|
Hauser Base
Hauser bases, also called magnesium amide bases, are magnesium compounds used in organic chemistry as Base (chemistry), bases for metalation, metalation reactions. These compounds were first described by Charles R. Hauser in 1947. Compared with organolithium reagents, the magnesium compounds have more covalent, and therefore less reactive, metal-ligand bonds. Consequently, they display a higher degree of functional group tolerance and a much greater chemoselectivity. Generally, Hauser bases are used at room temperature while reactions with organolithium reagents are performed at low temperatures, commonly at −78 °C. Structure Solid state structure Like all Grignard reaction, Grignard dimers, Hauser bases derived from 2,2,6,6-Tetramethylpiperidine, 2,2,6,6-tetramethylpiperidine (TMP) and Bis(trimethylsilyl)amine, HMDS are bridged by halides in the solid state. In contrast to Grignard reagents, dimeric amido bridged Hauser bases exist, too. All have one in common: the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Magnesium
Magnesium is a chemical element with the symbol Mg and atomic number 12. It is a shiny gray metal having a low density, low melting point and high chemical reactivity. Like the other alkaline earth metals (group 2 of the periodic table) it occurs naturally only in combination with other elements and it almost always has an oxidation state of +2. It reacts readily with air to form a thin passivation coating of magnesium oxide that inhibits further corrosion of the metal. The free metal burns with a brilliant-white light. The metal is obtained mainly by electrolysis of magnesium salts obtained from brine. It is less dense than aluminium and is used primarily as a component in strong and lightweight alloys that contain aluminium. In the cosmos, magnesium is produced in large, aging stars by the sequential addition of three helium nuclei to a carbon nucleus. When such stars explode as supernovas, much of the magnesium is expelled into the interstellar medium where it ma ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Heteroaromatic
In chemistry, aromaticity is a chemical property of cyclic ( ring-shaped), ''typically'' planar (flat) molecular structures with pi bonds in resonance (those containing delocalized electrons) that gives increased stability compared to saturated compounds having single bonds, and other geometric or connective non-cyclic arrangements with the same set of atoms. Aromatic rings are very stable and do not break apart easily. Organic compounds that are not aromatic are classified as aliphatic compounds—they might be cyclic, but only aromatic rings have enhanced stability. The term ''aromaticity'' with this meaning is historically related to the concept of having an aroma, but is a distinct property from that meaning. Since the most common aromatic compounds are derivatives of benzene (an aromatic hydrocarbon common in petroleum and its distillates), the word ''aromatic'' occasionally refers informally to benzene derivatives, and so it was first defined. Nevertheless, many non- ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aromatic
In chemistry, aromaticity is a chemical property of cyclic ( ring-shaped), ''typically'' planar (flat) molecular structures with pi bonds in resonance (those containing delocalized electrons) that gives increased stability compared to saturated compounds having single bonds, and other geometric or connective non-cyclic arrangements with the same set of atoms. Aromatic rings are very stable and do not break apart easily. Organic compounds that are not aromatic are classified as aliphatic compounds—they might be cyclic, but only aromatic rings have enhanced stability. The term ''aromaticity'' with this meaning is historically related to the concept of having an aroma, but is a distinct property from that meaning. Since the most common aromatic compounds are derivatives of benzene (an aromatic hydrocarbon common in petroleum and its distillates), the word ''aromatic'' occasionally refers informally to benzene derivatives, and so it was first defined. Nevertheless, many ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
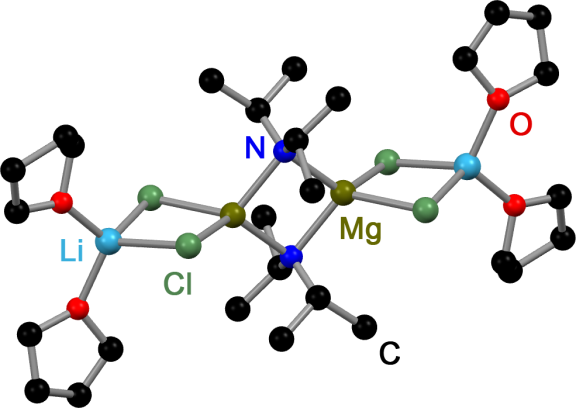
Turbo-Hauser Bases
''Turbo''-Hauser bases are Hauser base, amido magnesium halides that contain stoichiometric amounts of Lithium chloride, LiCl. These mixed Mg/Li amides of the type R2NMgCl⋅LiCl are used in organic chemistry as non-nucleophilic bases for metalation, metalation reactions of aromatic and heteroaromatic substrates. Compared to their LiCl free ancestors ''Turbo''-Hauser bases show an enhanced kinetic basicity, excellent regioselectivity, high functional group tolerance and a better solubility. Preparation Typically ''Turbo''-Hauser bases are prepared by treating an amine with a Grignard reaction, Grignard reagent and lithium chloride. In some cases they are prepared by treating a lithium amide with MgCl2: : : Common ''Turbo''-Hauser bases: R'2NH = ''i''Pr2NMgCl·LiCl (''i''Pr-''Turbo''-Hauser base), TMPMgCl·LiCl, TMP (''Turbo''-Hauser base or ''Knochel''-Hauser Base) Structure In solution, ''Turbo''-Hauser bases participate in temperature- and concentration-dependent equilibria. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Metal Amides
Metal amides (systematic name metal azanides) are a class of coordination compounds composed of a metal center with amide ligands of the form NR2−. Amide ligands have two electron pairs available for bonding. In principle, they can be terminal or bridging. In these two examples, the dimethylamido ligands are both bridging and terminal: File:Tris(dimethylamino)aluminium dimer.png, Tris(dimethylamino)aluminium dimer File:Tris(dimethylamino)gallium dimer.png, Tris(dimethylamino)gallium dimer File:Ti(NMe2)4.png, Tetrakis(dimethylamino)titanium File:Ta(NMe2)5.png, Pentakis(dimethylamido)tantalum In practice, bulky amide ligands have a lesser tendency to bridge. Amide ligands may participate in metal-ligand π-bonding giving a complex with the metal center being co-planar with the nitrogen and substituents. Metal bis(trimethylsilyl)amides form a significant subcategory of metal amide compounds. These compounds tend to be discrete and soluble in organic solvents. Alkali metal amid ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Grignard Reagent
A Grignard reagent or Grignard compound is a chemical compound with the general formula , where X is a halogen and R is an organic group, normally an alkyl or aryl. Two typical examples are methylmagnesium chloride and phenylmagnesium bromide . They are a subclass of the organomagnesium compounds. Grignard compounds are popular reagents in organic synthesis for creating new carbon-carbon bonds. For example, when reacted with another halogenated compound in the presence of a suitable catalyst, they typically yield and the magnesium halide as a byproduct; and the latter is insoluble in the solvents normally used. In this aspect, they are similar to organolithium reagents. Pure Grignard reagents are extremely reactive solids. They are normally handled as solutions in solvents such as diethyl ether or tetrahydrofuran; which are relatively stable as long as water is excluded. In such a medium, a Grignard reagent is invariably present as a complex with the magnesium atom conn ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Homoleptic
In inorganic chemistry, a homoleptic chemical compound is a metal compound with all ligands identical. The term uses the " homo-" prefix to indicate that something is the same for all. Any metal species which has more than one type of ligand is heteroleptic. Some compounds with names that suggest that they are homoleptic are in fact heteroleptic, because they have ligands in them which are not featured in the name. For instance dialkyl magnesium complexes, which are found in the equilibrium which exists in a solution of a Grignard reagent in an ether, have two ether ligands attached to each magnesium centre. Another example is a solution of trimethyl aluminium in an ether solvent (such as THF); similar chemistry should be expected for a triaryl or trialkyl borane. It is possible for some ligands such as DMSO to bind with two or more different coordination modes. It would still be reasonable to consider a complex which has only one type of ligand but with different coordination m ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Heteroleptic
In inorganic chemistry, a homoleptic chemical compound is a metal compound with all ligands identical. The term uses the " homo-" prefix to indicate that something is the same for all. Any metal species which has more than one type of ligand is heteroleptic. Some compounds with names that suggest that they are homoleptic are in fact heteroleptic, because they have ligands in them which are not featured in the name. For instance dialkyl magnesium complexes, which are found in the equilibrium which exists in a solution of a Grignard reagent in an ether, have two ether ligands attached to each magnesium centre. Another example is a solution of trimethyl aluminium in an ether solvent (such as THF); similar chemistry should be expected for a triaryl or trialkyl borane. It is possible for some ligands such as DMSO to bind with two or more different coordination modes. It would still be reasonable to consider a complex which has only one type of ligand but with different coordination m ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |


.png)