|
Isotope Fractionation
Isotope fractionation describes fractionation processes that affect the relative abundance of isotopes, phenomena which are taken advantage of in isotope geochemistry and other fields. Normally, the focus is on stable isotopes of the same element. Isotopic fractionation can be measured by isotope analysis, using isotope-ratio mass spectrometry or cavity ring-down spectroscopy to measure ratios of isotopes, an important tool to understand geochemical and biological systems. For example, biochemical processes cause changes in ratios of stable carbon isotopes incorporated into biomass. Definition Stable isotopes partitioning between two substances ''A'' and ''B'' can be expressed by the use of the isotopic fractionation factor (alpha): where ''R'' is the ratio of the heavy to light isotope (e.g., 2H/1H or 18O/16O). Values for alpha tend to be very close to 1. McGlynn, Shawn E.; "Biological Isotope Fractionation and Earth History: From Enzymes to Cells to Ecosystems" pp 59-79 in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
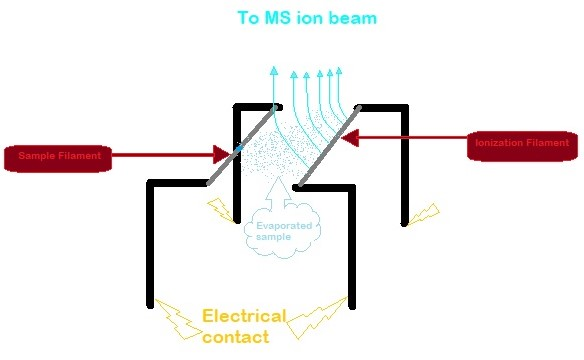
Thermal Ionization Mass Spectrometer
Thermal ionization mass spectrometry (TIMS) is also known as surface ionization and is a highly sensitive isotope mass spectrometry characterization technique. The isotopic ratios of radionuclides are used to get an accurate measurement for the elemental analysis of a sample. Singly charged ions of the sample are formed by the thermal ionization effect. A chemically purified liquid sample is placed on a metal filament which is then heated to evaporate the solvent. The removal of an electron from the purified sample is consequently achieved by heating the filament enough to release an electron, which then ionizes the atoms of the sample. TIMS utilizes a magnetic sector mass analyzer to separate the ions based on their mass to charge ratio. The ions gain velocity by an electrical potential gradient and are focused into a beam by electrostatic lenses. The ion beam then passes through the magnetic field of the electromagnet where it is partitioned into separate ion beams based on the io ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Kinetic Fractionation
Kinetic fractionation is an isotopic fractionation process that separates stable isotopes from each other by their mass during unidirectional processes. Biological processes are generally unidirectional and are very good examples of "kinetic" isotope reactions. All organisms preferentially use lighter isotopic species, because "energy costs" are lower, resulting in a significant fractionation between the substrate (heavier) and the biologically mediated product (lighter). As an example, photosynthesis preferentially takes up the light isotope of carbon 12C during assimilation of an atmospheric CO2 molecule. This kinetic isotope fractionation explains why plant material (and thus fossil fuels, which are derived from plants) is typically depleted in 13C by 25 per mil (2.5 per cent) relative to most inorganic carbon on Earth. A naturally occurring example of non-biological kinetic fractionation occurs during the evaporation of seawater to form clouds under conditions in which some pa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Springer Verlag
Springer Science+Business Media, commonly known as Springer, is a German multinational publishing company of books, e-books and peer-reviewed journals in science, humanities, technical and medical (STM) publishing. Originally founded in 1842 in Berlin, it expanded internationally in the 1960s, and through mergers in the 1990s and a sale to venture capitalists it fused with Wolters Kluwer and eventually became part of Springer Nature in 2015. Springer has major offices in Berlin, Heidelberg, Dordrecht, and New York City. History Julius Springer founded Springer-Verlag in Berlin in 1842 and his son Ferdinand Springer grew it from a small firm of 4 employees into Germany's then second largest academic publisher with 65 staff in 1872.Chronology ". Springer Science+Business Media. In 1964, Springer expanded its business internationally, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
John Wiley & Sons
John Wiley & Sons, Inc., commonly known as Wiley (), is an American multinational publishing company founded in 1807 that focuses on academic publishing and instructional materials. The company produces books, journals, and encyclopedias, in print and electronically, as well as online products and services, training materials, and educational materials for undergraduate, graduate, and continuing education students. History The company was established in 1807 when Charles Wiley opened a print shop in Manhattan. The company was the publisher of 19th century American literary figures like James Fenimore Cooper, Washington Irving, Herman Melville, and Edgar Allan Poe, as well as of legal, religious, and other non-fiction titles. The firm took its current name in 1865. Wiley later shifted its focus to scientific, technical, and engineering subject areas, abandoning its literary interests. Wiley's son John (born in Flatbush, New York, October 4, 1808; died in East Orange, N ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isotope Separation
Isotope separation is the process of concentrating specific isotopes of a chemical element by removing other isotopes. The use of the nuclides produced is varied. The largest variety is used in research (e.g. in chemistry where atoms of "marker" nuclide are used to figure out reaction mechanisms). By tonnage, separating natural uranium into enriched uranium and depleted uranium is the largest application. In the following text, mainly the uranium enrichment is considered. This process is crucial in the manufacture of uranium fuel for nuclear power plants, and is also required for the creation of uranium-based nuclear weapons. Plutonium-based weapons use plutonium produced in a nuclear reactor, which must be operated in such a way as to produce plutonium already of suitable isotopic mix or ''grade''. While different chemical elements can be purified through chemical processes, isotopes of the same element have nearly identical chemical properties, which makes this type of separation ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Water Vapor
(99.9839 °C) , - , Boiling point , , - , specific gas constant , 461.5 J/( kg·K) , - , Heat of vaporization , 2.27 MJ/kg , - , Heat capacity , 1.864 kJ/(kg·K) Water vapor, water vapour or aqueous vapor is the gaseous phase of water. It is one state of water within the hydrosphere. Water vapor can be produced from the evaporation or boiling of liquid water or from the sublimation of ice. Water vapor is transparent, like most constituents of the atmosphere. Under typical atmospheric conditions, water vapor is continuously generated by evaporation and removed by condensation. It is less dense than most of the other constituents of air and triggers convection currents that can lead to clouds. Being a component of Earth's hydrosphere and hydrologic cycle, it is particularly abundant in Earth's atmosphere, where it acts as a greenhouse gas and warming feedback, contributing more to total greenhouse effect than non-condensable gases such as carbon ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phase Transition
In chemistry, thermodynamics, and other related fields, a phase transition (or phase change) is the physical process of transition between one state of a medium and another. Commonly the term is used to refer to changes among the basic states of matter: solid, liquid, and gas, and in rare cases, plasma. A phase of a thermodynamic system and the states of matter have uniform physical properties. During a phase transition of a given medium, certain properties of the medium change as a result of the change of external conditions, such as temperature or pressure. This can be a discontinuous change; for example, a liquid may become gas upon heating to its boiling point, resulting in an abrupt change in volume. The identification of the external conditions at which a transformation occurs defines the phase transition point. Types of phase transition At the phase transition point for a substance, for instance the boiling point, the two phases involved - liquid and vapor, have ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Transient Kinetic Isotope Fractionation
Transient kinetic isotope effects (or fractionation) occur when the reaction leading to isotope fractionation does not follow pure first-order kinetics and therefore isotopic effects cannot be described with the classical equilibrium fractionation equations or with steady-state kinetic fractionation equations (also known as the Rayleigh equation). In these instances, the general equations for biochemical isotope kinetics (GEBIK) and the general equations for biochemical isotope fractionation (GEBIF) can be used. The GEBIK and GEBIF equations are the most generalized approach to describe isotopic effects in any chemical, catalytic reaction and biochemical reactions because they can describe isotopic effects in equilibrium reactions, kinetic chemical reactions and kinetic biochemical reactions. In the latter two cases, they can describe both stationary and non-stationary fractionation (i.e., variable and inverse fractionation). In general, isotopic effects depend on the number of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mass-independent Fractionation
Mass-independent isotope fractionation or Non-mass-dependent fractionation (NMD), refers to any chemical or physical process that acts to separate isotopes, where the amount of separation does not scale in proportion with the difference in the masses of the isotopes. Most isotopic fractionations (including typical kinetic fractionations and equilibrium fractionations) are caused by the effects of the mass of an isotope on atomic or molecular velocities, diffusivities or bond strengths. Mass-independent fractionation processes are less common, occurring mainly in photochemical and spin-forbidden reactions. Observation of mass-independently fractionated materials can therefore be used to trace these types of reactions in nature and in laboratory experiments. Mass-independent fractionation in nature The most notable examples of mass-independent fractionation in nature are found in the isotopes of oxygen and sulfur. The first example was discovered by Robert N. Clayton, Toshiko ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Equilibrium Fractionation
Equilibrium isotope fractionation is the partial separation of isotopes between two or more substances in chemical equilibrium. Equilibrium fractionation is strongest at low temperatures, and (along with kinetic isotope effects) forms the basis of the most widely used isotopic paleothermometers (or climate proxies): D/H and 18O/16O records from ice cores, and 18O/16O records from calcium carbonate. It is thus important for the construction of geologic temperature records. Isotopic fractionations attributed to equilibrium processes have been observed in many elements, from hydrogen ( D/H) to uranium ( 238U/235U). In general, the light elements (especially hydrogen, boron, carbon, nitrogen, oxygen and sulfur) are most susceptible to fractionation, and their isotopes tend to be separated to a greater degree than heavier elements. Definition Most equilibrium fractionations are thought to result from the reduction in vibrational energy (especially zero-point energy) when a more ma ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fractionation
Fractionation is a separation process in which a certain quantity of a mixture (of gases, solids, liquids, enzymes, or isotopes, or a suspension) is divided during a phase transition, into a number of smaller quantities (fractions) in which the composition varies according to a gradient. Fractions are collected based on differences in a specific property of the individual components. A common trait in fractionations is the need to find an optimum between the amount of fractions collected and the desired purity in each fraction. Fractionation makes it possible to isolate more than two components in a mixture in a single run. This property sets it apart from other separation techniques. Fractionation is widely employed in many branches of science and technology. Mixtures of liquids and gases are separated by fractional distillation by difference in boiling point. Fractionation of components also takes place in column chromatography by a difference in affinity between stationary ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isotopes Of Carbon
Carbon (6C) has 15 known isotopes, from to , of which and are stable. The longest-lived radioisotope is , with a half-life of years. This is also the only carbon radioisotope found in nature—trace quantities are formed cosmogenically by the reaction + → + . The most stable artificial radioisotope is , which has a half-life of . All other radioisotopes have half-lives under 20 seconds, most less than 200 milliseconds. The least stable isotope is , with a half-life of . List of isotopes , - , , style="text-align:right" , 6 , style="text-align:right" , 2 , , [] , proton emission, 2p , Subsequently decays by double proton emission to for a net reaction of → + 4 , 0+ , , , - , rowspan=3, , rowspan=3 style="text-align:right" , 6 , rowspan=3 style="text-align:right" , 3 , rowspan=3, , rowspan=3, , β+ () , , rowspan=3, 3/2− , rowspan=3, , rowspan=3, , - , β+α () , Immediately decays by proton emission to for a net reaction of → ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |


_(cropped).jpg)

