|
Fructose Bisphosphatase Deficiency
In fructose bisphosphatase deficiency, there is not enough fructose bisphosphatase for gluconeogenesis to occur correctly. Glycolysis (the breakdown of glucose) will still work, as it does not use this enzyme. History Early research into the disorder was conducted by a team led by Anthony S. Pagliara and Barbara Illingworth Brown at Washington University Medical Center, based on the case of an infant girl from Oak Ridge, Missouri. Presentation Without effective gluconeogenesis (GNG), hypoglycaemia will set in after about 12 hours of fasting. This is the time when liver glycogen stores have been exhausted, and the body has to rely on GNG. When given a dose of glucagon (which would normally increase blood glucose) nothing will happen, as stores are depleted and GNG doesn't work. (In fact, the patient would already have high glucagon levels.) There is no problem with the metabolism of glucose or galactose, but fructose and glycerol cannot be used by the liver to maintain blood gluc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fructose 1,6-bisphosphate
Fructose 1,6-bisphosphate, also known as Harden-Young ester, is fructose sugar phosphorylated on carbons 1 and 6 (i.e., is a fructosephosphate). The β-D-form of this compound is common in cells. Upon entering the cell, most glucose and fructose is converted to fructose 1,6-bisphosphate. In glycolysis Fructose 1,6-bisphosphate lies within the glycolysis metabolic pathway and is produced by phosphorylation of fructose 6-phosphate. It is, in turn, broken down into two compounds: glyceraldehyde 3-phosphate and dihydroxyacetone phosphate. It is an allosteric activator of pyruvate kinase through distinct interactions of binding and allostery at the enzyme's catalytic site ''The numbering of the carbon atoms indicates the fate of the carbons according to their position in fructose 6-phosphate.'' Isomerism Fructose 1,6-bisphosphate has only one biologically active isomer, the β-D-form. There are many other isomers, analogous to those of fructose. Iron chelation Fructos ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fatty Acid
In chemistry, particularly in biochemistry, a fatty acid is a carboxylic acid with an aliphatic chain, which is either saturated or unsaturated. Most naturally occurring fatty acids have an unbranched chain of an even number of carbon atoms, from 4 to 28. Fatty acids are a major component of the lipids (up to 70% by weight) in some species such as microalgae but in some other organisms are not found in their standalone form, but instead exist as three main classes of esters: triglycerides, phospholipids, and cholesteryl esters. In any of these forms, fatty acids are both important dietary sources of fuel for animals and important structural components for cells. History The concept of fatty acid (''acide gras'') was introduced in 1813 by Michel Eugène Chevreul, though he initially used some variant terms: ''graisse acide'' and ''acide huileux'' ("acid fat" and "oily acid"). Types of fatty acids Fatty acids are classified in many ways: by length, by saturation vs unsaturati ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
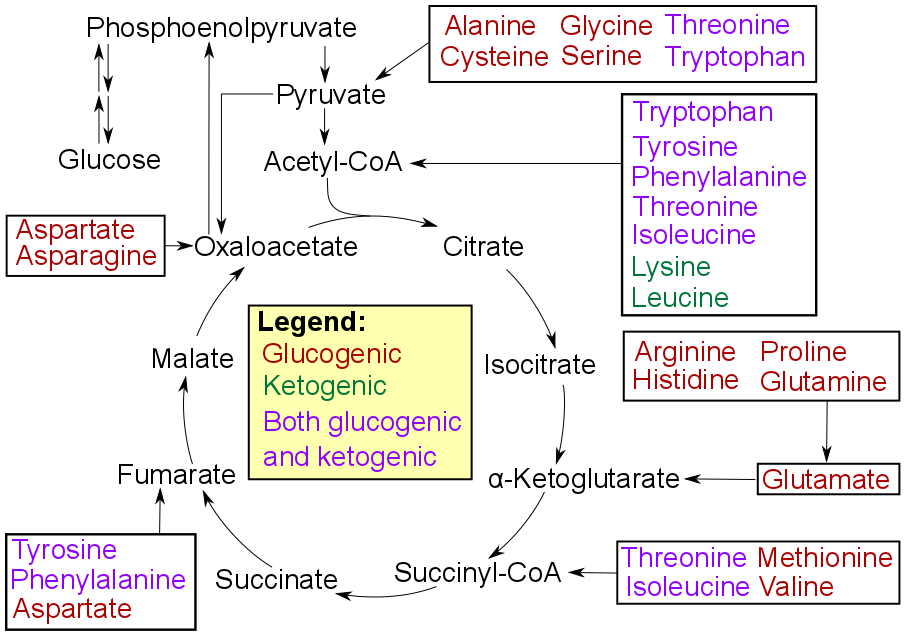
Gluconeogenesis
Gluconeogenesis (GNG) is a metabolic pathway that results in the generation of glucose from certain non-carbohydrate carbon substrates. It is a ubiquitous process, present in plants, animals, fungi, bacteria, and other microorganisms. In vertebrates, gluconeogenesis occurs mainly in the liver and, to a lesser extent, in the cortex of the kidneys. It is one of two primary mechanisms – the other being degradation of glycogen ( glycogenolysis) – used by humans and many other animals to maintain blood sugar levels, avoiding low levels (hypoglycemia). In ruminants, because dietary carbohydrates tend to be metabolized by rumen organisms, gluconeogenesis occurs regardless of fasting, low-carbohydrate diets, exercise, etc. In many other animals, the process occurs during periods of fasting, starvation, low-carbohydrate diets, or intense exercise. In humans, substrates for gluconeogenesis may come from any non-carbohydrate sources that can be converted to pyruvate or intermediates ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fructose
Fructose, or fruit sugar, is a Ketose, ketonic monosaccharide, simple sugar found in many plants, where it is often bonded to glucose to form the disaccharide sucrose. It is one of the three dietary monosaccharides, along with glucose and galactose, that are absorbed by the gut directly into the blood of the portal vein during digestion. The liver then converts both fructose and galactose into glucose, so that dissolved glucose, known as blood sugar, is the only monosaccharide present in circulating blood. Fructose was discovered by French chemist Augustin-Pierre Dubrunfaut in 1847. The name "fructose" was coined in 1857 by the English chemist William Allen Miller. Pure, dry fructose is a sweet, white, odorless, crystalline solid, and is the most water-soluble of all the sugars. Fructose is found in honey, tree and vine fruits, flowers, Berry, berries, and most List of root vegetables, root vegetables. Commercially, fructose is derived from sugar cane, sugar beets, and maize. Hi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sucrose
Sucrose, a disaccharide, is a sugar composed of glucose and fructose subunits. It is produced naturally in plants and is the main constituent of white sugar. It has the molecular formula . For human consumption, sucrose is extracted and refined from either sugarcane or sugar beet. Sugar mills – typically located in tropical regions near where sugarcane is grown – crush the cane and produce raw sugar which is shipped to other factories for refining into pure sucrose. Sugar beet factories are located in temperate climates where the beet is grown, and process the beets directly into refined sugar. The sugar-refining process involves washing the raw sugar crystals before dissolving them into a sugar syrup which is filtered and then passed over carbon to remove any residual colour. The sugar syrup is then concentrated by boiling under a vacuum and crystallized as the final purification process to produce crystals of pure sucrose that are clear, odorless, and sweet. Suga ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ketone Bodies
Ketone bodies are water-soluble molecules that contain the ketone groups produced from fatty acids by the liver (ketogenesis). Ketone bodies are readily transported into tissues outside the liver, where they are converted into acetyl-CoA (acetyl-Coenzyme A)—which then enters the citric acid cycle (Krebs cycle) and is oxidized for energy. These liver-derived ketone groups include acetoacetic acid (acetoacetate), beta-hydroxybutyrate, and acetone, a spontaneous breakdown product of acetoacetate (see graphic). Ketone bodies are produced by the liver during periods of caloric restriction of various scenarios: low food intake (fasting), carbohydrate restrictive diets, starvation, prolonged intense exercise, alcoholism, or during untreated (or inadequately treated) type 1 diabetes mellitus. Ketone bodies are produced in liver cells by the breakdown of fatty acids. They are released into the blood ''after'' glycogen stores in the liver have been depleted. (Glycogen stores typically ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acetyl CoA
Acetyl-CoA (acetyl coenzyme A) is a molecule that participates in many biochemical reactions in protein, carbohydrate and lipid metabolism. Its main function is to deliver the acetyl group to the citric acid cycle (Krebs cycle) to be oxidized for energy production. Coenzyme A (CoASH or CoA) consists of a β-mercaptoethylamine group linked to the vitamin pantothenic acid (B5) through an amide linkage and 3'-phosphorylated ADP. The acetyl group (indicated in blue in the structural diagram on the right) of acetyl-CoA is linked to the sulfhydryl substituent of the β-mercaptoethylamine group. This thioester linkage is a "high energy" bond, which is particularly reactive. Hydrolysis of the thioester bond is exergonic (−31.5 kJ/mol). CoA is acetylated to acetyl-CoA by the breakdown of carbohydrates through glycolysis and by the breakdown of fatty acids through β-oxidation. Acetyl-CoA then enters the citric acid cycle, where the acetyl group is oxidized to carbon dioxide and wa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Metabolic Acidosis
Metabolic acidosis is a serious electrolyte disorder characterized by an imbalance in the body's acid-base balance. Metabolic acidosis has three main root causes: increased acid production, loss of bicarbonate, and a reduced ability of the kidneys to excrete excess acids. Metabolic acidosis can lead to acidemia, which is defined as arterial blood pH that is lower than 7.35. Acidemia and acidosis are not mutually exclusive – pH and hydrogen ion concentrations also depend on the coexistence of other acid-base disorders; therefore, pH levels in people with metabolic acidosis can range from low to high. Acute metabolic acidosis, lasting from minutes to several days, often occurs during serious illnesses or hospitalizations, and is generally caused when the body produces an excess amount of organic acids (ketoacids in ketoacidosis, or lactic acid in lactic acidosis). A state of chronic metabolic acidosis, lasting several weeks to years, can be the result of impaired kidney function ( ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lactic Acid
Lactic acid is an organic acid. It has a molecular formula . It is white in the solid state and it is miscible with water. When in the dissolved state, it forms a colorless solution. Production includes both artificial synthesis as well as natural sources. Lactic acid is an alpha-hydroxy acid (AHA) due to the presence of a hydroxyl group adjacent to the carboxyl group. It is used as a synthetic intermediate in many organic synthesis industries and in various biochemical industries. The conjugate base of lactic acid is called lactate (or the lactate anion). The name of the derived acyl group is lactoyl. In solution, it can ionize by loss of a proton to produce the lactate ion . Compared to acetic acid, its p''K'' is 1 unit less, meaning lactic acid is ten times more acidic than acetic acid. This higher acidity is the consequence of the intramolecular hydrogen bonding between the α-hydroxyl and the carboxylate group. Lactic acid is chiral, consisting of two enantiomers. One ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pyruvate
Pyruvic acid (CH3COCOOH) is the simplest of the alpha-keto acids, with a carboxylic acid and a ketone functional group. Pyruvate, the conjugate base, CH3COCOO−, is an intermediate in several metabolic pathways throughout the cell. Pyruvic acid can be made from glucose through glycolysis, converted back to carbohydrates (such as glucose) via gluconeogenesis, or to fatty acids through a reaction with acetyl-CoA. It can also be used to construct the amino acid alanine and can be converted into ethanol or lactic acid via fermentation. Pyruvic acid supplies energy to cells through the citric acid cycle (also known as the Krebs cycle) when oxygen is present (aerobic respiration), and alternatively ferments to produce lactate when oxygen is lacking. Chemistry In 1834, Théophile-Jules Pelouze distilled tartaric acid and isolated glutaric acid and another unknown organic acid. Jöns Jacob Berzelius characterized this other acid the following year and named pyruvic acid because it ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fatty Liver
Fatty liver disease (FLD), also known as hepatic steatosis, is a condition where excess fat builds up in the liver. Often there are no or few symptoms. Occasionally there may be tiredness or pain in the upper right side of the abdomen. Complications may include cirrhosis, liver cancer, and esophageal varices. There are two types of fatty liver disease: non-alcoholic fatty liver disease (NAFLD) and alcoholic liver disease. NAFLD is made up of simple fatty liver and non-alcoholic steatohepatitis (NASH). The primary risks include alcohol, type 2 diabetes, and obesity. Other risk factors include certain medications such as glucocorticoids, and hepatitis C. It is unclear why some people with NAFLD develop simple fatty liver and others develop NASH. Diagnosis is based on the medical history supported by blood tests, medical imaging, and occasionally liver biopsy. Treatment of NAFLD is generally by dietary changes and exercise to bring about weight loss. In those who are severely affec ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Triacylglyceride
A triglyceride (TG, triacylglycerol, TAG, or triacylglyceride) is an ester derived from glycerol and three fatty acids (from ''tri-'' and ''glyceride''). Triglycerides are the main constituents of body fat in humans and other vertebrates, as well as vegetable fat. They are also present in the blood to enable the bidirectional transference of adipose fat and blood glucose from the liver, and are a major component of human skin oils. Many types of triglycerides exist. One specific classification focuses on saturated and unsaturated types. Saturated fats have ''no'' C=C groups; unsaturated fats feature one or more C=C groups. Unsaturated fats tend to have a lower melting point than saturated analogues; as a result, they are often liquid at room temperature. Chemical structure Triglycerides are tri-esters consisting of a glycerol bound to three fatty acid molecules. Alcohols have a hydroxyl (HO–) group. Organic acids have a carboxyl (–COOH) group. Alcohols and organic ac ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |