|
Fluoroform
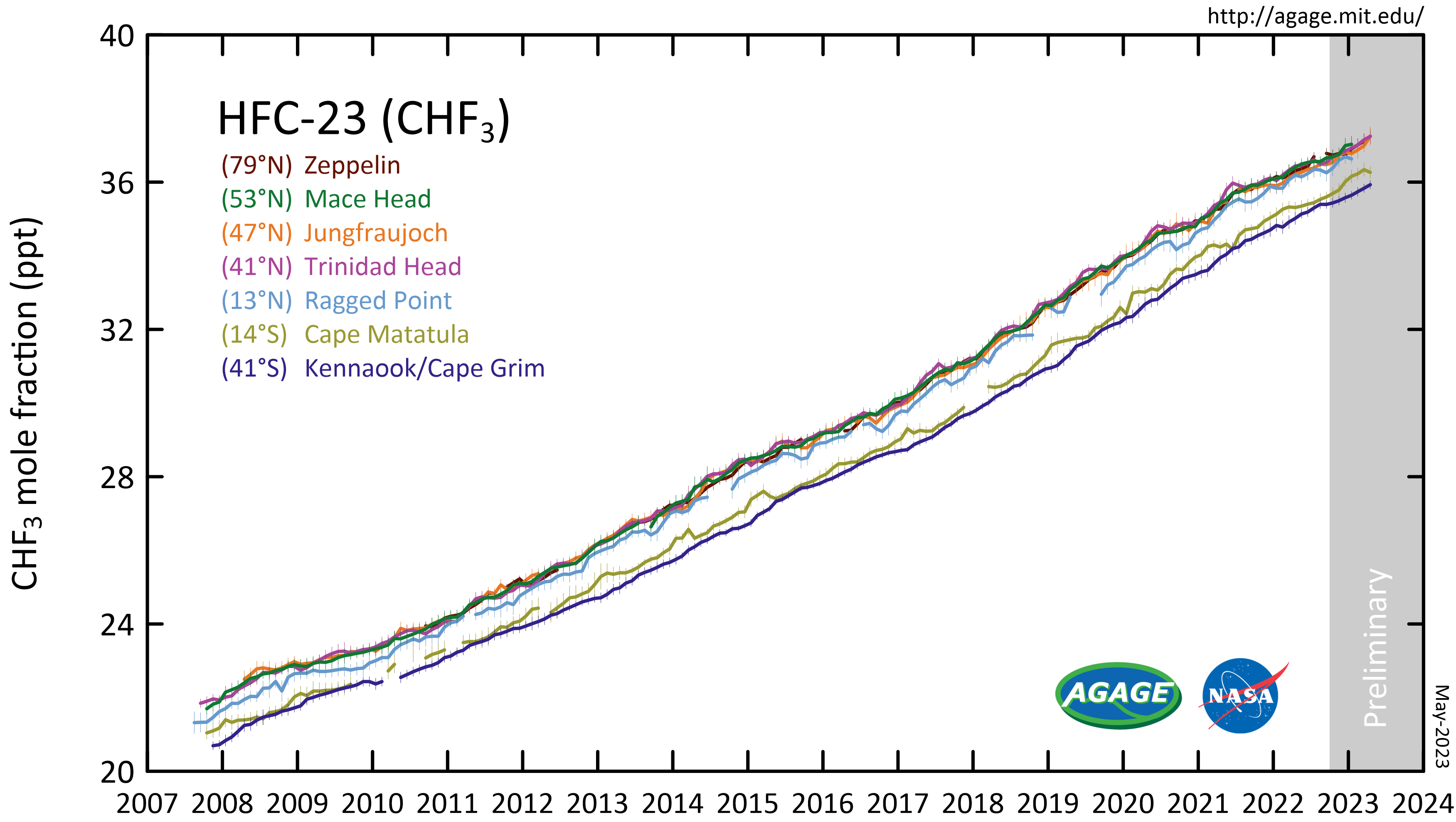
Trifluoromethane or fluoroform is the chemical compound with the formula CHF3. It is one of the " haloforms", a class of compounds with the formula CHX3 (X = halogen) with C3v symmetry. Fluoroform is used in diverse applications in organic synthesis. It is not an ozone depleter but is a greenhouse gas. Synthesis About 20M kg/y are produced industrially as both a by-product of and precursor to the manufacture of Teflon. It is produced by reaction of chloroform with HF: :CHCl3 + 3 HF → CHF3 + 3 HCl It is also generated biologically in small amounts apparently by decarboxylation of trifluoroacetic acid. Historical Fluoroform was first obtained by Maurice Meslans in the violent reaction of iodoform with dry silver fluoride in 1894. The reaction was improved by Otto Ruff by substitution of silver fluoride by a mixture of mercury fluoride and calcium fluoride. The exchange reaction works with iodoform and bromoform, and the exchange of the first two halogen atoms by ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
FE-13
Trifluoromethane or fluoroform is the chemical compound with the formula CHF3. It is one of the "trihalomethane, haloforms", a class of compounds with the formula CHX3 (X = halogen) with C3v symmetry group, symmetry. Fluoroform is used in diverse applications in organic synthesis. It is not an ozone depleter but is a greenhouse gas. Synthesis About 20M kg/y are produced industrially as both a by-product of and precursor to the manufacture of Teflon. It is produced by reaction of chloroform with Hydrogen_fluoride, HF: :CHCl3 + 3 HF → CHF3 + 3 HCl It is also generated biologically in small amounts apparently by decarboxylation of trifluoroacetic acid. Historical Fluoroform was first obtained by Maurice Meslans in the violent reaction of iodoform with dry silver fluoride in 1894. The reaction was improved by Otto Ruff by substitution of silver fluoride by a mixture of Mercury(II) fluoride, mercury fluoride and calcium fluoride. The exchange reaction works with iodofo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Trihalomethane
In chemistry, trihalomethanes (THMs) are chemical compounds in which three of the four hydrogen atoms of methane () are replaced by halogen atoms. Many trihalomethanes find uses in industry as solvents or refrigerants. THMs are also environmental pollutants, and many are considered carcinogenic. Trihalomethanes with all the same halogen atoms are called haloforms. Table of common trihalomethanes Industrial uses Only chloroform has significant applications of the haloforms. In the predominant application, chloroform is required for the production of tetrafluoroethylene, precursor to teflon. Chloroform is fluorinated by reaction with hydrogen fluoride to produce chlorodifluoromethane (R-22). Pyrolysis of chlorodifluoromethane (at 550-750 °C) yields TFE, with difluorocarbene as an intermediate. :CHCl3 + 2 HF -> CHClF2 + 2 HCl :2 CHClF2 -> C2F4 + 2 HCl Refrigerants and solvents Trifluoromethane and chlorodifluoromethane are both used as refrigerants. Trihalomethanes re ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bromoform
Bromoform (CHBr3) is a brominated organic solvent, colorless liquid at room temperature, with a high refractive index, very high density, and sweet odor is similar to that of chloroform. It is one of the four haloforms, the others being fluoroform, chloroform, and iodoform. Bromoform can be prepared by the haloform reaction using acetone and sodium hypobromite, by the electrolysis of potassium bromide in ethanol, or by treating chloroform with aluminium bromide. Currently its main use is as a laboratory reagent. Structure The molecule adopts tetrahedral molecular geometry with C3v symmetry. Uses Only small quantities of bromoform are currently produced industrially in the United States. In the past, it was used as a solvent, sedative and flame retardant, but now it is mainly used as a laboratory reagent, for example as an extraction solvent. Bromoform also has medical uses; injections of bromoform are sometimes used instead of epinephrine to treat severe asthma cases. Bromo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Halon 1301
Bromotrifluoromethane, commonly known as Halon 1301, R13B1, Halon 13B1 or BTM, is an organic halide with the chemical formula C Br F3. It is used for gaseous fire suppression as a far less toxic alternative to bromochloromethane. Table of physical properties Uses Halon 1301 was developed in a joint venture between the U.S. Army and DuPont in 1954, and introduced as an effective gaseous fire suppression fixed systems agent in the 1960s, and was used around valuable materials, such as aircraft, mainframe computers, and telecommunication switching centers, usually in total flooding systems . It was also widely used in the maritime industry to add a third level of protection should the main and emergency fire pumps become inoperable or ineffective. Halon 1301 was never widely used in portables outside military and spacecraft applications, due to its limited range, and invisible discharge. It does not produce the characteristic white cloud like CO2 and is difficult to direct ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
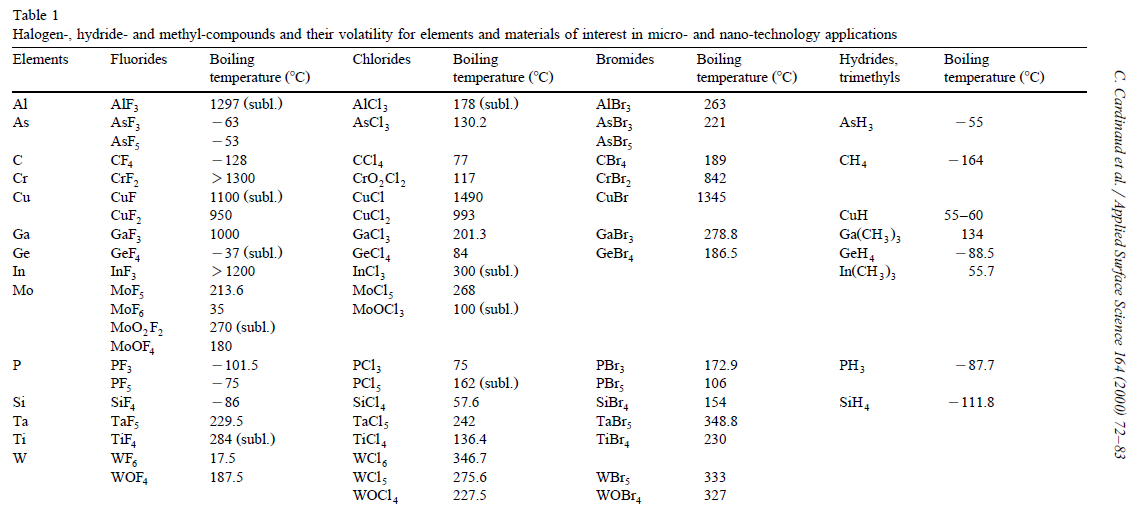
Halogen
The halogens () are a group in the periodic table consisting of five or six chemically related elements: fluorine (F), chlorine (Cl), bromine (Br), iodine (I), astatine (At), and tennessine (Ts). In the modern IUPAC nomenclature, this group is known as group 17. The word "halogen" means "salt former" (or "salt maker"). When halogens react with metals, they produce a wide range of salts, including calcium fluoride, sodium chloride (common table salt), silver bromide and potassium iodide. The group of halogens is the only periodic table group that contains elements in three of the main states of matter at standard temperature and pressure. All of the halogens form acids when bonded to hydrogen. Most halogens are typically produced from minerals or salts. The middle halogens—chlorine, bromine, and iodine—are often used as disinfectants. Organobromides are the most important class of flame retardants, while elemental halogens are dangerous and can be toxic. History The fl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Calcium Fluoride
Calcium fluoride is the inorganic compound of the elements calcium and fluorine with the formula CaF2. It is a white insoluble solid. It occurs as the mineral fluorite (also called fluorspar), which is often deeply coloured owing to impurities. Chemical structure The compound crystallizes in a cubic motif called the fluorite structure. Ca2+ centres are eight-coordinate, being centered in a cube of eight F− centres. Each F− centre is coordinated to four Ca2+ centres in the shape of a tetrahedron. Although perfectly packed crystalline samples are colorless, the mineral is often deeply colored due to the presence of F-centers. The same crystal structure is found in numerous ionic compounds with formula AB2, such as CeO2, cubic ZrO2, UO2, ThO2, and PuO2. In the corresponding anti-structure, called the antifluorite structure, anions and cations are swapped, such as Be2C. Gas phase The gas phase is noteworthy for failing the predictions of VSEPR theory; the molecule is no ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
DuPont
DuPont de Nemours, Inc., commonly shortened to DuPont, is an American multinational chemical company first formed in 1802 by French-American chemist and industrialist Éleuthère Irénée du Pont de Nemours. The company played a major role in the development of Delaware and first arose as a major supplier of gunpowder. DuPont developed many polymers such as Vespel, neoprene, nylon, Corian, Teflon, Mylar, Kapton, Kevlar, Zemdrain, M5 fiber, Nomex, Tyvek, Sorona, Corfam and Lycra in the 20th century, and its scientists developed many chemicals, most notably Freon (chlorofluorocarbons), for the refrigerant industry. It also developed synthetic pigments and paints including ChromaFlair. In 2015, DuPont and the Dow Chemical Company agreed to a reorganization plan in which the two companies would merge and split into three. As a merged entity, DuPont simultaneously acquired Dow and renamed itself to DowDuPont on August 31, 2017, and after 18 months spin off the merged entity' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chlorotrifluoromethane
Chlorotrifluoromethane, R-13, CFC-13, or Freon 13, is a non-flammable, non-corrosive chlorofluorocarbon (CFC) and also a mixed halomethane. It is a man-made substance used primarily as a refrigerant. When released into the environment, CFC-13 has a high ozone depletion potential, high global warming potential, and long atmospheric lifetime. Preparation It can be prepared by reacting carbon tetrachloride with hydrogen fluoride in the presence of a catalytic amount of antimony pentachloride: CCl4 + 3HF → CClF3 + 3HCl This reaction can also produce trichlorofluoromethane (CCl3F), dichlorodifluoromethane (CCl2F2) and tetrafluoromethane (CF4). Production phaseout Per the international Montreal Protocol, CFC-13 began a phase out and replacement with alternative substances starting in the early 1990s that will culminate in a global ban on its production. The atmospheric abundance of CFC-13 rose from 3.0 parts per trillion (ppt) in year 2010 to 3.3 ppt in year 2020 based ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Refrigerant
A refrigerant is a working fluid used in the heat pump and refrigeration cycle, refrigeration cycle of air conditioning systems and heat pumps where in most cases they undergo a repeated phase transition from a liquid to a gas and back again. Refrigerants are heavily regulated due to their toxicity, flammability and the contribution of CFC and HCFC refrigerants to ozone depletion and that of HFC refrigerants to climate change. Refrigerants are used in a Direct Expansion (DX) system to transfer energy from one environment to another, typically from inside a building to outside (or vice versa) commonly known as an "air conditioner" or "heat pump". Refrigerants can carry per kg 10 times more energy than water and 50 times more than air. Refrigerants are controlled substances due to 1) High Pressures (100-145 psi), 2) Extreme temperatures (-50°C to 145°C), 3) Flammability A1 class non-flammable, A2/A2L class flammable & A3 class extremely flammable/explosive and 4) Toxicity B1-low ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
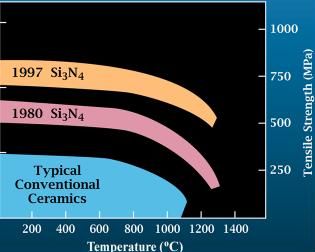
Silicon Nitride
Silicon nitride is a chemical compound of the elements silicon and nitrogen. is the most thermodynamically stable and commercially important of the silicon nitrides, and the term "silicon nitride" commonly refers to this specific composition. It is a white, high-melting-point solid that is relatively chemically inert, being attacked by dilute HF and hot . It is very hard (8.5 on the mohs scale). It has a high thermal stability with strong optical nonlinearities for all-optical applications. Production Silicon nitride is prepared by heating powdered silicon between 1300 °C and 1400 °C in a nitrogen atmosphere: :3 Si + 2 → The silicon sample weight increases progressively due to the chemical combination of silicon and nitrogen. Without an iron catalyst, the reaction is complete after several hours (~7), when no further weight increase due to nitrogen absorption (per gram of silicon) is detected. In addition to , several other silicon nitride phases (with chemical ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Silicon Oxide
Silicon oxide may refer to either of the following: *Silicon dioxide or quartz, SiO2, very well characterized *Silicon monoxide Silicon monoxide is the chemical compound with the formula SiO where silicon is present in the oxidation state +2. In the vapour phase, it is a diatomic molecule. It has been detected in stellar objects and has been described as the most common o ..., SiO, not very well characterized {{Short pages monitor ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Plasma Etching
Plasma etching is a form of plasma processing used to fabricate integrated circuits. It involves a high-speed stream of glow discharge (plasma) of an appropriate gas mixture being shot (in pulses) at a sample. The plasma source, known as etch species, can be either charged (ions) or neutral (atoms and radicals). During the process, the plasma generates volatile etch products at room temperature from the chemical reactions between the elements of the material etched and the reactive species generated by the plasma. Eventually the atoms of the shot element embed themselves at or just below the surface of the target, thus modifying the physical properties of the target. Mechanisms Plasma generation A plasma is a high energetic condition in which a lot of processes can occur. These processes happen because of electrons and atoms. To form the plasma electrons have to be accelerated to gain energy. Highly energetic electrons transfer the energy to atoms by collisions. Three different pr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |