|
GLRX5
Glutaredoxin 5, also known as GLRX5, is a protein which in humans is encoded by the ''GLRX5'' gene located on chromosome 14. This gene encodes a mitochondrial protein, which is evolutionarily conserved. It is involved in the biogenesis of iron- sulfur clusters, which are required for normal iron homeostasis. Mutations in this gene are associated with autosomal recessive pyridoxine-refractory sideroblastic anemia. Structure The GLRX5 gene contains 2 exons and encodes for a protein that is 13 kDa in size. The protein is highly expressed in erythroid cells. Crystal structure of the GLRX5 protein reveals that the protein likely exists as a tetramer with two Fe-S clusters buried in the interior. Function GLRX5 is a mitochondrial protein is conserved evolutionarily and plays a role in the formation of iron-sulfur clusters, which function to maintain iron homeostasis within the mitochondria and in the cell. GLRX5 is required for the steps in haem synthesis that involves mitoc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sideroblastic Anemia
Sideroblastic anemia, or sideroachrestic anemia, is a form of anemia in which the bone marrow produces ringed sideroblasts rather than healthy red blood cells (erythrocytes). In sideroblastic anemia, the body has iron available but cannot incorporate it into hemoglobin, which red blood cells need in order to transport oxygen efficiently. The disorder may be caused either by a genetic disorder or indirectly as part of myelodysplastic syndrome, which can develop into hematological malignancies (especially acute myeloid leukemia). Sideroblasts ('' sidero-'' + '' -blast'') are nucleated erythroblasts (precursors to mature red blood cells) with granules of iron accumulated in the mitochondria surrounding the nucleus. Normally, sideroblasts are present in the bone marrow, and enter the circulation after maturing into a normal erythrocyte. The presence of sideroblasts ''per se'' does not define sideroblastic anemia. Only the finding of ring (or ringed) sideroblasts characterizes sideroblas ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glutaredoxin
Glutaredoxins (also known as Thioltransferase) are small redox enzymes of approximately one hundred amino-acid residues that use glutathione as a cofactor. In humans this oxidation repair enzyme is also known to participate in many cellular functions, including redox signaling and regulation of glucose metabolism. Glutaredoxins are oxidized by substrates, and reduced non-enzymatically by glutathione. In contrast to thioredoxins, which are reduced by thioredoxin reductase, no oxidoreductase exists that specifically reduces glutaredoxins. Instead, glutaredoxins are reduced by the oxidation of glutathione. Reduced glutathione is then regenerated by glutathione reductase. Together these components compose the glutathione system. Like thioredoxin, which functions in a similar way, glutaredoxin possesses an active centre disulfide bond. It exists in either a reduced or an oxidized form where the two cysteine residues are linked in an intramolecular disulfide bond. Glutaredoxins function ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Protein
Proteins are large biomolecules and macromolecules that comprise one or more long chains of amino acid residues. Proteins perform a vast array of functions within organisms, including catalysing metabolic reactions, DNA replication, responding to stimuli, providing structure to cells and organisms, and transporting molecules from one location to another. Proteins differ from one another primarily in their sequence of amino acids, which is dictated by the nucleotide sequence of their genes, and which usually results in protein folding into a specific 3D structure that determines its activity. A linear chain of amino acid residues is called a polypeptide. A protein contains at least one long polypeptide. Short polypeptides, containing less than 20–30 residues, are rarely considered to be proteins and are commonly called peptides. The individual amino acid residues are bonded together by peptide bonds and adjacent amino acid residues. The sequence of amino acid residue ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mitochondrial
A mitochondrion (; ) is an organelle found in the cells of most Eukaryotes, such as animals, plants and fungi. Mitochondria have a double membrane structure and use aerobic respiration to generate adenosine triphosphate (ATP), which is used throughout the cell as a source of chemical energy. They were discovered by Albert von Kölliker in 1857 in the voluntary muscles of insects. The term ''mitochondrion'' was coined by Carl Benda in 1898. The mitochondrion is popularly nicknamed the "powerhouse of the cell", a phrase coined by Philip Siekevitz in a 1957 article of the same name. Some cells in some multicellular organisms lack mitochondria (for example, mature mammalian red blood cells). A large number of unicellular organisms, such as microsporidia, parabasalids and diplomonads, have reduced or transformed their mitochondria into other structures. One eukaryote, ''Monocercomonoides'', is known to have completely lost its mitochondria, and one multicellular organism, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glycine Encephalopathy
Glycine encephalopathy is a rare autosomal recessive disorder of glycine metabolism. After phenylketonuria, glycine encephalopathy is the second most common disorder of amino acid metabolism. The disease is caused by defects in the glycine cleavage system, an enzyme responsible for glycine catabolism. There are several forms of the disease, with varying severity of symptoms and time of onset. The symptoms are exclusively neurological in nature, and clinically this disorder is characterized by abnormally high levels of the amino acid glycine in bodily fluids and tissues, especially the cerebrospinal fluid. Glycine encephalopathy is sometimes referred to as "nonketotic hyperglycinemia" (NKH), as a reference to the biochemical findings seen in patients with the disorder, and to distinguish it from the disorders that cause "ketotic hyperglycinemia" (seen in propionic acidemia and several other inherited metabolic disorders). To avoid confusion, the term "glycine encephalopathy" is oft ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
ACO1
Aconitase 1, soluble is a protein that in humans is encoded by the ACO1 gene. Function The protein encoded by this gene is a bifunctional, cytosolic protein that functions as an essential enzyme in the TCA cycle and interacts with mRNA to control the levels of iron inside cells. When cellular iron levels are high, this protein binds to a 4Fe-4S cluster and functions as an aconitase Aconitase (aconitate hydratase; ) is an enzyme that catalyses the stereo-specific isomerization of citrate to isocitrate via ''cis''- aconitate in the tricarboxylic acid cycle, a non-redox-active process. Image:Citrate wpmp.png, Image:Cis- .... Aconitases are iron-sulfur proteins that function to catalyze the conversion of citrate to isocitrate. When cellular iron levels are low, the protein binds to iron-responsive elements (IREs), which are stem-loop structures found in the 5' UTR of ferritin mRNA, and in the 3' UTR of transferrin receptor mRNA. When the protein binds to IRE, it results in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hematopoiesis
Haematopoiesis (, from Greek , 'blood' and 'to make'; also hematopoiesis in American English; sometimes also h(a)emopoiesis) is the formation of blood cellular components. All cellular blood components are derived from haematopoietic stem cells. In a healthy adult person, approximately – new blood cells are produced daily in order to maintain steady state levels in the peripheral circulation.Semester 4 medical lectures at Uppsala University 2008 by Leif Jansson Process Haematopoietic stem cells (HSCs) Haematopoietic stem cells (HSCs) reside in the medulla of the bone (bone marrow) and have the unique ability to give rise to all of the different mature blood cell types and tissues. HSCs are self-renewing cells: when they differentiate, at least some of their daughter cells remain as HSCs so the pool of stem cells is not depleted. This phenomenon is called asymmetric division. The other daughters of HSCs ( myeloid and lymphoid progenitor cells) can follow any of the other ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Heme
Heme, or haem (pronounced / hi:m/ ), is a precursor to hemoglobin, which is necessary to bind oxygen in the bloodstream. Heme is biosynthesized in both the bone marrow and the liver. In biochemical terms, heme is a coordination complex "consisting of an iron ion coordinated to a porphyrin acting as a tetradentate ligand, and to one or two axial ligands." The definition is loose, and many depictions omit the axial ligands. Among the metalloporphyrins deployed by metalloproteins as prosthetic groups, heme is one of the most widely used and defines a family of proteins known as hemoproteins. Hemes are most commonly recognized as components of hemoglobin, the red pigment in blood, but are also found in a number of other biologically important hemoproteins such as myoglobin, cytochromes, catalases, heme peroxidase, and endothelial nitric oxide synthase. The word ''haem'' is derived from Greek ''haima'' meaning "blood". Function Hemoproteins have diverse biological functions incl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
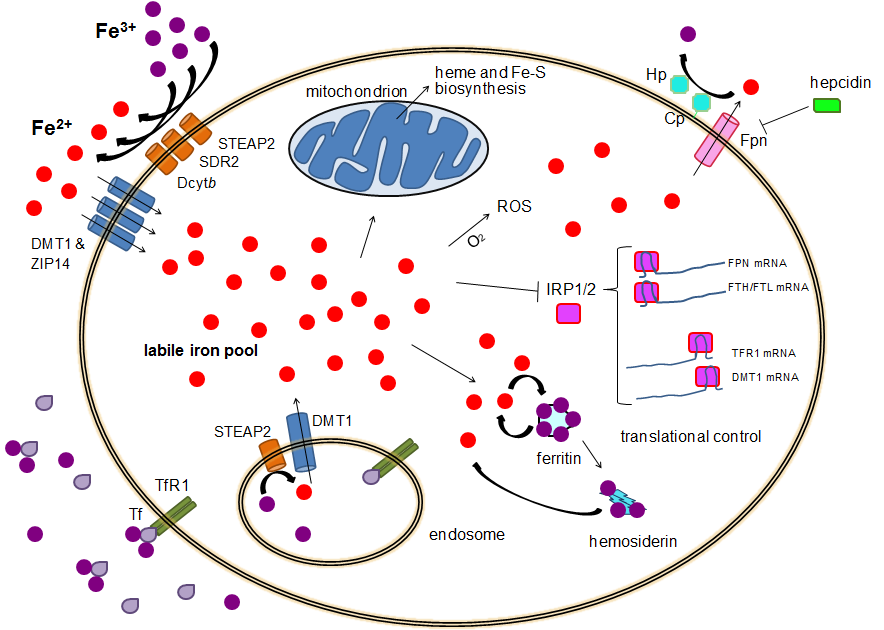
Iron Homeostasis
Human iron metabolism is the set of chemical reactions that maintain human homeostasis of iron at the systemic and cellular level. Iron is both necessary to the body and potentially toxic. Controlling iron levels in the body is a critically important part of many aspects of human health and disease. Hematologists have been especially interested in systemic iron metabolism because iron is essential for red blood cells, where most of the human body's iron is contained. Understanding iron metabolism is also important for understanding diseases of iron overload, such as hereditary hemochromatosis, and iron deficiency, such as iron-deficiency anemia. Importance of iron regulation Iron is an essential bioelement for most forms of life, from bacteria to mammals. Its importance lies in its ability to mediate electron transfer. In the ferrous state (Fe2+), iron acts as an electron donor, while in the ferric state (Fe3+) it acts as an acceptor. Thus, iron plays a vital role in the cataly ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iron-sulfur Clusters
Iron–sulfur proteins (or iron–sulphur proteins in British spelling) are proteins characterized by the presence of iron–sulfur clusters containing sulfide-linked di-, tri-, and tetrairon centers in variable oxidation states. Iron–sulfur clusters are found in a variety of metalloproteins, such as the ferredoxins, as well as NADH dehydrogenase, hydrogenases, coenzyme Q – cytochrome c reductase, succinate – coenzyme Q reductase and nitrogenase. Iron–sulfur clusters are best known for their role in the oxidation-reduction reactions of electron transport in mitochondria and chloroplasts. Both Complex I and Complex II of oxidative phosphorylation have multiple Fe–S clusters. They have many other functions including catalysis as illustrated by aconitase, generation of radicals as illustrated by SAM-dependent enzymes, and as sulfur donors in the biosynthesis of lipoic acid and biotin. Additionally, some Fe–S proteins regulate gene expression. Fe–S proteins are vulnerabl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Red Blood Cell
Red blood cells (RBCs), also referred to as red cells, red blood corpuscles (in humans or other animals not having nucleus in red blood cells), haematids, erythroid cells or erythrocytes (from Greek ''erythros'' for "red" and ''kytos'' for "hollow vessel", with ''-cyte'' translated as "cell" in modern usage), are the most common type of blood cell and the vertebrate's principal means of delivering oxygen (O2) to the body tissues—via blood flow through the circulatory system. RBCs take up oxygen in the lungs, or in fish the gills, and release it into tissues while squeezing through the body's capillaries. The cytoplasm of a red blood cell is rich in hemoglobin, an iron-containing biomolecule that can bind oxygen and is responsible for the red color of the cells and the blood. Each human red blood cell contains approximately 270 million hemoglobin molecules. The cell membrane is composed of proteins and lipids, and this structure provides properties essential for physiolo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Crystal Structure
In crystallography, crystal structure is a description of the ordered arrangement of atoms, ions or molecules in a crystal, crystalline material. Ordered structures occur from the intrinsic nature of the constituent particles to form symmetric patterns that repeat along the principal directions of Three-dimensional space (mathematics), three-dimensional space in matter. The smallest group of particles in the material that constitutes this repeating pattern is the unit cell of the structure. The unit cell completely reflects the symmetry and structure of the entire crystal, which is built up by repetitive Translation (geometry), translation of the unit cell along its principal axes. The translation vectors define the nodes of the Bravais lattice. The lengths of the principal axes, or edges, of the unit cell and the angles between them are the lattice constants, also called ''lattice parameters'' or ''cell parameters''. The symmetry properties of the crystal are described by the con ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |