|
Electroacoustic Phenomena
Electroacoustic phenomena arise when ultrasound propagates through a fluid containing ions. The associated particle motion generates electric signals because ions have electric charge. This coupling between ultrasound and electric field is called electroacoustic phenomena. The fluid might be a simple Newtonian liquid, or complex heterogeneous dispersion, emulsion or even a porous body. There are several different electroacoustic effects depending on the nature of the fluid.Dukhin, A.S. and Goetz, P.J. ''Characterization of liquids, nano- and micro- particulates and porous bodies using Ultrasound'', Elsevier, 2017 *Ion vibration current (IVI) and potential, an electric signal that arises when an acoustic wave propagates through a homogeneous fluid. * Streaming vibration current (SVI) and potential, an electric signal that arises when an acoustic wave propagates through a porous body in which the pores are filled with fluid. *Colloid vibration current (CVI) and potential, an electr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ultrasound
Ultrasound is sound waves with frequency, frequencies higher than the upper audible limit of human hearing range, hearing. Ultrasound is not different from "normal" (audible) sound in its physical properties, except that humans cannot hear it. This limit varies from person to person and is approximately 20 Hertz, kilohertz (20,000 hertz) in healthy young adults. Ultrasound devices operate with frequencies from 20 kHz up to several gigahertz. Ultrasound is used in many different fields. Ultrasonic devices are used to detect objects and measure distances. Ultrasound imaging or sonography is often used in medicine. In the nondestructive testing of products and structures, ultrasound is used to detect invisible flaws. Industrially, ultrasound is used for cleaning, mixing, and accelerating chemical processes. Animals such as bats and porpoises use ultrasound for locating Predation, prey and obstacles. History Acoustics, the science of sound, starts as far back as Pyth ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dispersion Technology
Dispersion Technology Inc is a scientific instrument manufacturer located in Bedford Hills, New York. It was founded in 1996 by Philip Goetz (former Chairman, retired in 2010) and Dr. Andrei Dukhin (current CEO). The company develops and sells analytical instruments intended for characterizing concentrated dispersions and emulsions, complying with the International Standards for acoustic particle sizing ISO 20998 ISO 20998-1:2006 Measurement and characterization of particles by acoustic methods -- Part 1: Concepts and procedures in ultrasonic attenuation spectroscopy -- Part 2: Guideline ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Condensed Matter Physics
Condensed matter physics is the field of physics that deals with the macroscopic and microscopic physical properties of matter, especially the solid and liquid phases which arise from electromagnetic forces between atoms. More generally, the subject deals with "condensed" phases of matter: systems of many constituents with strong interactions between them. More exotic condensed phases include the superconducting phase exhibited by certain materials at low temperature, the ferromagnetic and antiferromagnetic phases of spins on crystal lattices of atoms, and the Bose–Einstein condensate found in ultracold atomic systems. Condensed matter physicists seek to understand the behavior of these phases by experiments to measure various material properties, and by applying the physical laws of quantum mechanics, electromagnetism, statistical mechanics, and other theories to develop mathematical models. The diversity of systems and phenomena available for study makes condensed matter phy ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Colloidal Chemistry
Interface and colloid science is an interdisciplinary intersection of branches of chemistry, physics, nanoscience and other fields dealing with colloids, heterogeneous systems consisting of a mechanical mixture of particles between 1 nm and 1000 nm dispersed in a continuous medium. A colloidal solution is a heterogeneous mixture in which the particle size of the substance is intermediate between a true solution and a suspension, i.e. between 1–1000 nm. Smoke from a fire is an example of a colloidal system in which tiny particles of solid float in air. Just like true solutions, colloidal particles are small and cannot be seen by the naked eye. They easily pass through filter paper. But colloidal particles are big enough to be blocked by parchment paper or animal membrane. Interface and colloid science has applications and ramifications in the chemical industry, pharmaceuticals, biotechnology, ceramics, minerals, nanotechnology, and microfluidics, among others. There ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Mixtures
A chemical substance is a form of matter having constant chemical composition and characteristic properties. Some references add that chemical substance cannot be separated into its constituent elements by physical separation methods, i.e., without breaking chemical bonds. Chemical substances can be simple substances (substances consisting of a single chemical element), chemical compounds, or alloys. Chemical substances are often called 'pure' to set them apart from mixtures. A common example of a chemical substance is pure water; it has the same properties and the same ratio of hydrogen to oxygen whether it is isolated from a river or made in a laboratory. Other chemical substances commonly encountered in pure form are diamond (carbon), gold, table salt ( sodium chloride) and refined sugar ( sucrose). However, in practice, no substance is entirely pure, and chemical purity is specified according to the intended use of the chemical. Chemical substances exist as solids, liqu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Interface And Colloid Science
Interface and colloid science is an interdisciplinary intersection of branches of chemistry, physics, nanoscience and other fields dealing with colloids, heterogeneous systems consisting of a mechanical mixture of particles between 1 nm and 1000 nm dispersed in a continuous medium. A colloidal solution is a heterogeneous mixture in which the particle size of the substance is intermediate between a true solution and a suspension, i.e. between 1–1000 nm. Smoke from a fire is an example of a colloidal system in which tiny particles of solid float in air. Just like true solutions, colloidal particles are small and cannot be seen by the naked eye. They easily pass through filter paper. But colloidal particles are big enough to be blocked by parchment paper or animal membrane. Interface and colloid science has applications and ramifications in the chemical industry, pharmaceuticals, biotechnology, ceramics, minerals, nanotechnology, and microfluidics, among others. There ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dukhin Number
The Dukhin number () is a dimensionless quantity that characterizes the contribution of the surface conductivity to various electrokinetic and electroacoustic effects, as well as to electrical conductivity and permittivity of fluid heterogeneous systems. The number was named after Stanislav and Andrei Dukhin. Overview It was introduced by Lyklema in “Fundamentals of Interface and Colloid Science”.Lyklema, J.J. "Fundamentals of Interface and Colloid Science", vol.2, page.3.208, 1995 . A recent IUPAC Technical Report used this term explicitly and detailed several means of measurement in physical systems. The Dukhin number is a ratio of the surface conductivity \kappa^ to the fluid bulk electrical conductivity Km multiplied by particle size ''a'': : = \frac. There is another expression of this number that is valid when the surface conductivity is associated only with ions motion above the slipping plane in the double layer. In this case, the value of the surface conductivity ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
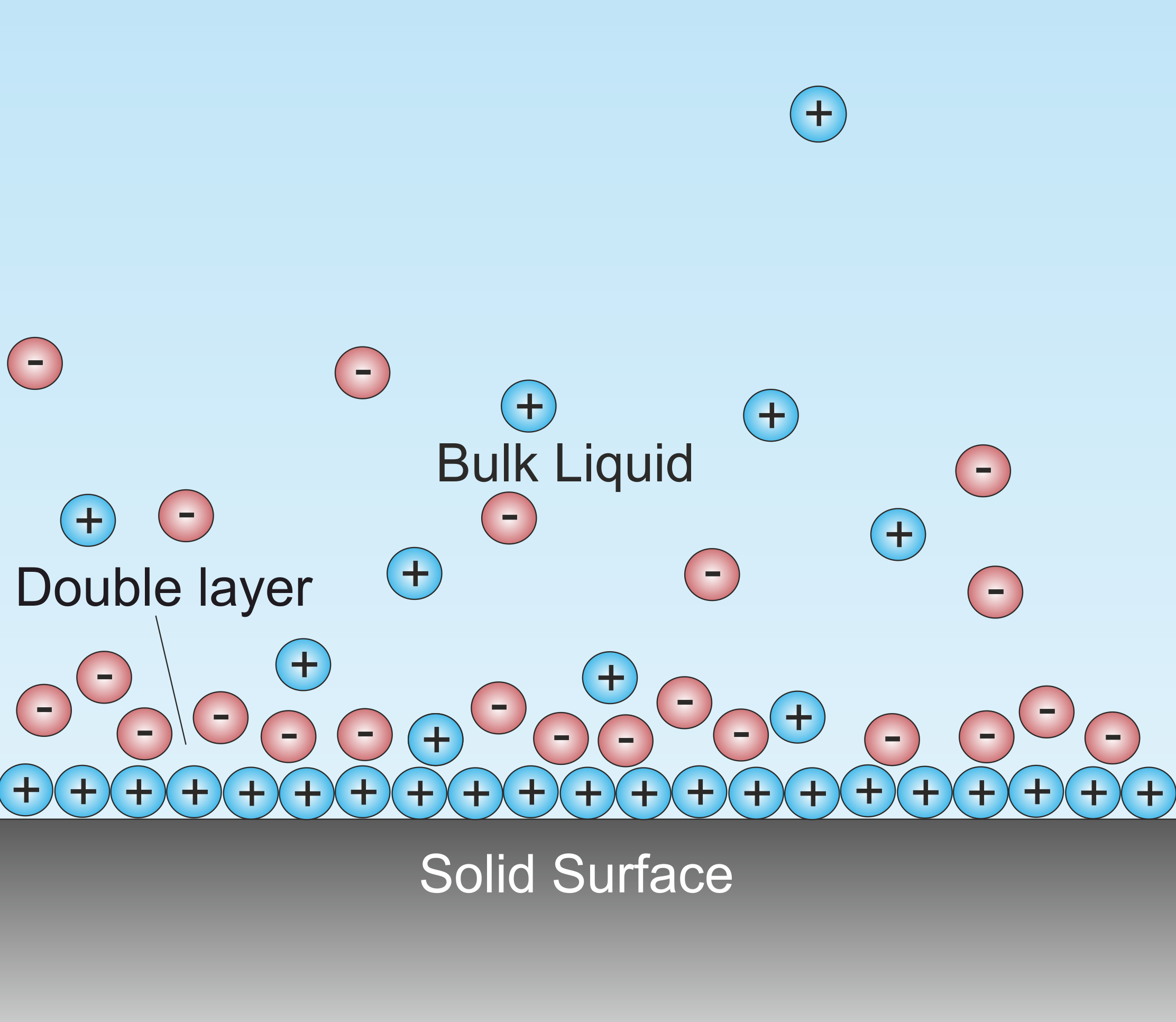
Surface Conductivity
Surface conductivity is an additional conductivity of an electrolyte in the vicinity of the charged interfaces. Surface and volume conductivity of liquids correspond to the electrically driven motion of ions in an electric field. A layer of counter ions of the opposite polarity to the surface charge exists close to the interface. It is formed due to attraction of counter-ions by the surface charges. This layer of higher ionic concentration is a part of the interfacial double layer. The concentration of the ions in this layer is higher as compared to the ionic strength of the liquid bulk. This leads to the higher electric conductivity of this layer. Smoluchowski was the first to recognize the importance of surface conductivity at the beginning of the 20th century. There is a detailed description of surface conductivity by Lyklema in "Fundamentals of Interface and Colloid Science" The Double Layer (DL) has two regions, according to the well established Gouy-Chapman-Stern model. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Debye Length
In plasmas and electrolytes, the Debye length \lambda_ (also called Debye radius), is a measure of a charge carrier's net electrostatic effect in a solution and how far its electrostatic effect persists. With each Debye length the charges are increasingly electrically screened and the electric potential decreases in magnitude by 1/ e. A Debye sphere is a volume whose radius is the Debye length. Debye length is an important parameter in plasma physics, electrolytes, and colloids (DLVO theory). The corresponding Debye screening wave vector k_=1/\lambda_ for particles of density n, charge q at a temperature T is given by k_^2=4\pi n q^2/(k_T) in Gaussian units. Expressions in MKS units will be given below. The analogous quantities at very low temperatures (T \to 0) are known as the Thomas–Fermi length and the Thomas–Fermi wave vector. They are of interest in describing the behaviour of electrons in metals at room temperature. The Debye length is named after the Dutch-America ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Double Layer (interfacial)
A double layer (DL, also called an electrical double layer, EDL) is a structure that appears on the surface of an object when it is exposed to a fluid. The object might be a solid particle, a gas bubble, a liquid droplet, or a porous body. The DL refers to two parallel layers of charge surrounding the object. The first layer, the surface charge (either positive or negative), consists of ions adsorbed onto the object due to chemical interactions. The second layer is composed of ions attracted to the surface charge via the Coulomb force, electrically screening the first layer. This second layer is loosely associated with the object. It is made of free ions that move in the fluid under the influence of electric attraction and thermal motion rather than being firmly anchored. It is thus called the "diffuse layer". Interfacial DLs are most apparent in systems with a large surface area to volume ratio, such as a colloid or porous bodies with particles or pores (respectively) on t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electrokinetic Potential
Zeta potential is the electrical potential at the slipping plane. This plane is the interface which separates mobile fluid from fluid that remains attached to the surface. Zeta potential is a scientific term for electrokinetic potential in colloidal dispersions. In the colloidal chemistry literature, it is usually denoted using the Greek letter zeta (ζ), hence ''ζ-potential''. The usual units are volts (V) or, more commonly, millivolts (mV). From a theoretical viewpoint, the zeta potential is the electric potential in the interfacial double layer (DL) at the location of the slipping plane relative to a point in the bulk fluid away from the interface. In other words, zeta potential is the potential difference between the dispersion medium and the stationary layer of fluid attached to the dispersed particle. The zeta potential is caused by the net electrical charge contained within the region bounded by the slipping plane, and also depends on the location of that plane. Th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electrophoresis
Electrophoresis, from Ancient Greek ἤλεκτρον (ḗlektron, "amber") and φόρησις (phórēsis, "the act of bearing"), is the motion of dispersed particles relative to a fluid under the influence of a spatially uniform electric field. Electrophoresis of positively charged particles (cations) is sometimes called cataphoresis, while electrophoresis of negatively charged particles (anions) is sometimes called anaphoresis. The electrokinetic phenomenon of electrophoresis was observed for the first time in 1807 by Russian professors Peter Ivanovich Strakhov and Ferdinand Frederic Reuss at Moscow University, who noticed that the application of a constant electric field caused clay particles dispersed in water to migrate. It is ultimately caused by the presence of a charged interface between the particle surface and the surrounding fluid. It is the basis for analytical techniques used in chemistry for separating molecules by size, charge, or binding affinity. Electropho ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |