|
Geodesic Polyarene
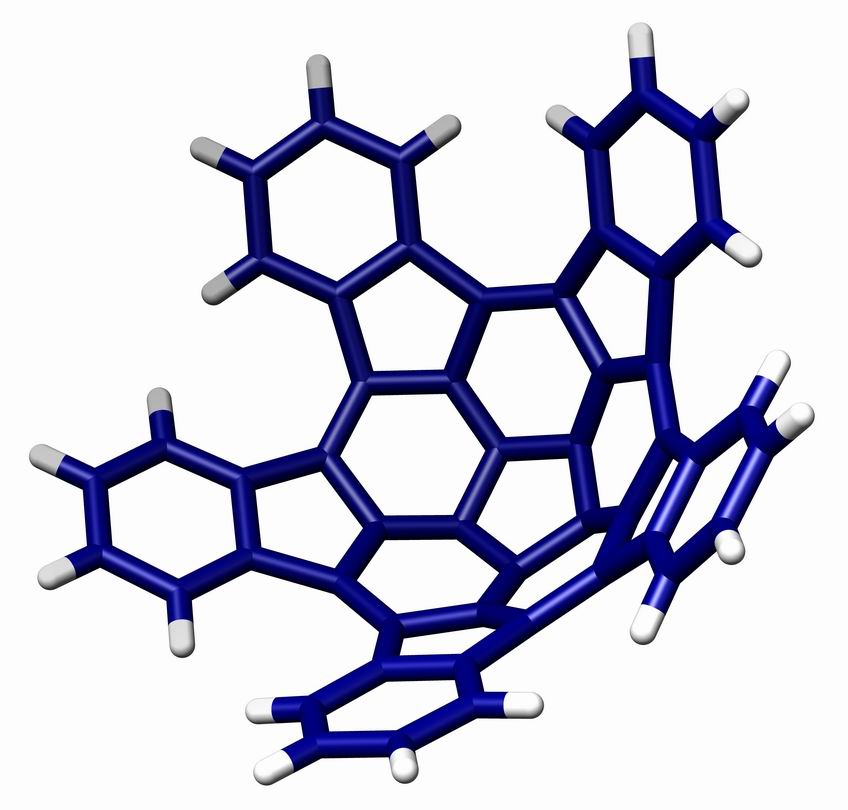
A geodesic polyarene in organic chemistry is a polycyclic aromatic hydrocarbon with curved convex or concave surfaces. Examples include fullerenes, nanotubes, corannulenes, helicenes and sumanene. The molecular orbitals of the carbon atoms in these systems are to some extent pyramidalized resulting a different pi electron density on either side of the molecule with consequences for reactivity. One member of this group of organic compounds, ''pentaindenocorannulene'' (depicted below), can be considered a large fullerene fragment. The experimentally obtained curvature and degree of pyramidalizion (12.6° for the carbons of the pentagon at the center ) are both actually larger than that of fullerene but according to its discoverers, the compound is relatively easy to synthesize starting from corannulene and a way is opened to produce larger such fragments by stitching. The crystal structure of pentaindenocorannulene has been obtained. An illustration of the crystal packing for p ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tris(dibenzylideneacetone)dipalladium(0)
Tris(dibenzylideneacetone)dipalladium(0) or d2(dba)3is an organopalladium compound. The compound is a complex of palladium(0) with dibenzylideneacetone (dba). It is a dark-purple/brown solid, which is modestly soluble in organic solvents. Because the dba ligands are easily displaced, the complex is used as a homogeneous catalyst in organic synthesis. Preparation and structure First reported in 1970, it is prepared from dibenzylideneacetone and sodium tetrachloropalladate. Because it is commonly recrystallized from chloroform, the complex is often supplied as the adduct d2(dba)3·CHCl3Jiro Tsuji and Ian J. S. Fairlamb "Tris(dibenzylideneacetone)dipalladium–Chloroform" E-EROS, 2008. The purity of samples can be variable. In d2(dba)3 the pair of Pd atoms are separated by 320 pm but are tied together by dba units. The Pd(0) centres are bound to the alkene parts of the dba ligands. Applications d2(dba)3is used as a source of soluble Pd(0), in particular as a catalyst f ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Corannulene
Corannulene is a polycyclic aromatic hydrocarbon with chemical formula C20 H10. The molecule consists of a cyclopentane ring fused with 5 benzene rings, so another name for it is irculene. It is of scientific interest because it is a geodesic polyarene and can be considered a fragment of buckminsterfullerene. Due to this connection and also its bowl shape, corannulene is also known as a buckybowl. Buckybowls are fragments of buckyballs. Corannulene exhibits a bowl-to-bowl inversion with an inversion barrier of 10.2 kcal/ mol (42.7 kJ/mol) at −64 °C. Synthesis Several synthetic routes exist to corannulene. Flash vacuum pyrolysis techniques generally have lower chemical yields than solution-chemistry syntheses, but offer routes to more derivatives. Corannulane was first isolated in 1966 by multistep organic synthesis. In 1971, the synthesis and properties of corannulane were reported. A flash vacuum pyrolysis method followed in 1991. One synthesis based on solution ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fullerene Chemistry
Fullerene chemistry is a field of organic chemistry devoted to the chemical properties of fullerenes. Research in this field is driven by the need to functionalize fullerenes and tune their properties. For example, fullerene is notoriously insoluble and adding a suitable group can enhance solubility. By adding a polymerizable group, a fullerene polymer can be obtained. Functionalized fullerenes are divided into two classes: exohedral fullerenes with substituents outside the cage and endohedral fullerenes with trapped molecules inside the cage. This article covers the chemistry of these so-called "buckyballs," while the chemistry of carbon nanotubes is covered in carbon nanotube chemistry. Chemical properties of fullerenes Fullerene or C60 is soccer-ball-shaped or ''Ih'' with 12 pentagons and 20 hexagons. According to Euler's theorem these 12 pentagons are required for closure of the carbon network consisting of ''n'' hexagons and C60 is the first stable fullerene because it i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Synthesis Of C50H10
Synthesis or synthesize may refer to: Science Chemistry and biochemistry *Chemical synthesis, the execution of chemical reactions to form a more complex molecule from chemical precursors **Organic synthesis, the chemical synthesis of organic compounds ***Total synthesis, the complete organic synthesis of complex organic compounds, usually without the aid of biological processes ***Convergent synthesis or linear synthesis, a strategy to improve the efficiency of multi-step chemical syntheses **Dehydration synthesis, a chemical synthesis resulting in the loss of a water molecule *Biosynthesis, the creation of an organic compound in a living organism, usually aided by enzymes **Photosynthesis, a biochemical reaction using a carbon molecule to produce an organic molecule, using sunlight as a catalyst **Chemosynthesis, the synthesis of biological compounds into organic waste, using methane or an oxidized molecule as a catalyst **Amino acid synthesis, the synthesis of an amino acid ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Flash Vacuum Pyrolysis
Flash vacuum pyrolysis (FVP) is a technique in organic synthesis. It entails heating a precursor molecule intensely and briefly. Two key parameters are the temperature and duration (or residence time), which are adjusted to optimize yield, conversion, and avoidance of intractable products. Often the experiment entails volatilizing a precursor, which is drawn through a "hot zone" followed by rapid condensation. The apparatus typically is conducted under dynamic vacuum. The hot zone must impart heat to the gaseous molecules, so it is generally packed with solids to induce gas-solid collisions. The packing material is generally chemically inert, such as quartz. The precursor (i) volatilizes with gentle heating and under vacuum, (ii) the precursor fragments or rearranges in the hot zone, and finally (iii) the products are collected by rapid cooling. Rapid post-reaction cooling and the dilution inherent in gases both suppress bimolecular degradation pathways. Examples The technique ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbon Nanotube
A scanning tunneling microscopy image of a single-walled carbon nanotube Rotating single-walled zigzag carbon nanotube A carbon nanotube (CNT) is a tube made of carbon with diameters typically measured in nanometers. ''Single-wall carbon nanotubes'' (''SWCNTs'') are one of the allotropes of carbon, intermediate between fullerene cages and flat graphene, with diameters in the range of a nanometre. Although not made this way, single-wall carbon nanotubes can be idealized as cutouts from a two-dimensional Hexagonal tiling, hexagonal lattice of carbon atoms rolled up along one of the Bravais lattice vectors of the hexagonal lattice to form a hollow cylinder. In this construction, periodic boundary conditions are imposed over the length of this roll-up vector to yield a helical lattice of seamlessly bonded carbon atoms on the cylinder surface. ''Multi-wall carbon nanotubes'' (''MWCNTs'') consisting of nested single-wall carbon nanotubes weakly bound together by van der Waals ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Synthesis Of Pentaindenocorannulene
Synthesis or synthesize may refer to: Science Chemistry and biochemistry *Chemical synthesis, the execution of chemical reactions to form a more complex molecule from chemical precursors **Organic synthesis, the chemical synthesis of organic compounds ***Total synthesis, the complete organic synthesis of complex organic compounds, usually without the aid of biological processes ***Convergent synthesis or linear synthesis, a strategy to improve the efficiency of multi-step chemical syntheses **Dehydration synthesis, a chemical synthesis resulting in the loss of a water molecule *Biosynthesis, the creation of an organic compound in a living organism, usually aided by enzymes **Photosynthesis, a biochemical reaction using a carbon molecule to produce an organic molecule, using sunlight as a catalyst **Chemosynthesis, the synthesis of biological compounds into organic waste, using methane or an oxidized molecule as a catalyst **Amino acid synthesis, the synthesis of an amino acid ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dimethylacetamide
Dimethylacetamide (DMAc or DMA) is the organic compound with the formula CH3C(O)N(CH3)2. This colorless, water-miscible, high-boiling liquid is commonly used as a polar solvent in organic synthesis. DMA is miscible with most other solvents, although it is poorly soluble in aliphatic hydrocarbons. Synthesis and production DMA is prepared commercially by the reaction of dimethylamine with acetic anhydride or acetic acid. Dehydration of the salt of dimethylamine and acetic acid also furnishes this compound: : CH3CO2H·HN(CH3)2 → H2O + CH3CON(CH3)2 Dimethylacetamide can also be produced by the reaction of dimethylamine with methyl acetate. : The separation and purification of the product is carried out by multistage distillation in rectification columns. DMA is obtained with essentially quantitive (99%) yield referred to methyl acetate. Reactions and applications The chemical reactions of dimethylacetamide are typical of ''N'',''N''-disubstituted amides. Hydrolysis of the acyl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
DBU (chemistry)
1,8-Diazabicyclo .4.0ndec-7-ene, or more commonly DBU, is a chemical compound and belongs to the class of amidine compounds. It is used in organic synthesis as a catalyst, a complexing ligand, and a non-nucleophilic base. Occurrence Although all commercially available DBU is produced synthetically, it may also be isolated from the sea sponge '' Niphates digitalis''. The biosynthesis of DBU has been proposed to begin with 1,6-hexanedial and 1,3-diaminopropane. Uses As a reagent in organic chemistry, DBU is used as a catalyst, a complexing ligand, and a non-nucleophilic base. It is also used as a curing agent for epoxy resins. It is used in the separation of fullerenes in conjunction with trimethylbenzene. It reacts with C70 and higher fullerenes, but not with to C60 It is also used as a catalyst in the production of polyurethanes. It also exhibited its dual character (base and nucleophile) in the synthesis of aryl- and styryl-terminal acetylenes. See also * 1,5-Diazabic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Heck Reaction
The Heck reaction (also called the Mizoroki–Heck reaction) is the chemical reaction of an unsaturated halide (or triflate) with an alkene in the presence of a base and a palladium catalyst (or palladium nanomaterial-based catalyst) to form a substituted alkene. It is named after Tsutomu Mizoroki and Richard F. Heck. Heck was awarded the 2010 Nobel Prize in Chemistry, which he shared with Ei-ichi Negishi and Akira Suzuki, for the discovery and development of this reaction. This reaction was the first example of a carbon-carbon bond-forming reaction that followed a Pd(0)/Pd(II) catalytic cycle, the same catalytic cycle that is seen in other Pd(0)-catalyzed cross-coupling reactions. The Heck reaction is a way to substitute alkenes. History The original reaction by Tsutomu Mizoroki (1971) describes the coupling between iodobenzene and styrene in methanol to form stilbene at 120 °C (autoclave) with potassium acetate base and palladium chloride catalysis. This work was an ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
dipalladium(0).jpg)

