|
Helium Trimer
The helium trimer (or trihelium) is a weakly bound molecule consisting of three helium atoms. Van der Waals forces link the atoms together. The combination of three atoms is much more stable than the two-atom helium dimer. The three-atom combination of helium-4 atoms is an Efimov state. Helium-3 is predicted to form a trimer, although ground state dimers containing helium-3 are completely unstable. Helium trimer molecules have been produced by expanding cold helium gas from a nozzle into a vacuum chamber. Such a set up also produces the helium dimer and other helium atom clusters. The existence of the molecule was proven by matter wave diffraction through a diffraction grating. Properties of the molecules can be discovered by Coulomb explosion imaging. In this process, a laser ionizes all three atoms simultaneously, which then fly away from each other due to electrostatic repulsion and are detected. The helium trimer is large, being more than 100 Å, which is even larger tha ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Van Der Waals Force
In molecular physics, the van der Waals force is a distance-dependent interaction between atoms or molecules. Unlike ionic or covalent bonds, these attractions do not result from a chemical electronic bond; they are comparatively weak and therefore more susceptible to disturbance. The van der Waals force quickly vanishes at longer distances between interacting molecules. Named after Dutch physicist Johannes Diderik van der Waals, the van der Waals force plays a fundamental role in fields as diverse as supramolecular chemistry, structural biology, polymer science, nanotechnology, surface science, and condensed matter physics. It also underlies many properties of organic compounds and molecular solids, including their solubility in polar and non-polar media. If no other force is present, the distance between atoms at which the force becomes repulsive rather than attractive as the atoms approach one another is called the van der Waals contact distance; this phenomenon resul ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Helium Dimer
The helium dimer is a van der Waals molecule with formula He2 consisting of two helium atoms. This chemical is the largest diatomic molecule—a molecule consisting of two atoms bonded together. The bond that holds this dimer together is so weak that it will break if the molecule rotates, or vibrates too much. It can only exist at very low cryogenic temperatures. Two excited helium atoms can also bond to each other in a form called an excimer. This was discovered from a spectrum of helium that contained bands first seen in 1912. Written as He2* with the * meaning an excited state, it is the first known Rydberg molecule. Several dihelium ions also exist, having net charges of negative one, positive one, and positive two. Two helium atoms can be confined together without bonding in the cage of a fullerene. Molecule Based on molecular orbital theory, He2 should not exist, and a chemical bond cannot form between the atoms. However, the van der Waals force exists between helium at ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Helium-4
Helium-4 () is a stable isotope of the element helium. It is by far the more abundant of the two naturally occurring isotopes of helium, making up about 99.99986% of the helium on Earth. Its nucleus is identical to an alpha particle, and consists of two protons and two neutrons. Alpha decay of heavy elements in the Earth's crust is the source of most naturally occurring helium-4 on Earth, produced after the planet cooled and solidified. While it is also produced by nuclear fusion in stars, most helium-4 in the Sun and in the universe is thought to have been produced by the Big Bang, and is referred to as " primordial helium". However, primordial helium-4 is largely absent from the Earth, having escaped during the high-temperature phase of Earth's formation. Helium-4 makes up about one quarter of the ordinary matter in the universe by mass, with almost all of the rest being hydrogen. When liquid helium-4 is cooled to below , it becomes a superfluid, with properties that are ver ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Efimov State
The Efimov effect is an effect in the quantum mechanics of few-body systems predicted by the Russian theoretical physicist V. N. Efimov in 1970. Efimov’s effect is where three identical bosons interact, with the prediction of an infinite series of excited three-body energy levels when a two-body state is exactly at the dissociation threshold. One corollary is that there exist bound states (called Efimov states) of three bosons even if the two-particle attraction is too weak to allow two bosons to form a pair. A (three-particle) Efimov state, where the (two-body) sub-systems are unbound, is often depicted symbolically by the Borromean rings. This means that if one of the particles is removed, the remaining two fall apart. In this case, the Efimov state is also called a Borromean state. Theory Efimov predicted that, as the pair interactions among three identical bosons approach resonance—that is, as the binding energy of some two-body bound state approaches zero or the scatteri ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Helium-3
Helium-3 (3He see also helion) is a light, stable isotope of helium with two protons and one neutron (the most common isotope, helium-4, having two protons and two neutrons in contrast). Other than protium (ordinary hydrogen), helium-3 is the only stable isotope of any element with more protons than neutrons. Helium-3 was discovered in 1939. Helium-3 occurs as a primordial nuclide, escaping from Earth's crust into its atmosphere and into outer space over millions of years. Helium-3 is also thought to be a natural nucleogenic and cosmogenic nuclide, one produced when lithium is bombarded by natural neutrons, which can be released by spontaneous fission and by nuclear reactions with cosmic rays. Some of the helium-3 found in the terrestrial atmosphere is also an artifact of atmospheric and underwater nuclear weapons testing. Much speculation has been made over the possibility of helium-3 as a future energy source. Unlike most nuclear fusion reactions, the fusion of helium-3 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Matter Wave
Matter waves are a central part of the theory of quantum mechanics, being an example of wave–particle duality. All matter exhibits wave-like behavior. For example, a beam of electrons can be diffracted just like a beam of light or a water wave. In most cases, however, the wavelength is too small to have a practical impact on day-to-day activities. The concept that matter behaves like a wave was proposed by French physicist Louis de Broglie () in 1924. It is also referred to as the ''de Broglie hypothesis''. Matter waves are referred to as ''de Broglie waves''. The ''de Broglie wavelength'' is the wavelength, , associated with a massive particle (i.e., a particle with mass, as opposed to a massless particle) and is related to its momentum, , through the Planck constant, : : \lambda = \frac=\frac. Wave-like behavior of matter was first experimentally demonstrated by George Paget Thomson's thin metal diffraction experiment, and independently in the Davisson–Germer experiment, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diffraction Grating
In optics, a diffraction grating is an optical component with a periodic structure that diffracts light into several beams travelling in different directions (i.e., different diffraction angles). The emerging coloration is a form of structural coloration. The directions or diffraction angles of these beams depend on the wave (light) incident angle to the diffraction grating, the spacing or distance between adjacent diffracting elements (e.g., parallel slits for a transmission grating) on the grating, and the wavelength of the incident light. The grating acts as a dispersive element. Because of this, diffraction gratings are commonly used in monochromators and spectrometers, but other applications are also possible such as optical encoders for high precision motion control and wavefront measurement. For typical applications, a reflective grating has ridges or ''rulings'' on its surface while a transmissive grating has transmissive or hollow slits on its surface. Such a grating ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Coulomb Explosion
In condensed-matter physics, Coulombic explosions are a mechanism for transforming energy in intense electromagnetic fields into atomic motion and are thus useful for controlled destruction of relatively robust molecules. The explosions are a prominent technique in laser-based machining, and appear naturally in certain high-energy reactions. Mechanism The Coulombic repulsion of particles having the same electric charge can break the bonds that hold solids together. When done with a narrow laser beam, a small amount of solid explodes into a plasma of ionized atomic particles. It may be shown that the Coulomb explosion occurs in the same critical parameter regime as the superradiant phase transition i.e. when the destabilizing interactions become overwhelming and dominate over the native oscillatory phonon solid cluster binding motions which is also characteristic for the diamond synthesis. With their low mass, outer valence electrons responsible for chemical bonding are easil ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electrostatic Repulsion
Electrostatics is a branch of physics that studies electric charges at rest ( static electricity). Since classical times, it has been known that some materials, such as amber, attract lightweight particles after rubbing. The Greek word for amber, (), was thus the source of the word 'electricity'. Electrostatic phenomena arise from the forces that electric charges exert on each other. Such forces are described by Coulomb's law. Even though electrostatically induced forces seem to be rather weak, some electrostatic forces are relatively large. The force between an electron and a proton, which together make up a hydrogen atom, is about 36 orders of magnitude stronger than the gravitational force acting between them. There are many examples of electrostatic phenomena, from those as simple as the attraction of plastic wrap to one's hand after it is removed from a package, to the apparently spontaneous explosion of grain silos, the damage of electronic components during manufact ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Equilateral Triangle
In geometry, an equilateral triangle is a triangle in which all three sides have the same length. In the familiar Euclidean geometry, an equilateral triangle is also equiangular; that is, all three internal angles are also congruent to each other and are each 60°. It is also a regular polygon, so it is also referred to as a regular triangle. Principal properties Denoting the common length of the sides of the equilateral triangle as a, we can determine using the Pythagorean theorem that: *The area is A=\frac a^2, *The perimeter is p=3a\,\! *The radius of the circumscribed circle is R = \frac *The radius of the inscribed circle is r=\frac a or r=\frac *The geometric center of the triangle is the center of the circumscribed and inscribed circles *The altitude (height) from any side is h=\frac a Denoting the radius of the circumscribed circle as ''R'', we can determine using trigonometry that: *The area of the triangle is \mathrm=\fracR^2 Many of these quantities have simple r ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Homonuclear Triatomic Molecules
Homonuclear molecules, or homonuclear species, are molecules composed of only one element. Homonuclear molecules may consist of various numbers of atoms. The size of the molecule an element can form depends on the element's properties, and some elements form molecules of more than one size. The most familiar homonuclear molecules are diatomic molecule, which consist of two atoms, although not all diatomic molecules are homonuclear. Homonuclear diatomic molecules include hydrogen (H2), oxygen (O2), nitrogen (N2) and all of the halogens. Ozone (O3) is a common triatomic homonuclear molecule. Homonuclear tetratomic molecules include arsenic (As4) and phosphorus (P4). Allotropes are different chemical forms of the same element (not containing any other element). In that sense, allotropes are all homonuclear. Many elements have multiple allotropic forms. In addition to the most common form of gaseous oxygen, O2, and ozone, there are other allotropes of oxygen. Sulfur forms several allo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
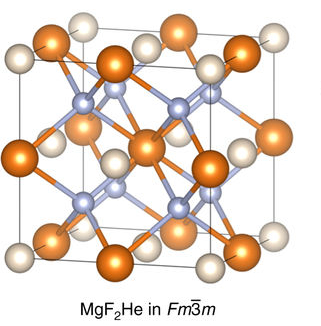
Helium Compounds
Helium is the smallest and the lightest noble gas and one of the most unreactive elements, so it was commonly considered that helium compounds cannot exist at all, or at least under normal conditions. Helium's first ionization energy of 24.57 eV is the highest of any element. Helium has a complete shell of electrons, and in this form the atom does not readily accept any extra electrons nor join with anything to make covalent compounds. The electron affinity is 0.080 eV, which is very close to zero. The helium atom is small with the radius of the outer electron shell at 0.29 Å. Helium is a very hard atom with a Pearson hardness of 12.3 eV. It has the lowest polarizability of any kind of atom, however, very weak van der Waals forces exist between helium and other atoms. This force may exceed repulsive forces, so at extremely low temperatures helium may form van der Waals molecules. Helium has the lowest boiling point (4.2 K) of any known substance. Repulsive fo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |


_0.002_K_region.png)