|
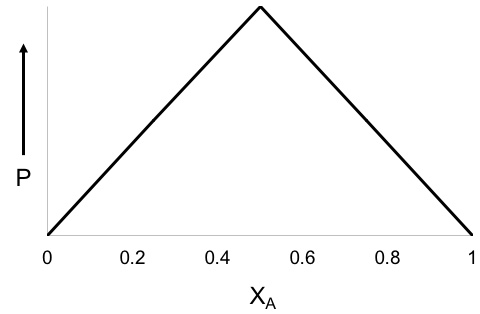
Job Plot
Within chemistry, a Job plot, otherwise known as the method of continuous variation or Job's method, is a method used in analytical chemistry to determine the stoichiometry of a binding event. The method is named after Paul Job and is also used in instrumental analysis and advanced chemical equilibrium texts and research articles. Job first published his method in 1928, while studying the associations of ions in solution. By plotting the UV absorbance of a solution of against the mole fraction of , he produced a graph which provided information about the equilibrium complexes present in solution. Theory In solutions where two species are present (i.e. species A and species B), one species (A) may bind to the other species (B). In some cases, more than one A will bind with a single B. One way to determine the amount of A binding to B is by using a Job plot. In this method, the sum of the molar concentrations of the two binding partners (e.g. a protein and ligand or a metal a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemistry
Chemistry is the science, scientific study of the properties and behavior of matter. It is a natural science that covers the Chemical element, elements that make up matter to the chemical compound, compounds made of atoms, molecules and ions: their composition, structure, properties, behavior and the changes they undergo during a Chemical reaction, reaction with other Chemical substance, substances. Chemistry also addresses the nature of chemical bonds in chemical compounds. In the scope of its subject, chemistry occupies an intermediate position between physics and biology. It is sometimes called the central science because it provides a foundation for understanding both Basic research, basic and Applied science, applied scientific disciplines at a fundamental level. For example, chemistry explains aspects of plant growth (botany), the formation of igneous rocks (geology), how atmospheric ozone is formed and how environmental pollutants are degraded (ecology), the properties ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Analytical Chemistry
Analytical chemistry studies and uses instruments and methods to separate, identify, and quantify matter. In practice, separation, identification or quantification may constitute the entire analysis or be combined with another method. Separation isolates analytes. Qualitative analysis identifies analytes, while quantitative analysis determines the numerical amount or concentration. Analytical chemistry consists of classical, wet chemical methods and modern, instrumental methods. Classical qualitative methods use separations such as precipitation, extraction, and distillation. Identification may be based on differences in color, odor, melting point, boiling point, solubility, radioactivity or reactivity. Classical quantitative analysis uses mass or volume changes to quantify amount. Instrumental methods may be used to separate samples using chromatography, electrophoresis or field flow fractionation. Then qualitative and quantitative analysis can be performed, often with t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Stoichiometry
Stoichiometry refers to the relationship between the quantities of reactants and products before, during, and following chemical reactions. Stoichiometry is founded on the law of conservation of mass where the total mass of the reactants equals the total mass of the products, leading to the insight that the relations among quantities of reactants and products typically form a ratio of positive integers. This means that if the amounts of the separate reactants are known, then the amount of the product can be calculated. Conversely, if one reactant has a known quantity and the quantity of the products can be empirically determined, then the amount of the other reactants can also be calculated. This is illustrated in the image here, where the balanced equation is: : Here, one molecule of methane reacts with two molecules of oxygen gas to yield one molecule of carbon dioxide and two molecules of water. This particular chemical equation is an example of complete combustion. St ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Instrumental Analysis
Instrumental analysis is a field of analytical chemistry that investigates analytes using scientific instruments. Spectroscopy Spectroscopy measures the interaction of the molecules with electromagnetic radiation. Spectroscopy consists of many different applications such as atomic absorption spectroscopy, atomic emission spectroscopy, ultraviolet-visible spectroscopy, X-ray fluorescence spectroscopy, infrared spectroscopy, Raman spectroscopy, nuclear magnetic resonance spectroscopy, photoemission spectroscopy, Mössbauer spectroscopy, and circular dichroism spectroscopy. Nuclear spectroscopy Methods of nuclear spectroscopy use properties of a nucleus to probe a material's properties, especially the material's local structure. Common methods include nuclear magnetic resonance spectroscopy (NMR), Mössbauer spectroscopy (MBS), and perturbed angular correlation (PAC). Mass spectrometry Mass spectrometry measures mass-to-charge ratio of molecules using electric and magnetic f ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Equilibrium
In a chemical reaction, chemical equilibrium is the state in which both the reactants and products are present in concentrations which have no further tendency to change with time, so that there is no observable change in the properties of the system. This state results when the forward reaction proceeds at the same rate as the reverse reaction. The reaction rates of the forward and backward reactions are generally not zero, but they are equal. Thus, there are no net changes in the concentrations of the reactants and products. Such a state is known as dynamic equilibrium. Historical introduction The concept of chemical equilibrium was developed in 1803, after Berthollet found that some chemical reactions are reversible. For any reaction mixture to exist at equilibrium, the rates of the forward and backward (reverse) reactions must be equal. In the following chemical equation, arrows point both ways to indicate equilibrium. A and B are reactant chemical species, S and T are p ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ultraviolet–visible Spectroscopy
UV spectroscopy or UV–visible spectrophotometry (UV–Vis or UV/Vis) refers to absorption spectroscopy or reflectance spectroscopy in part of the ultraviolet and the full, adjacent visible regions of the electromagnetic spectrum. Being relatively inexpensive and easily implemented, this methodology is widely used in diverse applied and fundamental applications. The only requirement is that the sample absorb in the UV-Vis region, i.e. be a chromophore. Absorption spectroscopy is complementary to fluorescence spectroscopy. Parameters of interest, besides the wavelength of measurement, are absorbance (A) or transmittance (%T) or reflectance (%R), and its change with time. Optical transitions Most molecules and ions absorb energy in the ultraviolet or visible range, i.e., they are chromophores. The absorbed photon excites an electron in the chromophore to higher energy molecular orbitals, giving rise to an excited state. For organic chromophores, four possible types of transitions ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mole Fraction
In chemistry, the mole fraction or molar fraction (''xi'' or ) is defined as unit of the amount of a constituent (expressed in moles), ''ni'', divided by the total amount of all constituents in a mixture (also expressed in moles), ''n''tot. This expression is given below: :x_i = \frac The sum of all the mole fractions is equal to 1: :\sum_^ n_i = n_\mathrm ; \ \sum_^ x_i = 1. The same concept expressed with a denominator of 100 is the mole percent, molar percentage or molar proportion (mol%). The mole fraction is also called the amount fraction. It is identical to the number fraction, which is defined as the number of molecules of a constituent ''Ni'' divided by the total number of all molecules ''N''tot. The mole fraction is sometimes denoted by the lowercase Greek letter (chi) instead of a Roman ''x''. For mixtures of gases, IUPAC recommends the letter ''y''. The National Institute of Standards and Technology of the United States prefers the term amount-of-substance fraction o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Job Plot 1
Work or labor (or labour in British English) is intentional activity people perform to support the needs and wants of themselves, others, or a wider community. In the context of economics, work can be viewed as the human activity that contributes (along with other factors of production) towards the goods and services within an economy. Work is fundamental to all societies, but can vary widely within and between them, from gathering in natural resources by hand, to operating complex technologies that substitute for physical or even mental effort by many human beings. All but the simplest tasks also require specific skills, equipment or tools, and other resources (such as material for manufacturing goods). Cultures and individuals across history have expressed a wide range of attitudes towards work. Outside of any specific process or industry, humanity has developed a variety of institutions for situating work in society. Besides objective differences, one culture may or ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |