|
IRE-BP
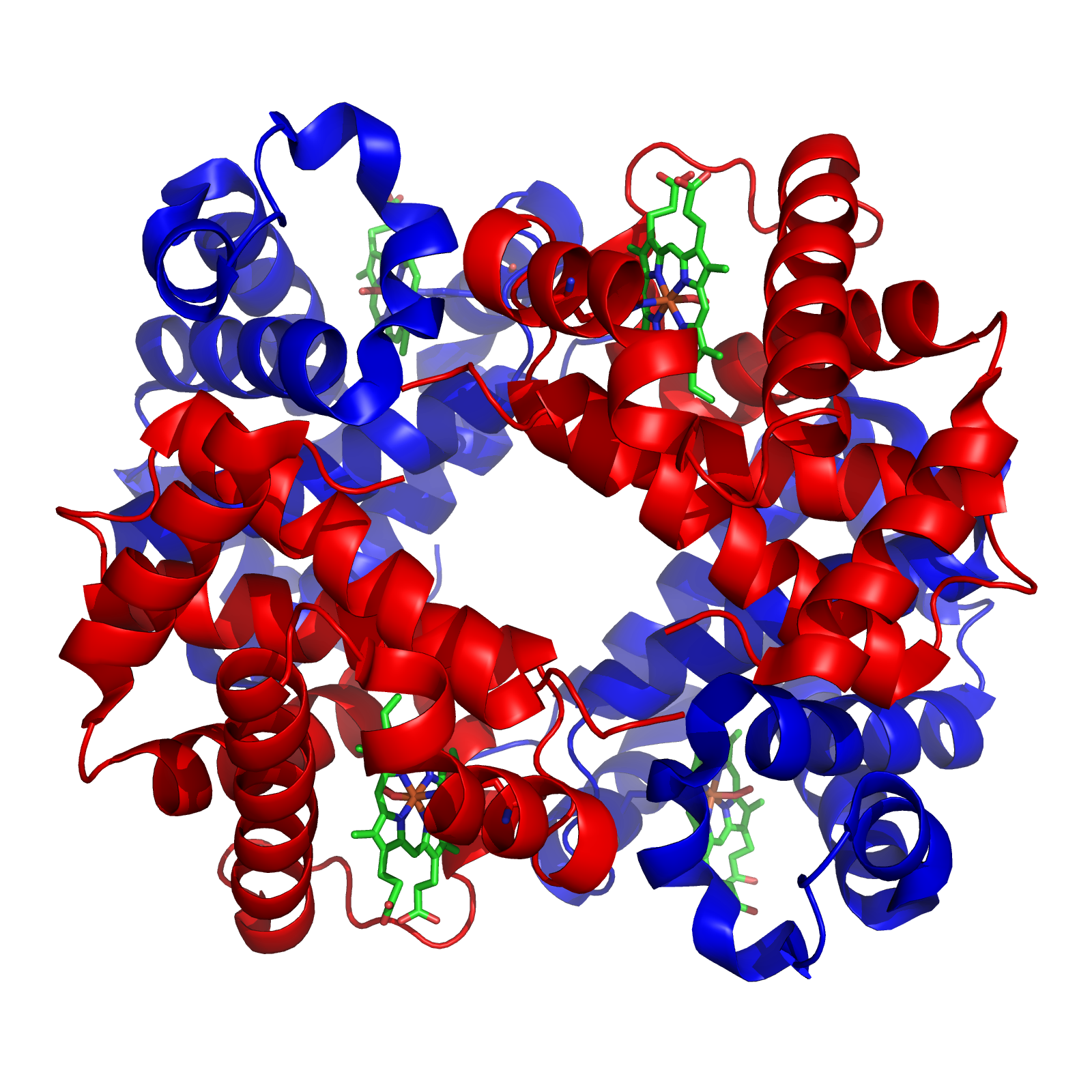
The iron-responsive element-binding proteins, also known as IRE-BP, IRBP, IRP and IFR , bind to iron-responsive elements (IREs) in the regulation of human iron metabolism. Function ACO1, or IRP1, is a bifunctional protein that functions as an iron-responsive element (IRE)-binding protein involved in the control of iron metabolism by binding mRNA to repress translation or degradation. It functions also as the cytoplasmic isoform of aconitase. Aconitases are iron-sulfur proteins that require a 4Fe-4S cluster for their enzymatic activity, in which they catalyze conversion of citrate to isocitrate. This structure was based on x-ray crystal diffraction. The resolution was 2.80 Å. This protein was harvested from the species ''Oryctolagus cuniculus'', more commonly known as a rabbit. This protein has a couple of conformational changes associated with it to explain the alternative functions as either mRNA regulator or as an enzyme. This information was obtained from the RCSB protein da ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aconitase
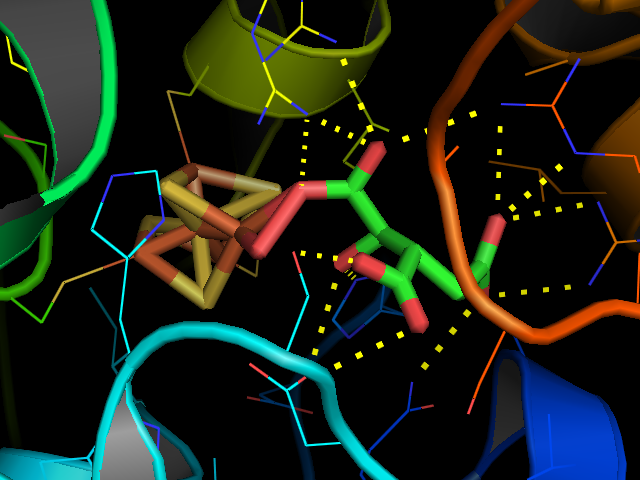
Aconitase (aconitate hydratase; ) is an enzyme that catalyses the stereo-specific isomerization of citrate to isocitrate via ''cis''- aconitate in the tricarboxylic acid cycle, a non-redox-active process. Image:Citrate wpmp.png, Image:Cis-Aconitate wpmp.png, Image:isocitric acid.svg, Structure Aconitase, displayed in the structures in the right margin of this page, has two slightly different structures, depending on whether it is activated or inactivated. In the inactive form, its structure is divided into four domains. Counting from the N-terminus, only the first three of these domains are involved in close interactions with the Fe-4Scluster, but the active site consists of residues from all four domains, including the larger C-terminal domain. The Fe-S cluster and a anion also reside in the active site. When the enzyme is activated, it gains an additional iron atom, creating a Fe-4Scluster. However, the structure of the rest of the enzyme is nearly unchanged; the c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aconitase
Aconitase (aconitate hydratase; ) is an enzyme that catalyses the stereo-specific isomerization of citrate to isocitrate via ''cis''- aconitate in the tricarboxylic acid cycle, a non-redox-active process. Image:Citrate wpmp.png, Image:Cis-Aconitate wpmp.png, Image:isocitric acid.svg, Structure Aconitase, displayed in the structures in the right margin of this page, has two slightly different structures, depending on whether it is activated or inactivated. In the inactive form, its structure is divided into four domains. Counting from the N-terminus, only the first three of these domains are involved in close interactions with the Fe-4Scluster, but the active site consists of residues from all four domains, including the larger C-terminal domain. The Fe-S cluster and a anion also reside in the active site. When the enzyme is activated, it gains an additional iron atom, creating a Fe-4Scluster. However, the structure of the rest of the enzyme is nearly unchanged; the c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iron Responsive Element
In molecular biology, the iron response element or iron-responsive element (IRE) is a short conserved stem-loop which is bound by iron response proteins (IRPs, also named IRE-BP or IRBP). The IRE is found in UTRs (untranslated regions) of various mRNAs whose products are involved in iron metabolism. For example, the mRNA of ferritin (an iron storage protein) contains one IRE in its 5' UTR. When iron concentration is low, IRPs bind the IRE in the ferritin mRNA and cause reduced translation rates. In contrast, binding to multiple IREs in the 3' UTR of the transferrin receptor (involved in iron acquisition) leads to increased mRNA stability. Mechanism of action The two leading theories describe how iron probably interacts to impact posttranslational control of transcription. The classical theory suggests that IRPs, in the absence of iron, bind avidly to the mRNA IRE. When iron is present, it interacts with the protein to cause it to release the mRNA. For example, In high iron ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iron-responsive Element
In molecular biology, the iron response element or iron-responsive element (IRE) is a short conserved stem-loop which is bound by iron response proteins (IRPs, also named IRE-BP or IRBP). The IRE is found in UTRs (untranslated regions) of various mRNAs whose products are involved in iron metabolism. For example, the mRNA of ferritin (an iron storage protein) contains one IRE in its 5' UTR. When iron concentration is low, IRPs bind the IRE in the ferritin mRNA and cause reduced translation rates. In contrast, binding to multiple IREs in the 3' UTR of the transferrin receptor (involved in iron acquisition) leads to increased mRNA stability. Mechanism of action The two leading theories describe how iron probably interacts to impact posttranslational control of transcription. The classical theory suggests that IRPs, in the absence of iron, bind avidly to the mRNA IRE. When iron is present, it interacts with the protein to cause it to release the mRNA. For example, In high iro ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isocitrate
Isocitric acid is a structural isomer of citric acid. Since citric acid and isocitric acid are structural isomers, they share similar physical and chemical properties. Due to these similar properties, it is difficult to separate the isomers. Salts and esters of isocitric acid are known as isocitrates. The isocitrate anion is a substrate of the citric acid cycle. Isocitrate is formed from citrate with the help of the enzyme aconitase Aconitase (aconitate hydratase; ) is an enzyme that catalyses the stereo-specific isomerization of citrate to isocitrate via ''cis''- aconitate in the tricarboxylic acid cycle, a non-redox-active process. Image:Citrate wpmp.png, Image:Cis- ..., and is acted upon by isocitrate dehydrogenase. Isocitric acid is commonly used as a marker to detect the authenticity and quality of fruit products, most often citrus juices. In authentic orange juice, for example, the ratio of citric acid to D-isocitric acid is usually less than 130. An isocitr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oryctolagus Cuniculus
The European rabbit (''Oryctolagus cuniculus'') or coney is a species of rabbit native to the Iberian Peninsula (including Spain, Portugal, and southwestern France), western France, and the northern Atlas Mountains in northwest Africa. It has been widely introduced elsewhere, often with devastating effects on local biodiversity. Its decline in its native range due to myxomatosis, rabbit calicivirus, overhunting and habitat loss has caused the decline of the Iberian lynx (''Lynx pardinus'') and Spanish imperial eagle (''Aquila adalberti''). It is known as an invasive species because it has been introduced to countries on all continents with the exception of Antarctica, and has caused many problems within the environment and ecosystems; in particular, European rabbits in Australia have had a devastating impact, due in part to the lack of natural predators there. The European rabbit is well known for digging networks of burrows, called warrens, where it spends most of its time whe ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
FBXL5
F-box/LRR-repeat protein 5 is a protein that in humans is encoded by the ''FBXL5'' gene. This gene encodes a member of the F-box protein family which is characterized by an approximately 40 amino acid motif, the F-box. The F-box proteins constitute one of the four subunits of ubiquitin protein ligase complex called SCFs (SKP1-cullin-F-box), which function in phosphorylation-dependent ubiquitination. The F-box proteins are divided into 3 classes: Fbws containing WD-40 domains, Fbls containing leucine-rich repeats, and Fbxs containing either different protein-protein interaction modules or no recognizable motifs. The protein encoded by this gene belongs to the Fbls class and, in addition to an F-box, contains several tandem leucine-rich repeats. Alternative splicing of this gene generates 2 transcript variants. FBXL5 is an iron sensor. It promotes IRP2 ubiquitination and then its degradation. References Further reading * * * * * * * * * * * * * {{gene-4-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Blood
Blood is a body fluid in the circulatory system of humans and other vertebrates that delivers necessary substances such as nutrients and oxygen to the cells, and transports metabolic waste products away from those same cells. Blood in the circulatory system is also known as ''peripheral blood'', and the blood cells it carries, ''peripheral blood cells''. Blood is composed of blood cells suspended in blood plasma. Plasma, which constitutes 55% of blood fluid, is mostly water (92% by volume), and contains proteins, glucose, mineral ions, hormones, carbon dioxide (plasma being the main medium for excretory product transportation), and blood cells themselves. Albumin is the main protein in plasma, and it functions to regulate the colloidal osmotic pressure of blood. The blood cells are mainly red blood cells (also called RBCs or erythrocytes), white blood cells (also called WBCs or leukocytes) and platelets (also called thrombocytes). The most abundant cells in vertebrate blo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
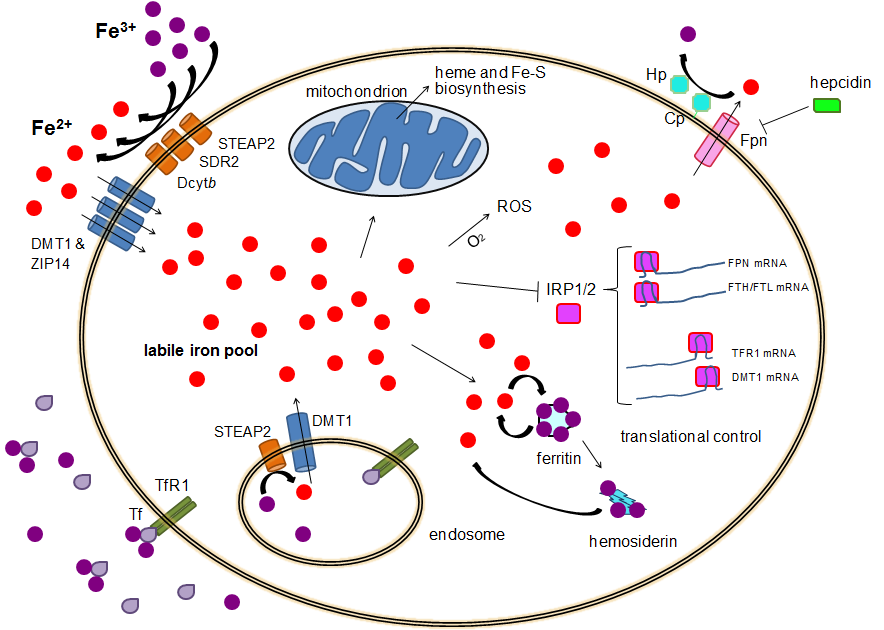
Human Iron Metabolism
Human iron metabolism is the set of chemical reactions that maintain human homeostasis of iron at the systemic and cellular level. Iron is both necessary to the body and potentially toxic. Controlling iron levels in the body is a critically important part of many aspects of human health and disease. Hematologists have been especially interested in systemic iron metabolism because iron is essential for red blood cells, where most of the human body's iron is contained. Understanding iron metabolism is also important for understanding diseases of iron overload, such as hereditary hemochromatosis, and iron deficiency, such as iron-deficiency anemia. Importance of iron regulation Iron is an essential bioelement for most forms of life, from bacteria to mammals. Its importance lies in its ability to mediate electron transfer. In the ferrous state (Fe2+), iron acts as an electron donor, while in the ferric state (Fe3+) it acts as an acceptor. Thus, iron plays a vital role in the catalys ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ferritin
Ferritin is a universal intracellular protein that stores iron and releases it in a controlled fashion. The protein is produced by almost all living organisms, including archaea, bacteria, algae, higher plants, and animals. It is the primary ''intracellular iron-storage protein'' in both prokaryotes and eukaryotes, keeping iron in a soluble and non-toxic form. In humans, it acts as a buffer against iron deficiency and iron overload. Ferritin is found in most tissues as a cytosolic protein, but small amounts are secreted into the serum where it functions as an iron carrier. Plasma ferritin is also an indirect marker of the total amount of iron stored in the body; hence, serum ferritin is used as a diagnostic test for iron-deficiency anemia. Aggregated ferritin transforms into a toxic form of iron called hemosiderin. Ferritin is a globular protein complex consisting of 24 protein subunits forming a hollow nanocage with multiple metal–protein interactions. Ferritin that is n ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Translation (genetics)
In molecular biology and genetics, translation is the process in which ribosomes in the cytoplasm or endoplasmic reticulum synthesize proteins after the process of transcription of DNA to RNA in the cell's nucleus. The entire process is called gene expression. In translation, messenger RNA (mRNA) is decoded in a ribosome, outside the nucleus, to produce a specific amino acid chain, or polypeptide. The polypeptide later folds into an active protein and performs its functions in the cell. The ribosome facilitates decoding by inducing the binding of complementary tRNA anticodon sequences to mRNA codons. The tRNAs carry specific amino acids that are chained together into a polypeptide as the mRNA passes through and is "read" by the ribosome. Translation proceeds in three phases: # Initiation: The ribosome assembles around the target mRNA. The first tRNA is attached at the start codon. # Elongation: The last tRNA validated by the small ribosomal subunit (''accommodation ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
MRNA
In molecular biology, messenger ribonucleic acid (mRNA) is a single-stranded molecule of RNA that corresponds to the genetic sequence of a gene, and is read by a ribosome in the process of Protein biosynthesis, synthesizing a protein. mRNA is created during the process of Transcription (biology), transcription, where an enzyme (RNA polymerase) converts the gene into primary transcript mRNA (also known as pre-mRNA). This pre-mRNA usually still contains introns, regions that will not go on to code for the final amino acid sequence. These are removed in the process of RNA splicing, leaving only exons, regions that will encode the protein. This exon sequence constitutes mature mRNA. Mature mRNA is then read by the ribosome, and, utilising amino acids carried by transfer RNA (tRNA), the ribosome creates the protein. This process is known as Translation (biology), translation. All of these processes form part of the central dogma of molecular biology, which describes the flow of genet ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
_(Sylvilagus_palustris).jpg)