|
Health Effects Of Radon
Radon, a radioactive, colorless, odorless, tasteless noble gas, has been studied by a number of scientific and medical bodies for its effects on health. A naturally-occurring gas formed as a decay product of radium, radon is one of the densest substances that remains a gas under normal conditions, and is considered to be a health hazard due to its radioactivity. Its most stable isotope, radon-222 (), has a half-life of 3.8 days. Due to its high radioactivity, it has been less well studied by chemists, but a few compounds are known. Radon is formed as part of the normal radioactive decay chain of uranium into . Uranium has been present since the Earth was formed, and its most common isotope has a very long half-life (4.5 billion years), which is the time required for one-half of uranium to break down. Thus, uranium and radon will continue to occur for millions of years at about the same concentrations as they do now. Radon is responsible for the majority of public exposure to ion ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Radon
Radon is a chemical element with the symbol Rn and atomic number 86. It is a radioactive, colourless, odourless, tasteless noble gas. It occurs naturally in minute quantities as an intermediate step in the normal radioactive decay chains through which thorium and uranium slowly decay into various short-lived radioactive elements and lead. Radon itself is the immediate decay product of radium. Its most stable isotope, 222Rn, has a half-life of only 3.8 days, making it one of the rarest elements. Since thorium and uranium are two of the most common radioactive elements on Earth, while also having three isotopes with half-lives on the order of several billion years, radon will be present on Earth long into the future despite its short half-life. The decay of radon produces many other short-lived nuclides, known as "radon daughters", ending at stable isotopes of lead. [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
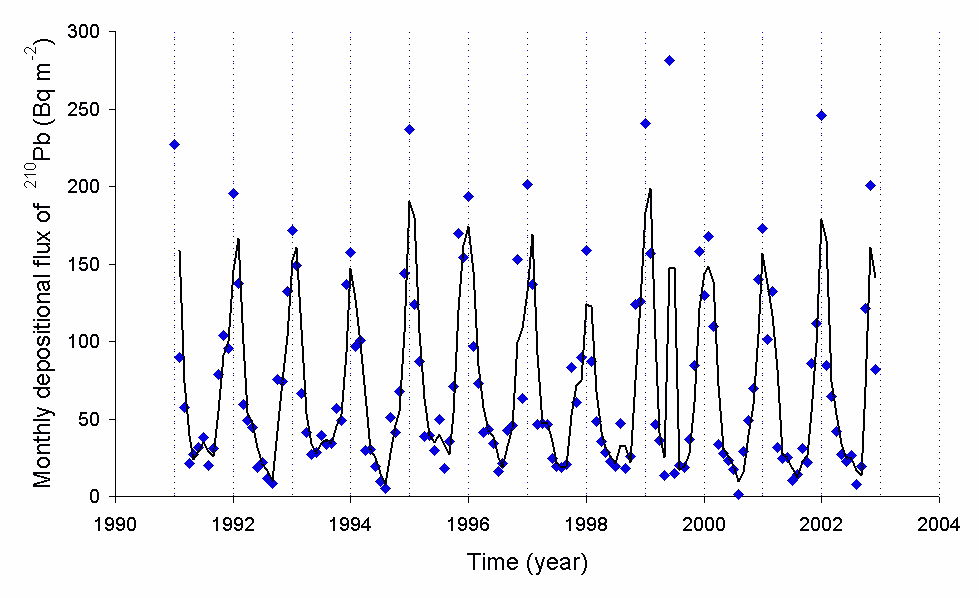
Lead210inairatjapan
Lead is a chemical element with the symbol Pb (from the Latin ) and atomic number 82. It is a heavy metal that is denser than most common materials. Lead is soft and malleable, and also has a relatively low melting point. When freshly cut, lead is a shiny gray with a hint of blue. It tarnishes to a dull gray color when exposed to air. Lead has the highest atomic number of any stable element and three of its isotopes are endpoints of major nuclear decay chains of heavier elements. Lead is toxic, even in small amounts, especially to children. Lead is a relatively unreactive post-transition metal. Its weak metallic character is illustrated by its amphoteric nature; lead and lead oxides react with acids and bases, and it tends to form covalent bonds. Compounds of lead are usually found in the +2 oxidation state rather than the +4 state common with lighter members of the carbon group. Exceptions are mostly limited to organolead compounds. Like the lighter members of the group, lea ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sand
Sand is a granular material composed of finely divided mineral particles. Sand has various compositions but is defined by its grain size. Sand grains are smaller than gravel and coarser than silt. Sand can also refer to a textural class of soil or soil type; i.e., a soil containing more than 85 percent sand-sized particles by mass. The composition of sand varies, depending on the local rock sources and conditions, but the most common constituent of sand in inland continental settings and non-tropical coastal settings is silica (silicon dioxide, or SiO2), usually in the form of quartz. Calcium carbonate is the second most common type of sand, for example, aragonite, which has mostly been created, over the past 500million years, by various forms of life, like coral and shellfish. For example, it is the primary form of sand apparent in areas where reefs have dominated the ecosystem for millions of years like the Caribbean. Somewhat more rarely, sand may be composed of calciu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Shale
Shale is a fine-grained, clastic sedimentary rock formed from mud that is a mix of flakes of clay minerals (hydrous aluminium phyllosilicates, e.g. kaolin, Al2 Si2 O5( OH)4) and tiny fragments (silt-sized particles) of other minerals, especially quartz and calcite.Blatt, Harvey and Robert J. Tracy (1996) ''Petrology: Igneous, Sedimentary and Metamorphic'', 2nd ed., Freeman, pp. 281–292 Shale is characterized by its tendency to split into thin layers ( laminae) less than one centimeter in thickness. This property is called '' fissility''. Shale is the most common sedimentary rock. The term ''shale'' is sometimes applied more broadly, as essentially a synonym for mudrock, rather than in the more narrow sense of clay-rich fissile mudrock. Texture Shale typically exhibits varying degrees of fissility. Because of the parallel orientation of clay mineral flakes in shale, it breaks into thin layers, often splintery and usually parallel to the otherwise indistinguishable beddin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Granite
Granite () is a coarse-grained (phaneritic) intrusive igneous rock composed mostly of quartz, alkali feldspar, and plagioclase. It forms from magma with a high content of silica and alkali metal oxides that slowly cools and solidifies underground. It is common in the continental crust of Earth, where it is found in igneous intrusions. These range in size from dikes only a few centimeters across to batholiths exposed over hundreds of square kilometers. Granite is typical of a larger family of ''granitic rocks'', or ''granitoids'', that are composed mostly of coarse-grained quartz and feldspars in varying proportions. These rocks are classified by the relative percentages of quartz, alkali feldspar, and plagioclase (the QAPF classification), with true granite representing granitic rocks rich in quartz and alkali feldspar. Most granitic rocks also contain mica or amphibole minerals, though a few (known as leucogranites) contain almost no dark minerals. Granite is nearly alway ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thorium
Thorium is a weakly radioactive metallic chemical element with the symbol Th and atomic number 90. Thorium is silvery and tarnishes black when it is exposed to air, forming thorium dioxide; it is moderately soft and malleable and has a high melting point. Thorium is an electropositive actinide whose chemistry is dominated by the +4 oxidation state; it is quite reactive and can ignite in air when finely divided. All known thorium isotopes are unstable. The most stable isotope, 232Th, has a half-life of 14.05 billion years, or about the age of the universe; it decays very slowly via alpha decay, starting a decay chain named the thorium series that ends at stable 208 Pb. On Earth, thorium and uranium are the only significantly radioactive elements that still occur naturally in large quantities as primordial elements. Thorium is estimated to be over three times as abundant as uranium in the Earth's crust, and is chiefly refined from monazite sands as a by-product of extracti ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bad Gastein
Bad Gastein (; formerly ''Badgastein''; Southern Bavarian: ''Bod Goschdei'') is a spa town in the district of St. Johann im Pongau District, St. Johann im Pongau, in the Austrian state of Salzburg (state), Salzburg. Picturesquely situated in a high valley of the Hohe Tauern mountain range, it is known for the Gastein Waterfall and a variety of ''Belle Époque'' hotel buildings. Geography Bad Gastein is located in the historic ''Pongau'' region, the municipal area of about is the largest in St. Johann im Pongau District. It stretches along the upper Gastein Valley following the course of the Gastein Ache creek, a right tributary of the Salzach river. The valley separates the Hohe Tauern Ankogel Group in the east from the Goldberg Group in the west. The town centre is located at the Gastein Falls, about above sea level. It is characterised by numerous historic multi-storey hotel buildings erected on the steep slopes. The Gastein municipality comprises the Cadastral community, ca ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
The Linde Group
Linde plc is a global multinational chemical company founded in Germany and, since 2018, domiciled in Ireland and headquartered in the United Kingdom. Linde is the world's largest industrial gas company by market share and revenue. It serves customers in the healthcare, petroleum refining, manufacturing, food, beverage carbonation, fiber-optics, steel making, aerospace, material handling equipment (MHE), chemicals, electronics and water treatment industries. The company's primary business is the manufacturing and distribution of atmospheric gases, including oxygen, nitrogen, argon, rare gases, and process gases, including carbon dioxide, helium, hydrogen, ammonia, electronic gases, specialty gases, and acetylene. The company was formed by the 2018 merger of Linde AG of Germany (founded in 1879) and Praxair (founded in 1907 as Linde Air Products Company) of the United States. The resulting holding company was incorporated in Ireland, with principal executive offices in Guildfor ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Picogram
To help compare different orders of magnitude, the following lists describe various mass levels between 10−59 kg and 1052 kg. The least massive thing listed here is a graviton, and the most massive thing is the observable universe. Typically, an object having greater mass will also have greater weight (see mass versus weight), especially if the objects are subject to the same gravitational field strength. Units of mass The table at right is based on the kilogram (kg), the base unit of mass in the International System of Units ( SI). The kilogram is the only standard unit to include an SI prefix (''kilo-'') as part of its name. The ''gram'' (10−3 kg) is an SI derived unit of mass. However, the ''names'' of all SI mass units are based on ''gram'', rather than on ''kilogram''; thus 103 kg is a ''megagram'' (106 g), not a *''kilokilogram''. The ''tonne'' (t) is an SI-compatible unit of mass equal to a megagram (''Mg''), or 103 kg. The unit is in common use for m ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Radon Concentration Next To Uranium Mine
Radon is a chemical element with the symbol Rn and atomic number 86. It is a radioactive, colourless, odourless, tasteless noble gas. It occurs naturally in minute quantities as an intermediate step in the normal radioactive decay chains through which thorium and uranium slowly decay into various short-lived radioactive elements and lead. Radon itself is the immediate decay product of radium. Its most stable isotope, 222Rn, has a half-life of only 3.8 days, making it one of the rarest elements. Since thorium and uranium are two of the most common radioactive elements on Earth, while also having three isotopes with half-lives on the order of several billion years, radon will be present on Earth long into the future despite its short half-life. The decay of radon produces many other short-lived nuclides, known as "radon daughters", ending at stable isotopes of lead. [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Radioisotope
A radionuclide (radioactive nuclide, radioisotope or radioactive isotope) is a nuclide that has excess nuclear energy, making it unstable. This excess energy can be used in one of three ways: emitted from the nucleus as gamma radiation; transferred to one of its electrons to release it as a conversion electron; or used to create and emit a new particle (alpha particle or beta particle) from the nucleus. During those processes, the radionuclide is said to undergo radioactive decay. These emissions are considered ionizing radiation because they are energetic enough to liberate an electron from another atom. The radioactive decay can produce a stable nuclide or will sometimes produce a new unstable radionuclide which may undergo further decay. Radioactive decay is a random process at the level of single atoms: it is impossible to predict when one particular atom will decay. However, for a collection of atoms of a single nuclide the decay rate, and thus the half-life (''t''1/2) for t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
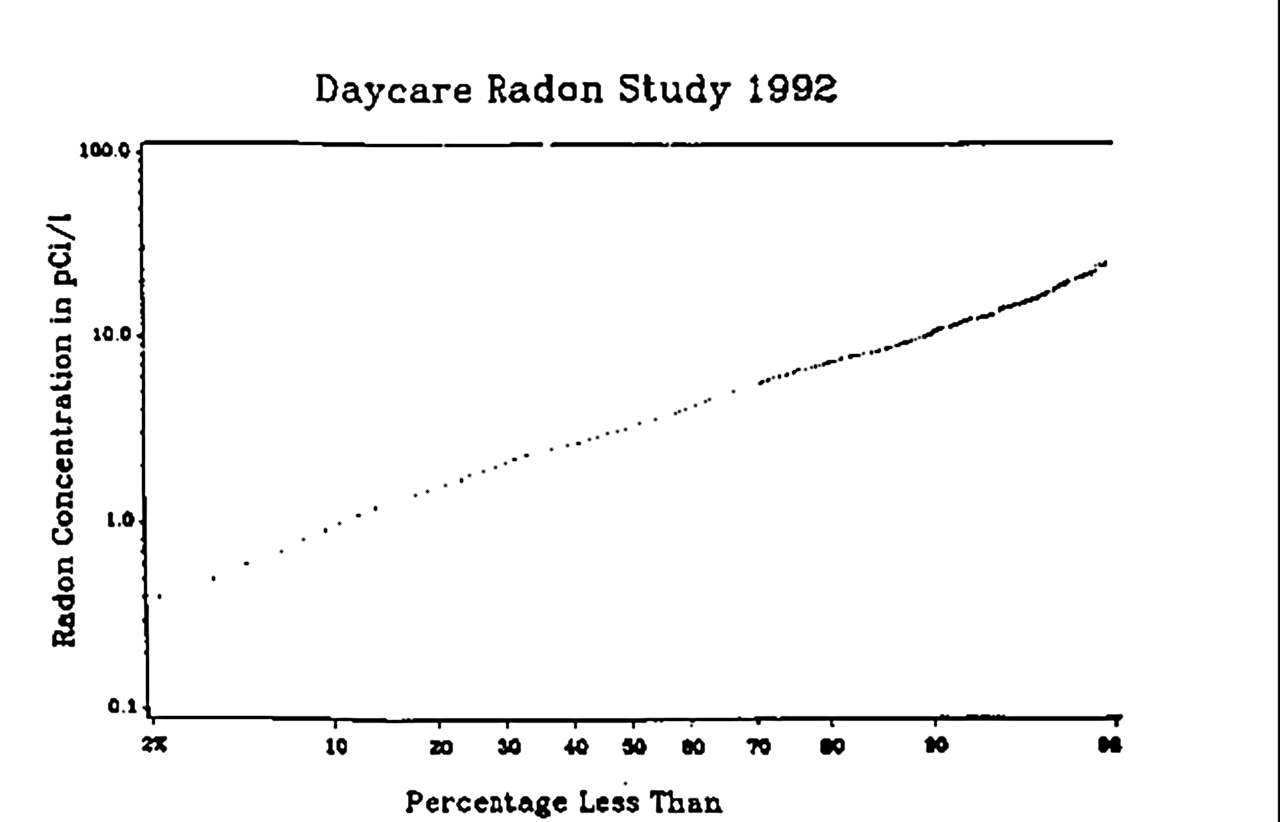
Working Level
Working level (WL) is a historical unit of concentration of radioactive decay products of radon, applied to uranium mining environment. One working level refers to the concentration of short-lived decay products of radon in equilibrium with 3,700 Bq/m (100 pCi/L) in air. These decay products would emit 1.3 × 10 MeV in complete decay. The Nuclear Regulatory Commission uses this definition. Working level month (WLM) is a closely related quantity, referring to exposure to one working level for 170 hours per month. This comes from assuming a 40-hour work week. In 2002, the NRC regulations limited exposure in mines to 0.3 WL, which was comparable with the standards of International Commission on Radiological Protection The International Commission on Radiological Protection (ICRP) is an independent, international, non-governmental organization, with the mission to protect people, animals, and the environment from the harmful effects of ionising radiation. Its r ... at the time. R ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |