|
Davies Equation
The Davies equation is an empirical extension of Debye–Hückel theory which can be used to calculate activity coefficients of electrolyte solutions at relatively high concentrations at 25 °C. The equation, originally published in 1938, was refined by fitting to experimental data. The final form of the equation gives the mean molal activity coefficient of an electrolyte that dissociates into ions having charges and as a function of ionic strength : :-\log f_\pm = 0.5 z_1 z_2\left(\frac - 0.30 I \right). The second term, , goes to zero as the ionic strength goes to zero, so the equation reduces to the Debye–Hückel equation at low concentration. However, as concentration increases, the second term becomes increasingly important, so the Davies equation can be used for solutions too concentrated to allow the use of the Debye–Hückel equation. For 1:1 electrolytes the difference between measured values and those calculated with this equation is about 2% of the value for ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Debye–Hückel Theory
The Debye–Hückel theory was proposed by Peter Debye and Erich Hückel as a theoretical explanation for departures from ideality in solutions of electrolytes and plasmas. It is a linearized Poisson–Boltzmann model, which assumes an extremely simplified model of electrolyte solution but nevertheless gave accurate predictions of mean activity coefficients for ions in dilute solution. The Debye–Hückel equation provides a starting point for modern treatments of non-ideality of electrolyte solutions. Overview In the chemistry of electrolyte solutions, an ideal solution is a solution whose colligative properties are proportional to the concentration of the solute. Real solutions may show departures from this kind of ideality. In order to accommodate these effects in the thermodynamics of solutions, the concept of activity was introduced: the properties are then proportional to the activities of the ions. Activity, ''a'', is proportional to concentration, ''c''. The proport ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
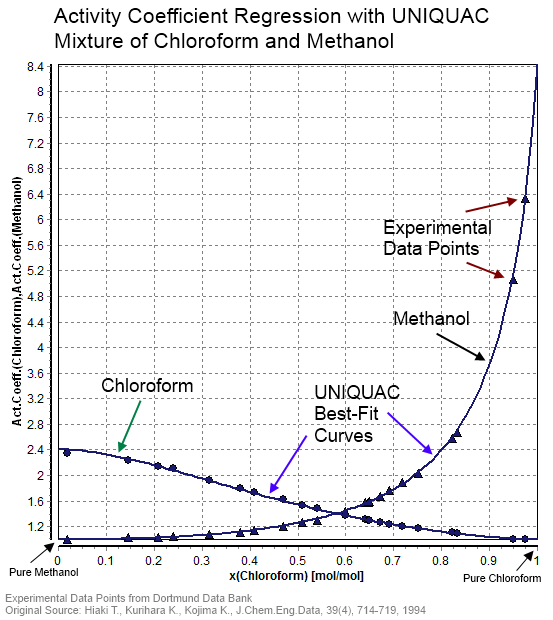
Activity Coefficient
In thermodynamics, an activity coefficient is a factor used to account for deviation of a mixture of chemical substances from ideal behaviour. In an ideal mixture, the microscopic interactions between each pair of chemical species are the same (or macroscopically equivalent, the enthalpy change of solution and volume variation in mixing is zero) and, as a result, properties of the mixtures can be expressed directly in terms of simple concentrations or partial pressures of the substances present e.g. Raoult's law. Deviations from ideality are accommodated by modifying the concentration by an ''activity coefficient''. Analogously, expressions involving gases can be adjusted for non-ideality by scaling partial pressures by a fugacity coefficient. The concept of activity coefficient is closely linked to that of activity in chemistry. Thermodynamic definition The chemical potential, \mu_\mathrm, of a substance B in an ideal mixture of liquids or an ideal solution is given by :\mu_ ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electrolyte
An electrolyte is a medium containing ions that is electrically conducting through the movement of those ions, but not conducting electrons. This includes most soluble salts, acids, and bases dissolved in a polar solvent, such as water. Upon dissolving, the substance separates into cations and anions, which disperse uniformly throughout the solvent. Solid-state electrolytes also exist. In medicine and sometimes in chemistry, the term electrolyte refers to the substance that is dissolved. Electrically, such a solution is neutral. If an electric potential is applied to such a solution, the cations of the solution are drawn to the electrode that has an abundance of electrons, while the anions are drawn to the electrode that has a deficit of electrons. The movement of anions and cations in opposite directions within the solution amounts to a current. Some gases, such as hydrogen chloride (HCl), under conditions of high temperature or low pressure can also function as electrolytes. El ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Molal
Molality is a measure of the number of moles of solute in a solution corresponding to 1 kg or 1000 g of solvent. This contrasts with the definition of molarity which is based on a specified volume of solution. A commonly used unit for molality in chemistry is mol/ kg. A solution of concentration 1 mol/kg is also sometimes denoted as ''1 molal''. The unit ''mol/kg'' requires that molar mass be expressed in ''kg/mol'', instead of the usual ''g/mol'' or ''kg/kmol''. Definition The molality (''b''), of a solution is defined as the amount of substance (in moles) of solute, ''n''solute, divided by the mass (in kg) of the solvent, ''m''solvent: :b = \frac In the case of solutions with more than one solvent, molality can be defined for the mixed solvent considered as a pure pseudo-solvent. Instead of mole solute per kilogram solvent as in the binary case, units are defined as mole solute per kilogram mixed solvent. Origin The term ''molality'' is formed in analogy to ''mo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ionic Strength
The ionic strength of a solution is a measure of the concentration of ions in that solution. Ionic compounds, when dissolved in water, dissociate into ions. The total electrolyte concentration in solution will affect important properties such as the dissociation constant or the solubility of different salts. One of the main characteristics of a solution with dissolved ions is the ionic strength. Ionic strength can be molar (mol/L solution) or molal (mol/kg solvent) and to avoid confusion the units should be stated explicitly. The concept of ionic strength was first introduced by Lewis and Randall in 1921 while describing the activity coefficients of strong electrolytes. Quantifying ionic strength The molar ionic strength, ''I'', of a solution is a function of the concentration of ''all'' ions present in that solution. :I = \begin\frac\end\sum_^ c_i z_i^ where one half is because we are including both cations and anions, ''c''i is the molar concentration of ion i (M, mol/L), ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Debye–Hückel Equation
The chemists Peter Debye and Erich Hückel noticed that solutions that contain ionic solutes do not behave ideally even at very low concentrations. So, while the concentration of the solutes is fundamental to the calculation of the dynamics of a solution, they theorized that an extra factor that they termed gamma is necessary to the calculation of the activity coefficients of the solution. Hence they developed the Debye–Hückel equation and Debye–Hückel limiting law. The activity is only proportional to the concentration and is altered by a factor known as the activity coefficient \gamma. This factor takes into account the interaction energy of ions in solution. Debye–Hückel limiting law In order to calculate the activity a_C of an ion C in a solution, one must know the concentration and the activity coefficient: a_C = \gamma \frac\mathrm\mathrm, where * \gamma is the activity coefficient of C, * \mathrm is the concentration of the chosen ''standard state'', e.g. 1 mol ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ion Pair
In chemistry, ion association is a chemical reaction whereby ions of opposite electric charge come together in solution to form a distinct chemical entity. Ion associates are classified, according to the number of ions that associate with each other, as ion pairs, ion triplets, etc. Ion pairs are also classified according to the nature of the interaction as contact, solvent-shared or solvent-separated. The most important factor to determine the extent of ion association is the dielectric constant of the solvent. Ion associates have been characterized by means of vibrational spectroscopy, as introduced by Niels Bjerrum, and dielectric-loss spectroscopy. Classification of ion pairs ''Ion pairs'' are formed when a cation and anion, which are present in a solution of an ionizable substance, come together to form a discrete chemical species. There are three distinct types of ''ion pairs'', depending on the extent of solvation of the two ions. For example, magnesium sulphate exist ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Osmotic Coefficient
An osmotic coefficient \phi is a quantity which characterises the deviation of a solvent from ideal behaviour, referenced to Raoult's law. It can be also applied to solutes. Its definition depends on the ways of expressing chemical composition of mixtures. The osmotic coefficient based on molality ''m'' is defined by: \phi = \frac and on a mole fraction basis by: \phi = -\frac where \mu_A^* is the chemical potential of the pure solvent and \mu_A is the chemical potential of the solvent in a solution, ''M''A is its molar mass, ''x''A its mole fraction, ''R'' the gas constant and ''T'' the temperature in Kelvin. The latter osmotic coefficient is sometimes called the rational osmotic coefficient. The values for the two definitions are different, but since \ln x_A = - \ln \left(1 + M_A \sum_i m_i \right) \approx - M_A \sum_i m_i, the two definitions are similar, and in fact both approach 1 as the concentration goes to zero. Applications For liquid solutions, the osmotic coeffi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pitzer Equations
Pitzer equations are important for the understanding of the behaviour of ions dissolved in natural waters such as rivers, lakes and sea-water. They were first described by physical chemist Kenneth Pitzer. The parameters of the Pitzer equations are linear combinations of parameters, of a virial expansion of the excess Gibbs free energy, which characterise interactions amongst ions and solvent. The derivation is thermodynamically rigorous at a given level of expansion. The parameters may be derived from various experimental data such as the osmotic coefficient, mixed ion activity coefficients, and salt solubility. They can be used to calculate mixed ion activity coefficients and water activities in solutions of high ionic strength for which the Debye–Hückel theory is no longer adequate. They are more rigorous than the equations of specific ion interaction theory (SIT theory), but Pitzer parameters are more difficult to determine experimentally than SIT parameters. Historical deve ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |