|
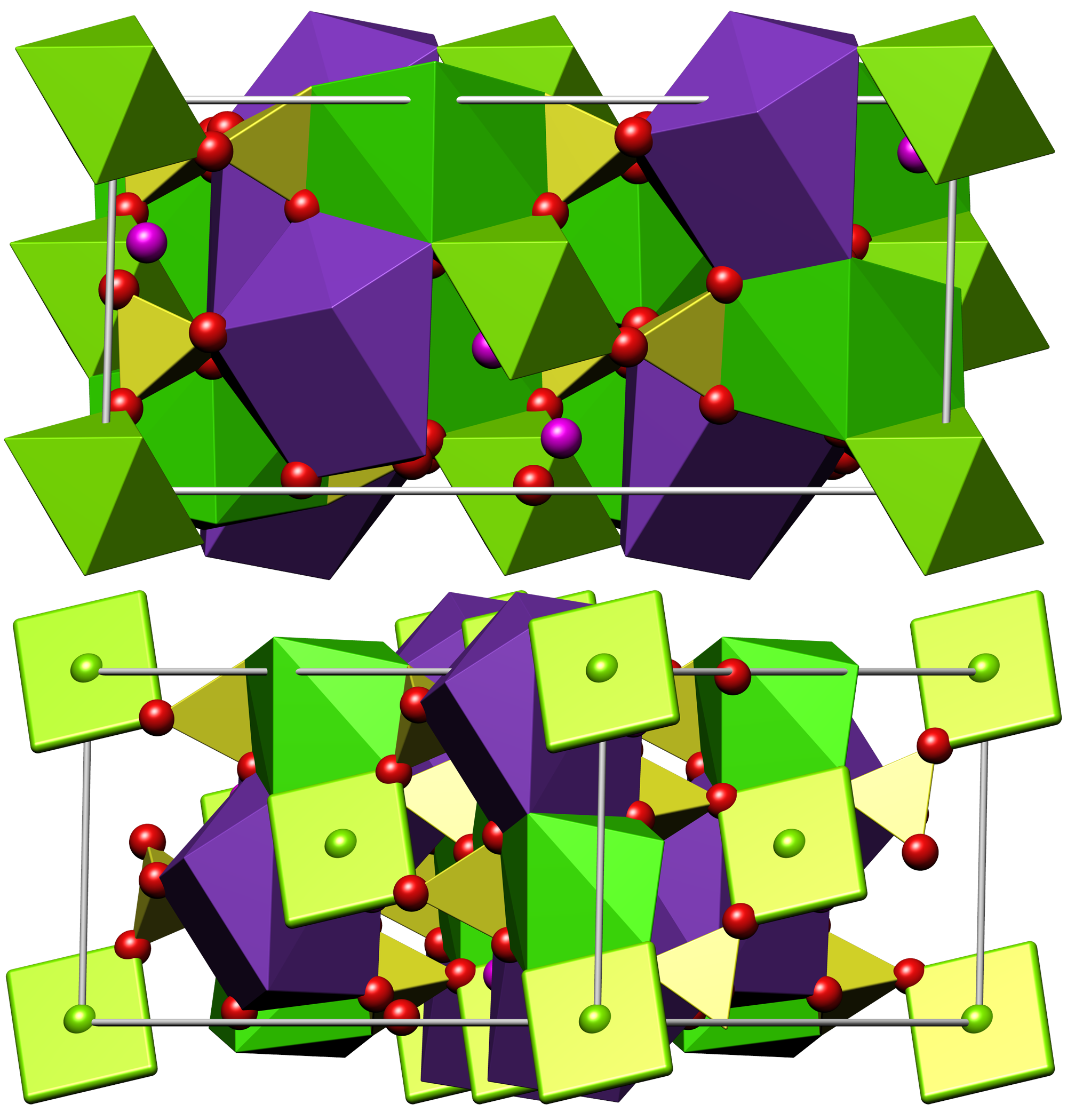
Glauberite
Glauberite is a monoclinic sodium calcium sulfate mineral with the formula Na2 Ca( S O4)2. It was first described in 1808 for material from the El Castellar Mine, Villarrubia de Santiago, Toledo, Castile-La Mancha, Spain. It was named for the extracted Glauber's salts after the German alchemist Johann Rudolf Glauber (1604–1668). Glauberite often forms in continental and marine evaporite deposits, but may also form from hydrothermal deposits, as mineral sublimates deposited near fumaroles, in amygdules in basalt, and in nitrate deposits in arid climates. It occurs associated with halite, polyhalite, anhydrite, gypsum, thenardite, mirabilite, sassolite and blodite. Because of its solubility, glauberite is often dissolved away from the crystal matrix leaving a distinctly shaped hollow cast. Its mineral composition is readily altered into other minerals as pseudomorphs. Gypsum pseudomorphs are common due to increased humidity. Glauberite, its cast impressions, and its pseudo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfate Mineral
The sulfate minerals are a class of minerals that include the sulfate ion () within their structure. The sulfate minerals occur commonly in primary evaporite depositional environments, as gangue minerals in hydrothermal veins and as secondary minerals in the oxidizing zone of sulfide mineral deposits. The chromate and manganate minerals have a similar structure and are often included with the sulfates in mineral classification systems.Klein, Cornelis and Cornelius S. Hurlbut, 1985, ''Manual of Mineralogy,'' 20th ed., John Wiley and Sons, New York, pp. 347–354 . Sulfate minerals include: *Anhydrous sulfates **Barite BaSO4 ** Celestite SrSO4 **Anglesite PbSO4 **Anhydrite CaSO4 **Hanksite Na22K(SO4)9(CO3)2Cl *Hydroxide and hydrous sulfates **Gypsum CaSO4·2H2O **Chalcanthite CuSO4·5H2O **Kieserite MgSO4·H2O ** Starkeyite MgSO4·4H2O **Hexahydrite MgSO4·6H2O **Epsomite MgSO4·7H2O **Meridianiite MgSO4·11H2O **Melanterite FeSO4·7H2O **Antlerite Cu3SO4(OH)4 **Brochantite Cu4SO4 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfate Minerals
The sulfate minerals are a class of minerals that include the sulfate ion () within their structure. The sulfate minerals occur commonly in primary evaporite depositional environments, as gangue minerals in hydrothermal Vein (geology), veins and as secondary minerals in the Redox, oxidizing zone of sulfide mineral deposits. The Chromate ion, chromate and manganate minerals have a similar structure and are often included with the sulfates in mineral classification systems.Klein, Cornelis and Cornelius S. Hurlbut, 1985, ''Manual of Mineralogy,'' 20th ed., John Wiley and Sons, New York, pp. 347–354 . Sulfate minerals include: *Anhydrous sulfates **Barite BaSO4 **Celestite SrSO4 **Anglesite PbSO4 **Anhydrite CaSO4 **Hanksite Na22K(SO4)9(CO3)2Cl *Hydroxide and hydrous sulfates **Gypsum CaSO4·2H2O **Chalcanthite CuSO4·5H2O **Kieserite MgSO4·H2O **Starkeyite MgSO4·4H2O **Hexahydrite MgSO4·6H2O **Epsomite MgSO4·7H2O **Meridianiite MgSO4·11H2O **Melanterite FeSO4·7H2O **Antlerite ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Volcanic Sublimate
A volcano is a rupture in the crust of a planetary-mass object, such as Earth, that allows hot lava, volcanic ash, and gases to escape from a magma chamber below the surface. On Earth, volcanoes are most often found where tectonic plates are diverging or converging, and most are found underwater. For example, a mid-ocean ridge, such as the Mid-Atlantic Ridge, has volcanoes caused by divergent tectonic plates whereas the Pacific Ring of Fire has volcanoes caused by convergent tectonic plates. Volcanoes can also form where there is stretching and thinning of the crust's plates, such as in the East African Rift and the Wells Gray-Clearwater volcanic field and Rio Grande rift in North America. Volcanism away from plate boundaries has been postulated to arise from upwelling diapirs from the core–mantle boundary, deep in the Earth. This results in hotspot volcanism, of which the Hawaiian hotspot is an example. Volcanoes are usually not created where two tectonic plates slide pa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Solubility
In chemistry, solubility is the ability of a substance, the solute, to form a solution with another substance, the solvent. Insolubility is the opposite property, the inability of the solute to form such a solution. The extent of the solubility of a substance in a specific solvent is generally measured as the concentration of the solute in a saturated solution, one in which no more solute can be dissolved. At this point, the two substances are said to be at the solubility equilibrium. For some solutes and solvents, there may be no such limit, in which case the two substances are said to be " miscible in all proportions" (or just "miscible"). The solute can be a solid, a liquid, or a gas, while the solvent is usually solid or liquid. Both may be pure substances, or may themselves be solutions. Gases are always miscible in all proportions, except in very extreme situations,J. de Swaan Arons and G. A. M. Diepen (1966): "Gas—Gas Equilibria". ''Journal of Chemical Physics'', ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sassolite
Sassolite is a borate mineral, specifically the mineral form of boric acid. It is usually white to gray, and colourless in transmitted light. It can also take on a yellow colour from sulfur impurities, or brown from iron oxides. History and occurrence Its mineral form was first described in 1800, and was named after Sasso Pisano, Castelnuovo Val di Cecina, Pisa Province, Tuscany, Italy where it was found. The mineral may be found in lagoons throughout Tuscany and Sasso. It is also found in the Lipari Islands and the US state of Nevada. It occurs in volcanic fumaroles and hot springs, deposited from steam, as well as in bedded sedimentary evaporite deposits. See also * List of minerals * Borax Borax is a salt (ionic compound), a hydrated borate of sodium, with chemical formula often written . It is a colorless crystalline solid, that dissolves in water to make a basic solution. It is commonly available in powder or granular form, ... References External links B ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mirabilite
Mirabilite, also known as Glauber's salt, is a hydrous sodium sulfate mineral with the chemical formula Na2SO4·10H2O. It is a vitreous, colorless to white monoclinic mineral that forms as an evaporite from sodium sulfate-bearing brines. It is found around saline springs and along saline playa lakes. Associated minerals include gypsum, halite, thenardite, trona, glauberite, and epsomite. Mirabilite is unstable and quickly dehydrates in dry air, the prismatic crystals turning into a white powder, thenardite (Na2SO4). In turn, thenardite can also absorb water and converts to mirabilite. Mirabilite is used as a purgative and anti-inflammatory remedy in the Traditional Chinese medicine; in Mandarin, it is called máng xiāo. The name 'mirabilite' is based on the phrase ''"Sal mirabilis"'' (Latin for "wonderful salt") used by Johann Rudolph Glauber when he inadvertently synthesized mirabilite. Mirabilite is found in several areas within the Mammoth Cave system, where it appears to ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gypsum
Gypsum is a soft sulfate mineral composed of calcium sulfate dihydrate, with the chemical formula . It is widely mined and is used as a fertilizer and as the main constituent in many forms of plaster, blackboard or sidewalk chalk, and drywall. Alabaster, a fine-grained white or lightly tinted variety of gypsum, has been used for sculpture by many cultures including Ancient Egypt, Mesopotamia, Ancient Rome, the Byzantine Empire, and the Nottingham alabasters of Medieval England. Gypsum also crystallizes as translucent crystals of selenite. It forms as an evaporite mineral and as a hydration product of anhydrite. The Mohs scale of mineral hardness defines gypsum as hardness value 2 based on scratch hardness comparison. Etymology and history The word ''gypsum'' is derived from the Greek word (), "plaster". Because the quarries of the Montmartre district of Paris have long furnished burnt gypsum (calcined gypsum) used for various purposes, this dehydrated gypsum became known ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Anhydrite
Anhydrite, or anhydrous calcium sulfate, is a mineral with the chemical formula CaSO4. It is in the orthorhombic crystal system, with three directions of perfect cleavage parallel to the three planes of symmetry. It is not isomorphous with the orthorhombic barium ( baryte) and strontium ( celestine) sulfates, as might be expected from the chemical formulas. Distinctly developed crystals are somewhat rare, the mineral usually presenting the form of cleavage masses. The Mohs hardness is 3.5, and the specific gravity is 2.9. The color is white, sometimes greyish, bluish, or purple. On the best developed of the three cleavages, the lustre is pearly; on other surfaces it is glassy. When exposed to water, anhydrite readily transforms to the more commonly occurring gypsum, (CaSO4·2H2O) by the absorption of water. This transformation is reversible, with gypsum or calcium sulfate hemihydrate forming anhydrite by heating to around under normal atmospheric conditions. Anhydrite is comm ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polyhalite
Polyhalite is an evaporite mineral, a hydrated sulfate of potassium, calcium and magnesium with formula: . Polyhalite crystallizes in the triclinic system, although crystals are very rare. The normal habit is massive to fibrous. It is typically colorless, white to gray, although it may be brick red due to iron oxide inclusions. It has a Mohs hardness of 3.5 and a specific gravity of 2.8. It occurs in sedimentary marine evaporites and is a major potassium ore mineral in the Carlsbad deposits of New Mexico. It is also present as a 2–3% contaminant of Himalayan salt. Polyhalite was first described in 1818 for specimens from its type locality in Salzburg, Austria. The name comes from the German ''Polyhalit'', which comes from the Ancient Greek words (''polys'') and ' (''hals''), which mean "many" and "salt", and the German ending ''-it'' (which comes from the Latin ending -ites, which originally also came from Greek), which is used like the English ending ''-ite'' to form the nam ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Halite
Halite (), commonly known as rock salt, is a type of salt, the mineral (natural) form of sodium chloride ( Na Cl). Halite forms isometric crystals. The mineral is typically colorless or white, but may also be light blue, dark blue, purple, pink, red, orange, yellow or gray depending on inclusion of other materials, impurities, and structural or isotopic abnormalities in the crystals. It commonly occurs with other evaporite deposit minerals such as several of the sulfates, halides, and borates. The name ''halite'' is derived from the Ancient Greek word for "salt", ἅλς (''háls''). Occurrence Halite dominantly occurs within sedimentary rocks where it has formed from the evaporation of seawater or salty lake water. Vast beds of sedimentary evaporite minerals, including halite, can result from the drying up of enclosed lakes and restricted seas. Such salt beds may be hundreds of meters thick and underlie broad areas. Halite occurs at the surface today in playas in regio ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |