|
Farnesylpyrophosphate
Farnesyl pyrophosphate (FPP), also known as farnesyl diphosphate (FDP), is an intermediate in the biosynthesis of terpenes and terpenoids such as Sterol, sterols and Carotenoid, carotenoids. It is also used in the synthesis of CoQ (part of the electron transport chain), as well as dehydrodolichol diphosphate (a precursor of dolichol, which transports proteins to the ER lumen for N-glycosylation, ''N''-glycosylation). Biosynthesis Farnesyl pyrophosphate synthase (a prenyl transferase) catalyzes sequential condensation reactions of dimethylallyl pyrophosphate with 2 units of 3-isopentenyl pyrophosphate to form farnesyl pyrophosphate, as is shown in the following two steps: * Dimethylallyl pyrophosphate reacts with 3-isopentenyl pyrophosphate to form geranyl pyrophosphate: * Geranyl pyrophosphate then reacts with another molecule of 3-isopentenyl pyrophosphate to form farnesyl pyrophosphate Pharmacology The above reactions are inhibited by bisphosphonates (used for osteoporosis). ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Terpene
Terpenes () are a class of natural products consisting of compounds with the formula (C5H8)n for n > 1. Comprising more than 30,000 compounds, these unsaturated hydrocarbons are produced predominantly by plants, particularly conifers. Terpenes are further classified by the number of carbons: monoterpenes (C10), sesquiterpenes (C15), diterpenes (C20), as examples. The terpene alpha-pinene, is a major component of the common solvent, turpentine. History and terminology The term ''terpene'' was coined in 1866 by the German chemist August Kekulé to denote all hydrocarbons having the empirical formula C10H16, of which camphene was one. Previously, many hydrocarbons having the empirical formula C10H16 had been called "camphene", but many other hydrocarbons of the same composition had had different names. Kekulé coined the term "terpene" in order to reduce the confusion. The name "terpene" is a shortened form of "terpentine", an obsolete spelling of "turpentine". Although sometimes ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Geranylgeranyl Pyrophosphate
Geranylgeranyl pyrophosphate is an intermediate in the biosynthesis of diterpenes and diterpenoids. It is also the precursor to carotenoids, gibberellins, tocopherols, and chlorophylls. It is also a precursor to geranylgeranylated proteins, which is its primary use in human cells. It is formed from farnesyl pyrophosphate by the addition of an isoprene unit from isopentenyl pyrophosphate. In ''Drosophila'', geranylgeranyl pyrophosphate is synthesised by HMG-CoA encoded by the Columbus gene. Geranylgeranyl pyrophosphate is utilised as a chemoattractant for migrating germ cells that have traversed the midgut epithelia. The attractant signal is produced at the gonadal precursors, directing the germ cells to these sites, where they will differentiate into eggs and spermatozoa (sperm). Related compounds * Farnesyl pyrophosphate * Geranylgeraniol * Geranyl pyrophosphate Geranyl pyrophosphate (GPP), also known as geranyl diphosphate (GDP), is the pyrophosphate ester of the terpenoi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Geranyl Pyrophosphate
Geranyl pyrophosphate (GPP), also known as geranyl diphosphate (GDP), is the pyrophosphate ester of the terpenoid geraniol. Its salts are colorless. It is a precursor to many natural products. Occurrence GPP is an intermediate in the isoprenoid biosynthesis pathway that produces longer prenyl chains such as farnesyl pyrophosphate and geranylgeranyl pyrophosphate as well as many terpenes. It can be prepared in the laboratory from geraniol. Related compounds * Geraniol * Farnesyl pyrophosphate * Geranylgeranyl pyrophosphate See also * Dimethylallyltranstransferase Dimethylallyltranstransferase (DMATT), also known as farnesylpyrophosphate synthase (FPPS) or as farnesyldiphosphate synthase (FDPS), is an enzyme that in humans is encoded by the FDPS gene and catalyzes the transformation of dimethylallylpyr ... References Further reading *Kulkarni RS, Pandit SS, Chidley HG, Nagel R, Schmidt A, Gershenzon J, Pujari KH, Giri AP and Gupta VS, 2013Characterization of three n ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Farnesol
Farnesol is a natural 15-carbon organic compound which is an acyclic sesquiterpene alcohol. Under standard conditions, it is a colorless liquid. It is hydrophobic, and thus insoluble in water, but miscible with oils. Farnesol is produced from 5-carbon isoprene compounds in both plants and animals. Phosphate-activated derivatives of farnesol are the building blocks of possibly all acyclic sesquiterpenoids. These compounds are doubled to form 30-carbon squalene, which is the precursor for steroids in plants, animals, and fungi. Farnesol and its derivatives are important starting compounds for natural and artificial organic synthesis. Uses Farnesol is present in many essential oils such as citronella, neroli, cyclamen, lemon grass, tuberose, rose, musk, balsam, and tolu. It is used in perfumery to emphasize the odors of sweet, floral perfumes. It enhances perfume scent by acting as a co-solvent that regulates the volatility of the odorants. It is especially used in lilac perfume ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Farnesene
The term farnesene refers to a set of six closely related chemical compounds which all are sesquiterpenes. α-Farnesene and β-farnesene are isomers, differing by the location of one double bond. α-Farnesene is 3,7,11-trimethyl-1,3,6,10-dodecatetraene and β-farnesene is 7,11-dimethyl-3-methylene-1,6,10-dodecatriene. The alpha form can exist as four stereoisomers that differ about the geometry of two of its three internal double bonds (the stereoisomers of the third internal double bond are identical). The beta isomer exists as two stereoisomers about the geometry of its central double bond. Two of the α-farnesene stereoisomers are reported to occur in nature. (''E'',''E'')-α-Farnesene is the most common isomer. It is found in the coating of apples, and other fruits, and it is responsible for the characteristic green apple odour. Its oxidation by air forms compounds that are damaging to the fruit. The oxidation products injure cell membranes which eventually causes cell dea ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
TRPV3
Transient receptor potential cation channel, subfamily V, member 3, also known as TRPV3, is a human gene encoding the protein of the same name. The TRPV3 protein belongs to a family of nonselective cation channels that function in a variety of processes, including temperature sensation and vasoregulation. The thermosensitive members of this family are expressed in subsets of human sensory neurons that terminate in the skin, and are activated at distinct physiological temperatures. This channel is activated at temperatures between 22 and 40 degrees C. The gene lies in close proximity to another family member (TRPV1) gene on chromosome 17, and the two encoded proteins are thought to associate with each other to form heteromeric channels. Function The TRPV3 channel has wide tissue expression that is especially high in the skin (keratinocytes) but also in the brain. It functions as a molecular sensor for innocuous warm temperatures. Mice lacking these protein are unable to sense ele ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
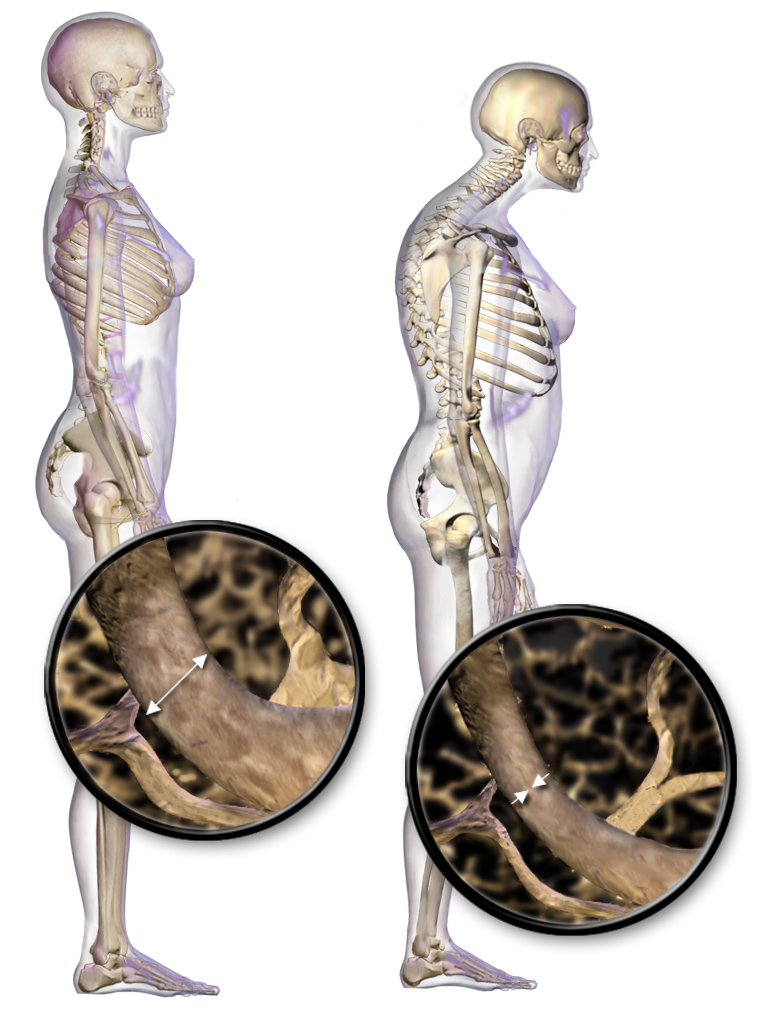
Osteoporosis
Osteoporosis is a systemic skeletal disorder characterized by low bone mass, micro-architectural deterioration of bone tissue leading to bone fragility, and consequent increase in fracture risk. It is the most common reason for a broken bone among the elderly. Bones that commonly break include the vertebrae in the spine, the bones of the forearm, and the hip. Until a broken bone occurs there are typically no symptoms. Bones may weaken to such a degree that a break may occur with minor stress or spontaneously. After the broken bone heals, the person may have chronic pain and a decreased ability to carry out normal activities. Osteoporosis may be due to lower-than-normal maximum bone mass and greater-than-normal bone loss. Bone loss increases after the menopause due to lower levels of estrogen, and after ' andropause' due to lower levels of testosterone. Osteoporosis may also occur due to a number of diseases or treatments, including alcoholism, anorexia, hyperthyroidism, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bisphosphonate
Bisphosphonates are a class of drugs that prevent the loss of bone density, used to treat osteoporosis and similar diseases. They are the most commonly prescribed drugs used to treat osteoporosis. They are called bisphosphonates because they have two phosphonate () groups. They are thus also called diphosphonates ('' bis-'' or '' di-'' + ''phosphonate''). Evidence shows that they reduce the risk of fracture in post-menopausal women with osteoporosis. Bone tissue undergoes constant remodeling and is kept in balance (homeostasis) by osteoblasts creating bone and osteoclasts destroying bone. Bisphosphonates inhibit the digestion of bone by encouraging osteoclasts to undergo apoptosis, or cell death, thereby slowing bone loss. The uses of bisphosphonates include the prevention and treatment of osteoporosis, Paget's disease of bone, bone metastasis (with or without hypercalcemia), multiple myeloma, primary hyperparathyroidism, osteogenesis imperfecta, fibrous dysplasia, and o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Terpenoid
The terpenoids, also known as isoprenoids, are a class of naturally occurring organic chemicals derived from the 5-carbon compound isoprene and its derivatives called terpenes, diterpenes, etc. While sometimes used interchangeably with "terpenes", terpenoids contain additional functional groups, usually containing oxygen. When combined with the hydrocarbon terpenes, terpenoids comprise about 80,000 compounds. They are the largest class of plant secondary metabolites, representing about 60% of known natural products. Many terpenoids have substantial pharmacological bioactivity and are therefore of interest to medicinal chemists. Plant terpenoids are used for their aromatic qualities and play a role in traditional herbal remedies. Terpenoids contribute to the scent of eucalyptus, the flavors of cinnamon, cloves, and ginger, the yellow color in sunflowers, and the red color in tomatoes. Well-known terpenoids include citral, menthol, camphor, salvinorin A in the plant '' Salvia di ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Geranyl Pyrophosphate
Geranyl pyrophosphate (GPP), also known as geranyl diphosphate (GDP), is the pyrophosphate ester of the terpenoid geraniol. Its salts are colorless. It is a precursor to many natural products. Occurrence GPP is an intermediate in the isoprenoid biosynthesis pathway that produces longer prenyl chains such as farnesyl pyrophosphate and geranylgeranyl pyrophosphate as well as many terpenes. It can be prepared in the laboratory from geraniol. Related compounds * Geraniol * Farnesyl pyrophosphate * Geranylgeranyl pyrophosphate See also * Dimethylallyltranstransferase Dimethylallyltranstransferase (DMATT), also known as farnesylpyrophosphate synthase (FPPS) or as farnesyldiphosphate synthase (FDPS), is an enzyme that in humans is encoded by the FDPS gene and catalyzes the transformation of dimethylallylpyr ... References Further reading *Kulkarni RS, Pandit SS, Chidley HG, Nagel R, Schmidt A, Gershenzon J, Pujari KH, Giri AP and Gupta VS, 2013Characterization of three n ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |