|
Glucose Syrup
Glucose syrup, also known as confectioner's glucose, is a syrup made from the hydrolysis of starch. Glucose is a sugar. Maize (corn) is commonly used as the source of the starch in the US, in which case the syrup is called "corn syrup", but glucose syrup is also made from potatoes and wheat, and less often from barley, rice and cassava.p. 21 Glucose syrup containing over 90% glucose is used in industrial fermentation, but syrups used in confectionery contain varying amounts of glucose, maltose and higher oligosaccharides, depending on the grade, and can typically contain 10% to 43% glucose. Glucose syrup is used in foods to sweeten, soften texture and add volume. By converting some glucose in corn syrup into fructose (using an enzymatic process), a sweeter product, high fructose corn syrup can be produced. Glucose syrup was first made in 1811 in Russia by Gottlieb Kirchhoff using heat and sulfuric acid. Types Depending on the method used to hydrolyse the starch and on ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Corn Syrup
Corn syrup is a food syrup that is made from the starch of corn/maize and contains varying amounts of sugars: glucose, maltose and higher oligosaccharides, depending on the grade. Corn syrup is used in foods to soften Mouthfeel, texture, add volume, prevent crystallization of sugar, and enhance flavor. Most table syrups are typically based with corn syrup. It can be processed into high-fructose corn syrup (HFCS) by using the enzyme xylose isomerase, D-xylose isomerase to convert a large proportion of its glucose into sweeter fructose. The more general term glucose syrup is often used synonymously with corn syrup, since glucose syrup in the United States is most commonly made from corn starch. Technically, glucose syrup is any liquid starch hydrolysis, hydrolysate of mono-, di-, and higher-saccharides and can be made from any source of starch: wheat, tapioca and potatoes are the most common other sources. Commercial preparation Historically, corn syrup was produced by combining ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oligosaccharide
An oligosaccharide (; ) is a carbohydrate, saccharide polymer containing a small number (typically three to ten) of monosaccharides (simple sugars). Oligosaccharides can have many functions including Cell–cell recognition, cell recognition and cell adhesion. They are normally present as glycans: oligosaccharide chains are linked to lipids or to compatible amino acid side chains in proteins, by ''N''- or ''O''-glycoside, glycosidic bonds. ''N''-Linked oligosaccharides are always pentasaccharides attached to asparagine via a beta linkage to the amine nitrogen of the side chain.. Alternately, O-linked glycosylation, ''O''-linked oligosaccharides are generally attached to threonine or serine on the alcohol group of the side chain. Not all natural oligosaccharides occur as components of glycoproteins or glycolipids. Some, such as the raffinose series, occur as storage or transport carbohydrates in plants. Others, such as maltodextrins or cellodextrins, result from the microbial break ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Maltotriose
Maltotriose is a trisaccharide (three-part sugar) consisting of three glucose molecules linked with α-1,4 glycosidic bonds. It is most commonly produced by the digestive enzyme alpha-amylase (a common enzyme in human saliva) on amylose in starch. The creation of both maltotriose and maltose during this process is due to the random manner in which alpha amylase hydrolyses α-1,4 glycosidic bonds. It is the shortest chain oligosaccharide that can be classified as maltodextrin Maltodextrin is a name shared by two different families of chemicals. Both families are glucose polymers (also called ''dextrose polymers'' or ''Dextrin, dextrins''), but have little chemical or nutritional similarity. The digestible maltodextr .... References Trisaccharides Types of sugar {{Carbohydrate-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Enzymatic Hydrolysis
In biochemistry, enzymatic hydrolysis is a process in which enzymes facilitate the cleavage of bonds in molecules with the addition of the elements of water (i.e. hydrolysis). It plays an important role in the digestion of food. It may be used to help provide renewable energy, as with cellulosic ethanol.Ethanol from biomass by enzymatic hydrolysis - JD Wright - Chemical Engineering Progress, 1988 osti.gov/ref> See also * Acid hydrolysis *Alkaline hydrolysis Hydrolysis (; ) is any chemical reaction in which a molecule of water breaks one or more chemical bonds. The term is used broadly for substitution, elimination, and solvation reactions in which water is the nucleophile. Biological hydrolysis ... * Digestion enzyme References Digestive system {{enzyme-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydroxymethylfurfural
Hydroxymethylfurfural (HMF), also known as 5-(hydroxymethyl)furfural, is an organic compound formed by the dehydration of reducing sugars. It is a white low-melting solid (although commercial samples are often yellow) which is highly soluble in both water and organic solvents. The molecule consists of a furan ring, containing both aldehyde and alcohol functional groups. HMF can form in sugar-containing food, particularly as a result of heating or cooking. Its formation has been the topic of significant study as HMF was regarded as being potentially carcinogenic to humans. However, so far in vivo genotoxicity was negative. No relevance for humans concerning carcinogenic and genotoxic effects can be derived. HMF is classified as a food improvement agent and is primarily being used in the food industry in form of a food additive as a biomarker as well as a flavoring agent for food products. It is also produced industrially on a modest scale as a carbon-neutral feedstock for the pro ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bitter (taste)
The gustatory system or sense of taste is the sensory system that is partially responsible for the perception of taste. Taste is the perception stimulated when a substance in the mouth reacts chemically with taste receptor cells located on taste buds in the oral cavity, mostly on the tongue. Taste, along with the sense of smell and trigeminal nerve stimulation (registering texture, pain, and temperature), determines flavors of food and other substances. Humans have taste receptors on taste buds and other areas, including the upper surface of the tongue and the epiglottis. The gustatory cortex is responsible for the perception of taste. The tongue is covered with thousands of small bumps called papillae, which are visible to the naked eye. Within each papilla are hundreds of taste buds. The exceptions to this is the filiform papillae that do not contain taste buds. There are between 2000 and 5000Boron, W.F., E.L. Boulpaep. 2003. Medical Physiology. 1st ed. Elsevier ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
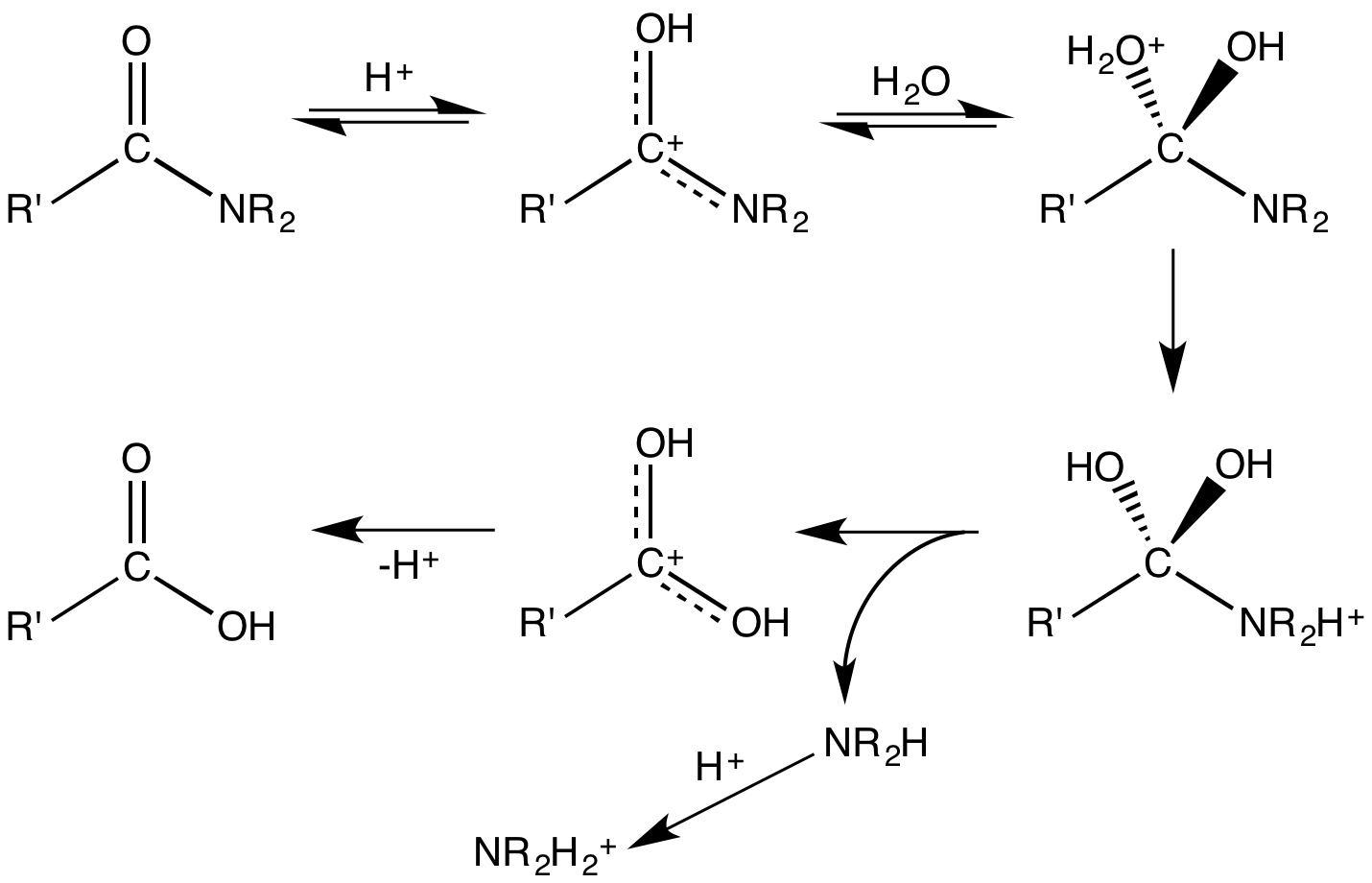
Acid Hydrolysis
Hydrolysis (; ) is any chemical reaction in which a molecule of water breaks one or more chemical bonds. The term is used broadly for substitution, elimination, and solvation reactions in which water is the nucleophile. Biological hydrolysis is the cleavage of biomolecules where a water molecule is consumed to effect the separation of a larger molecule into component parts. When a carbohydrate is broken into its component sugar molecules by hydrolysis (e.g., sucrose being broken down into glucose and fructose), this is recognized as saccharification. Hydrolysis reactions can be the reverse of a condensation reaction in which two molecules join into a larger one and eject a water molecule. Thus hydrolysis adds water to break down, whereas condensation builds up by removing water. Types Usually hydrolysis is a chemical process in which a molecule of water is added to a substance. Sometimes this addition causes both the substance and water molecule to split into two parts. In ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Reducing Sugar
A reducing sugar is any sugar that is capable of acting as a reducing agent. In an alkaline solution, a reducing sugar forms some aldehyde or ketone, which allows it to act as a reducing agent, for example in Benedict's reagent. In such a reaction, the sugar becomes a carboxylic acid. All monosaccharides are reducing sugars, along with some disaccharides, some oligosaccharides, and some polysaccharides. The monosaccharides can be divided into two groups: the aldoses, which have an aldehyde group, and the ketoses, which have a ketone group. Ketoses must first tautomerize to aldoses before they can act as reducing sugars. The common dietary monosaccharides galactose, glucose and fructose are all reducing sugars. Disaccharides are formed from two monosaccharides and can be classified as either reducing or nonreducing. Nonreducing disaccharides like sucrose and trehalose have glycosidic bonds between their anomeric carbons and thus cannot convert to an open-chain form with an ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dextrose Equivalent
Dextrose equivalent (DE) is a measure of the amount of reducing sugars present in a sugar product, expressed as a percentage on a dry basis relative to dextrose. The dextrose equivalent gives an indication of the average degree of polymerisation (DP) for starch sugars. As a rule of thumb, DE × DP = 120. In all glucose polymers, from the native starch to glucose syrup, the molecular chain ends with a reducing sugar, containing a free aldehyde in its linear form. As the starch is hydrolysed, the molecules become shorter and more reducing sugars are present. Therefore, the dextrose equivalent describes the degree of conversion of starch to dextrose. The standard method of determining the dextrose equivalent is the Lane-Eynon titration, based on the reduction of copper(II) sulfate in an alkaline tartrate solution, an application of Fehling's test. Examples: *A maltodextrin with a DE of 10 would have 10% of the reducing power of dextrose which has a DE of 100. *Maltose, a disac ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrolysis
Hydrolysis (; ) is any chemical reaction in which a molecule of water breaks one or more chemical bonds. The term is used broadly for substitution reaction, substitution, elimination reaction, elimination, and solvation reactions in which water is the nucleophile. Biological hydrolysis is the cleavage of Biomolecule, biomolecules where a water molecule is consumed to effect the separation of a larger molecule into component parts. When a carbohydrate is broken into its component sugar molecules by hydrolysis (e.g., sucrose being broken down into glucose and fructose), this is recognized as saccharification. Hydrolysis reactions can be the reverse of a condensation reaction in which two molecules join into a larger one and eject a water molecule. Thus hydrolysis adds water to break down, whereas condensation builds up by removing water. Types Usually hydrolysis is a chemical process in which a molecule of water is added to a substance. Sometimes this addition causes both the su ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfuric Acid
Sulfuric acid (American spelling and the preferred IUPAC name) or sulphuric acid (English in the Commonwealth of Nations, Commonwealth spelling), known in antiquity as oil of vitriol, is a mineral acid composed of the elements sulfur, oxygen, and hydrogen, with the molecular formula . It is a colorless, odorless, and Viscosity, viscous liquid that is Miscibility, miscible with water. Pure sulfuric acid does not occur naturally due to its Dehydration reaction, strong affinity to water vapor; it is Hygroscopy, hygroscopic and readily absorbs water vapor from the Atmosphere of Earth, air. Concentrated sulfuric acid is a strong oxidant with powerful dehydrating properties, making it highly corrosive towards other materials, from rocks to metals. Phosphorus pentoxide is a notable exception in that it is not dehydrated by sulfuric acid but, to the contrary, dehydrates sulfuric acid to sulfur trioxide. Upon addition of sulfuric acid to water, a considerable amount of heat is releas ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gottlieb Kirchhoff
Gottlieb Sigismund Constantin Kirchhoff (, Transliteration of Russian, tr. Konstantin Sigizmundovich Kirhgof; 19 February 1764 – 14 February 1833) was a Russian chemist of German origin. In 1792–1802, Assistant Director and then Director of the Head Pharmacy at Saint Petersburg. Corresponding member (1807–1812) and since 1812 Full member of the Petersburg Academy of Sciences (Russia). In 1811, he became the first person to convert starch into a sugar (corn syrup), by heating it with sulfuric acid in acid-catalyzed reaction.See: * Scherer (1811"Kirchhof's entdeckte Zubereitung des Zuckers und Syrups aus Buchweizen"(Kirchhoff's discovery of a preparation of sugar and syrup from buckwheat), ''Bulletin des Neuesten und Wissenwürdigsten aus der Naturwissenschaft'' … (Bulletin of the most recent and most useful to know [news] from science … ), 9 : 262–263. * Kirchhoff (1811) ''Mémoires de l'Académie impériale des sciences de St. Pétersbourg'', 4 : 27.From page 27: ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |