|
Iron Overdose
Iron poisoning typically occurs from ingestion of excess iron that results in acute toxicity. Mild symptoms which occur within hours include vomiting, diarrhea, abdominal pain, and drowsiness. In more severe cases, symptoms can include tachypnea, low blood pressure, seizures, or coma. If left untreated, iron poisoning can lead to multi-organ failure resulting in permanent organ damage or death. Iron is available over the counter as a single entity supplement in an iron salt form or in combination with vitamin supplements and is commonly used in the treatment of anemias. Overdoses on iron can be categorized as unintentional ingestion which is predominantly associated with children or intentional ingestion involving suicide attempts in adults. Unintentional ingestion of iron containing drug products are a major cause of mortality in children under the age of 6 years old in the United States. As a response, in 1997 the US Food and Drug Administration (FDA) implemented a regulation req ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acute Toxicity
Acute toxicity describes the adverse effects of a substance that result either from a single exposure or from multiple exposures in a short period of time (usually less than 24 hours). To be described as ''acute'' toxicity, the adverse effects should occur within 14 days of the administration of the substance. Acute toxicity is distinguished from chronic toxicity, which describes the adverse health effects from repeated exposures, often at lower levels, to a substance over a longer time period (months or years). It is widely considered unethical to use humans as test subjects for acute (or chronic) toxicity research. However, some information can be gained from investigating accidental human exposures (e.g., factory accidents). Otherwise, most acute toxicity data comes from animal testing or, more recently, ''in vitro'' testing methods and inference from data on similar substances. Measures of acute toxicity Regulatory values Limits for short-term exposure, such as ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
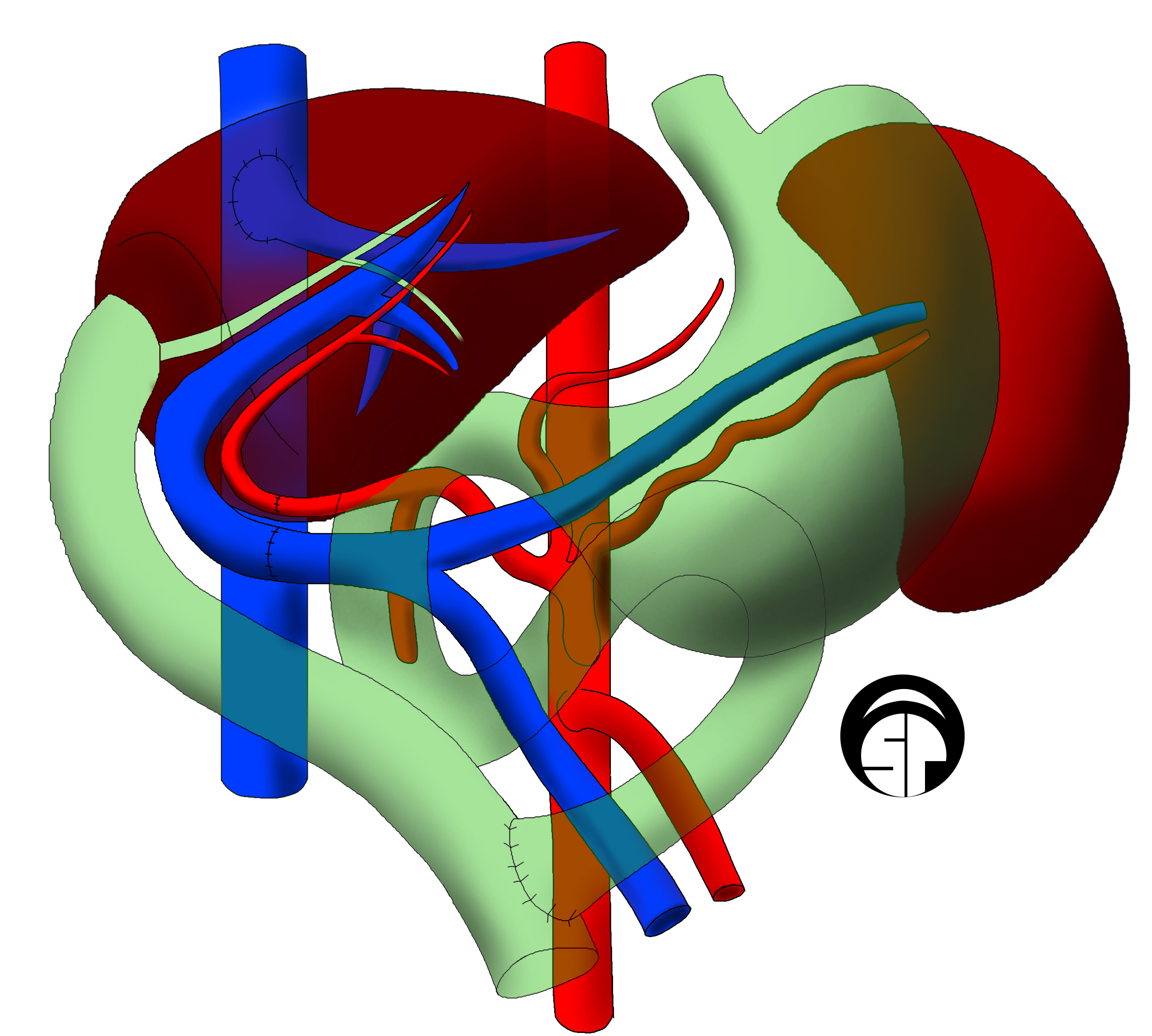
Liver Transplantation
Liver transplantation or hepatic transplantation is the replacement of a diseased liver with the healthy liver from another person ( allograft). Liver transplantation is a treatment option for end-stage liver disease and acute liver failure, although availability of donor organs is a major limitation. The most common technique is orthotopic transplantation, in which the native liver is removed and replaced by the donor organ in the same anatomic position as the original liver. The surgical procedure is complex, requiring careful harvest of the donor organ and meticulous implantation into the recipient. Liver transplantation is highly regulated, and only performed at designated transplant medical centers by highly trained transplant physicians and supporting medical team. The duration of the surgery ranges from 4 to 18 hours depending on outcome. Favorable outcomes require careful screening for eligible recipient, as well as a well-calibrated live or cadaveric donor match. Me ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ferritin
Ferritin is a universal intracellular protein that stores iron and releases it in a controlled fashion. The protein is produced by almost all living organisms, including archaea, bacteria, algae, higher plants, and animals. It is the primary ''intracellular iron-storage protein'' in both prokaryotes and eukaryotes, keeping iron in a soluble and non-toxic form. In humans, it acts as a buffer against iron deficiency and iron overload. Ferritin is found in most tissues as a cytosolic protein, but small amounts are secreted into the serum where it functions as an iron carrier. Plasma ferritin is also an indirect marker of the total amount of iron stored in the body; hence, serum ferritin is used as a diagnostic test for iron-deficiency anemia. Aggregated ferritin transforms into a toxic form of iron called hemosiderin. Ferritin is a globular protein complex consisting of 24 protein subunits forming a hollow nanocage with multiple metal–protein interactions. Ferritin th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Transferrin
Transferrins are glycoproteins found in vertebrates which bind to and consequently mediate the transport of iron (Fe) through blood plasma. They are produced in the liver and contain binding sites for two Fe3+ ions. Human transferrin is encoded by the ''TF'' gene and produced as a 76 kDa glycoprotein. Transferrin glycoproteins bind iron tightly, but reversibly. Although iron bound to transferrin is less than 0.1% (4 mg) of total body iron, it forms the most vital iron pool with the highest rate of turnover (25 mg/24 h). Transferrin has a molecular weight of around 80 kDa and contains two specific high-affinity Fe(III) binding sites. The affinity of transferrin for Fe(III) is extremely high ( association constant is 1020 M−1 at pH 7.4) but decreases progressively with decreasing pH below neutrality. Transferrins are not limited to only binding to iron but also to different metal ions. These glycoproteins are located in various bodily fluids of vertebrates. Some inv ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iron(III)
In chemistry, iron(III) refers to the element iron in its +3 oxidation state. In ionic compounds (salts), such an atom may occur as a separate cation (positive ion) denoted by Fe3+. The adjective ferric or the prefix ferri- is often used to specify such compounds — as in "ferric chloride" for iron(III) chloride, . The adjective " ferrous" is used instead for iron(II) salts, containing the cation Fe2+. The word ferric is derived from the Latin word ''ferrum'' for iron. Iron(III) metal centres also occur in coordination complexes, such as in the anion ferrioxalate, , where three bidentate oxalate ions surrounding the metal centre; or, in organometallic compounds, such as the ferrocenium cation , where two cyclopentadienyl anions are bound to the FeIII centre. Iron is almost always encountered in the oxidation states 0 (as in the metal), +2, or +3. Iron(III) is usually the most stable form in air, as illustrated by the pervasiveness of rust, an insoluble iron(III)-contain ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iron(II)
In chemistry, iron(II) refers to the element iron in its +2 oxidation state. In ionic compounds (salts), such an atom may occur as a separate cation (positive ion) denoted by Fe2+. The adjective ferrous or the prefix ferro- is often used to specify such compounds — as in "ferrous chloride" for iron(II) chloride, . The adjective "ferric" is used instead for iron(III) salts, containing the cation or Fe3+. The word ferrous is derived from the Latin word ''ferrum'' for iron. Iron(II) atoms may also occur as coordination complexes, such as the polymer iron(II) oxalate dihydrate, or ; and organometallic compounds, such as the neutral molecule ferrocene, or . Iron is almost always encountered in the oxidation states 0 (as in the metal), +2, or +3. Solid iron(II) salts are relatively stable in air, but in the presence of air and water they tend to oxidize to iron(III) salts that include hydroxide () or oxide () anions. Iron(II) and life All known forms of life require ir ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hemoglobin
Hemoglobin (haemoglobin BrE) (from the Greek word αἷμα, ''haîma'' 'blood' + Latin ''globus'' 'ball, sphere' + ''-in'') (), abbreviated Hb or Hgb, is the iron-containing oxygen-transport metalloprotein present in red blood cells (erythrocytes) of almost all vertebrates (the exception being the fish family Channichthyidae) as well as the tissues of some invertebrates. Hemoglobin in blood carries oxygen from the respiratory organs (''e.g.'' lungs or gills) to the rest of the body (''i.e.'' tissues). There it releases the oxygen to permit aerobic respiration to provide energy to power functions of an organism in the process called metabolism. A healthy individual human has 12to 20grams of hemoglobin in every 100mL of blood. In mammals, the chromoprotein makes up about 96% of the red blood cells' dry content (by weight), and around 35% of the total content (including water). Hemoglobin has an oxygen-binding capacity of 1.34mL O2 per gram, which increases the total blood oxygen ca ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Liver
The liver is a major organ only found in vertebrates which performs many essential biological functions such as detoxification of the organism, and the synthesis of proteins and biochemicals necessary for digestion and growth. In humans, it is located in the right upper quadrant of the abdomen, below the diaphragm. Its other roles in metabolism include the regulation of glycogen storage, decomposition of red blood cells, and the production of hormones. The liver is an accessory digestive organ that produces bile, an alkaline fluid containing cholesterol and bile acids, which helps the breakdown of fat. The gallbladder, a small pouch that sits just under the liver, stores bile produced by the liver which is later moved to the small intestine to complete digestion. The liver's highly specialized tissue, consisting mostly of hepatocytes, regulates a wide variety of high-volume biochemical reactions, including the synthesis and breakdown of small and complex molecu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Brain
The brain is an organ that serves as the center of the nervous system in all vertebrate and most invertebrate animals. It consists of nervous tissue and is typically located in the head ( cephalization), usually near organs for special senses such as vision, hearing and olfaction. Being the most specialized organ, it is responsible for receiving information from the sensory nervous system, processing those information (thought, cognition, and intelligence) and the coordination of motor control (muscle activity and endocrine system). While invertebrate brains arise from paired segmental ganglia (each of which is only responsible for the respective body segment) of the ventral nerve cord, vertebrate brains develop axially from the midline dorsal nerve cord as a vesicular enlargement at the rostral end of the neural tube, with centralized control over all body segments. All vertebrate brains can be embryonically divided into three parts: the forebrain (prosencep ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cardiogenic Shock
Cardiogenic shock (CS) is a medical emergency resulting from inadequate blood flow due to the dysfunction of the ventricles of the heart.Textbooks of Internal MedicinHarrison's Principles of Internal Medicine 16th Edition, The McGraw-Hill CompaniesCecil Textbook of Medicine by Lee Goldman, Dennis Ausiello, 22nd Edition (2003), W. B. Saunders CompanyThe Oxford Textbook of Medicine Edited by David A. Warrell, Timothy M. Cox and John D. Firth with Edward J. Benz, Fourth Edition (2003), Oxford University Press, Shock: An Overview PDF by Michael L. Cheatham, MD, Ernest F.J. Block, MD, Howard G. Smith, MD, John T. Promes, MD, Surgical Critical Care Service, Department of Surgical Education, |
Tachycardia
Tachycardia, also called tachyarrhythmia, is a heart rate that exceeds the normal resting rate. In general, a resting heart rate over 100 beats per minute is accepted as tachycardia in adults. Heart rates above the resting rate may be normal (such as with exercise) or abnormal (such as with electrical problems within the heart). Complications Tachycardia can lead to fainting. When the rate of blood flow becomes too rapid, or fast blood flow passes on damaged endothelium, it increases the friction within vessels resulting in turbulence and other disturbances. According to the Virchow's triad, this is one of the three conditions that can lead to thrombosis (i.e., blood clots within vessels). Causes Some causes of tachycardia include: * Adrenergic storm * Anaemia * Anxiety * Atrial fibrillation * Atrial flutter * Atrial tachycardia * Atrioventricular reentrant tachycardia * AV nodal reentrant tachycardia * Brugada syndrome * Circulatory shock and its various ca ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hypovolemic Shock
Hypovolemic shock is a form of shock caused by severe hypovolemia (insufficient blood volume or extracellular fluid in the body). It could be the result of severe dehydration through a variety of mechanisms or blood loss. Hypovolemic shock is a medical emergency; if left untreated, the insufficient blood flow can cause damage to organs, leading to multiple organ failure. In treating hypovolemic shock, it is important to determine the cause of the underlying hypovolemia, which may be the result of bleeding or other fluid losses. To minimize ischemic damage to tissues, treatment involves quickly replacing lost blood or fluids, with consideration of both rate and the type of fluids used. Tachycardia, a fast heart rate, is typically the first abnormal vital sign. When resulting from blood loss, trauma is the most common root cause, but severe blood loss can also happen in various body systems without clear traumatic injury. The body in hypovolemic shock prioritizes getting o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |


-oxide-sample.jpg)
_oxide.jpg)



