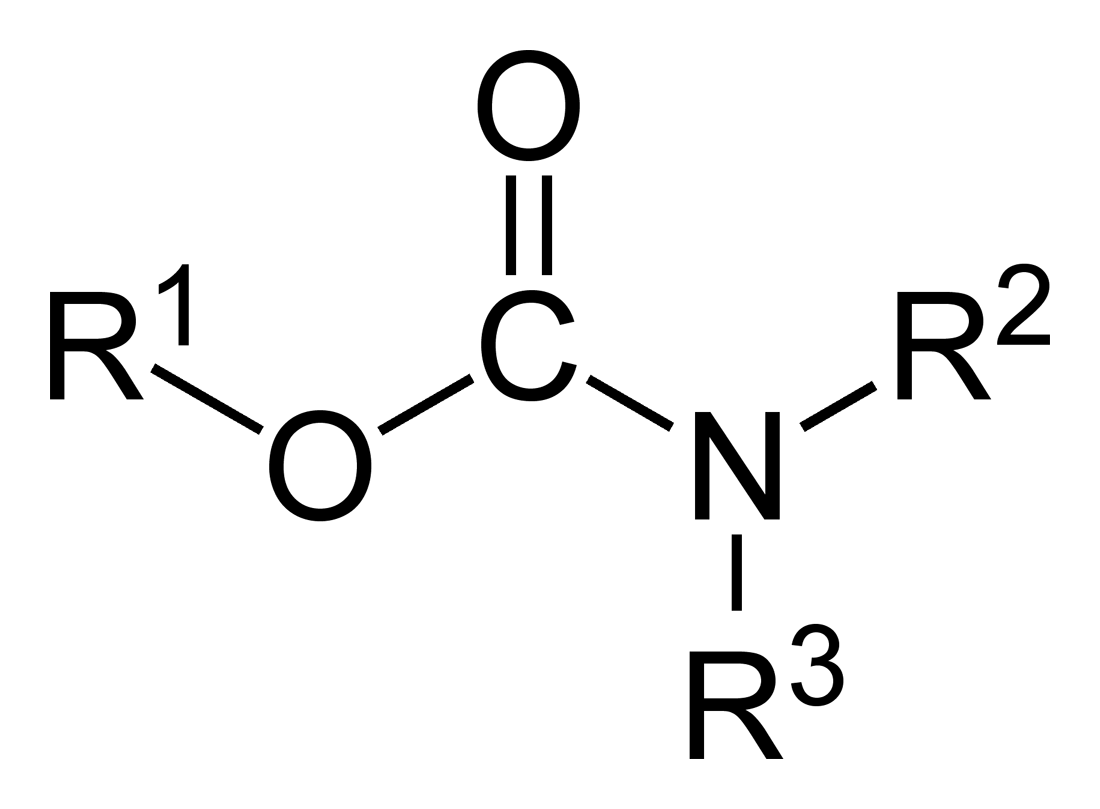
Carbamates
In organic chemistry, a carbamate is a category of organic compounds with the general formula and structure , which are formally derived from carbamic acid (). The term includes organic compounds (e.g., the ester ethyl carbamate), formally obtained by replacing one or more of the hydrogen atoms by other organic functional groups; as well as salts with the carbamate anion (e.g. ammonium carbamate). Polymers whose units are joined by carbamate groups are an important family of plastics, the polyurethanes. Properties While carbamic acids are unstable, many carbamate esters or ionic) are stable and well known. Equilibrium with carbonate and bicarbonate In water solutions, the carbamate anion slowly equilibrates with the ammonium cation and the carbonate or bicarbonate anions: : : Calcium carbamate is soluble in water, whereas calcium carbonate is not. Adding a calcium salt to an ammonium carbamate/carbonate solution will precipitate some calcium carbonate immediately, a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ureas
220 px, B vitamin, is a urea. In chemistry, ureas are a class of organic compounds with the formula (R2N)2CO where R = H, alkyl, aryl, etc. Thus, in addition to describing the specific chemical compound urea ((H2N)2CO), urea is the name of a functional group that is found in many compounds and materials of both practical and theoretical interest. Generally ureas are colorless crystalline solids, which, owing to the presence of fewer hydrogen bonds, exhibit melting points lower than that of urea itself. Synthesis Ureas can be prepared many methods, but rarely by direct carbonation, which is the route to urea itself. Instead, methods can be classified according those that assemble the urea functionality and those that start with preformed urea. Assembly of N-substituted urea functionality Phosgenation entails the reaction of amines with phosgene, proceeding via the isocyanate (or carbamoyl chloride) as an intermediate: :COCl2 + R2NH → R2NC(O)Cl + HCl :COCl2 + RNH2 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |