|
Dissipate
In thermodynamics, dissipation is the result of an irreversible process that takes place in homogeneous thermodynamic systems. In a dissipative process, energy (internal, bulk flow kinetic, or system potential) transforms from an initial form to a final form, where the capacity of the final form to do thermodynamic work is less than that of the initial form. For example, heat transfer is dissipative because it is a transfer of internal energy from a hotter body to a colder one. Following the second law of thermodynamics, the entropy varies with temperature (reduces the capacity of the combination of the two bodies to do work), but never decreases in an isolated system. These processes produce entropy at a certain rate. The entropy production rate times ambient temperature gives the dissipated power. Important examples of irreversible processes are: heat flow through a thermal resistance, fluid flow through a flow resistance, diffusion (mixing), chemical reactions, and electric ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Heat Flow
Heat transfer is a discipline of thermal engineering that concerns the generation, use, conversion, and exchange of thermal energy (heat) between physical systems. Heat transfer is classified into various mechanisms, such as thermal conduction, thermal convection, thermal radiation, and transfer of energy by phase changes. Engineers also consider the transfer of mass of differing chemical species (mass transfer in the form of advection), either cold or hot, to achieve heat transfer. While these mechanisms have distinct characteristics, they often occur simultaneously in the same system. Heat conduction, also called diffusion, is the direct microscopic exchanges of kinetic energy of particles (such as molecules) or quasiparticles (such as lattice waves) through the boundary between two systems. When an object is at a different temperature from another body or its surroundings, heat flows so that the body and the surroundings reach the same temperature, at which point they are in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Heat Transfer
Heat transfer is a discipline of thermal engineering that concerns the generation, use, conversion, and exchange of thermal energy (heat) between physical systems. Heat transfer is classified into various mechanisms, such as thermal conduction, thermal convection, thermal radiation, and transfer of energy by phase changes. Engineers also consider the transfer of mass of differing chemical species (mass transfer in the form of advection), either cold or hot, to achieve heat transfer. While these mechanisms have distinct characteristics, they often occur simultaneously in the same system. Heat conduction, also called diffusion, is the direct microscopic exchanges of kinetic energy of particles (such as molecules) or quasiparticles (such as lattice waves) through the boundary between two systems. When an object is at a different temperature from another body or its surroundings, heat flows so that the body and the surroundings reach the same temperature, at which point they are in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
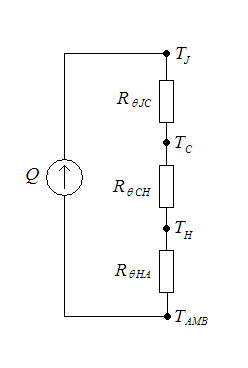
Thermal Resistance
Thermal resistance is a heat property and a measurement of a temperature difference by which an object or material resists a heat flow. Thermal resistance is the reciprocal of thermal conductance. * (Absolute) thermal resistance ''R'' in kelvins per watt (K/W) is a property of a particular component. For example, a characteristic of a heat sink. * Specific thermal resistance or thermal resistivity ''Rλ'' in kelvin–metres per watt (K⋅m/W), is a material constant. * Thermal insulance has the units square metre kelvin per watt (m2⋅K/W) in SI units or square foot degree Fahrenheit–hours per British thermal unit (ft2⋅°F⋅h/Btu) in imperial units. It is the thermal resistance of unit area of a material. In terms of insulation, it is measured by the R-value. Absolute thermal resistance Absolute thermal resistance is the temperature difference across a structure when a unit of heat energy flows through it in unit time. It is the reciprocal of thermal conductance. The ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electrical Resistance And Conductance
The electrical resistance of an object is a measure of its opposition to the flow of electric current. Its reciprocal quantity is , measuring the ease with which an electric current passes. Electrical resistance shares some conceptual parallels with mechanical friction. The SI unit of electrical resistance is the ohm (), while electrical conductance is measured in siemens (S) (formerly called the 'mho' and then represented by ). The resistance of an object depends in large part on the material it is made of. Objects made of electrical insulators like rubber tend to have very high resistance and low conductance, while objects made of electrical conductors like metals tend to have very low resistance and high conductance. This relationship is quantified by resistivity or conductivity. The nature of a material is not the only factor in resistance and conductance, however; it also depends on the size and shape of an object because these properties are extensive rather than int ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Joule Heating
Joule heating, also known as resistive, resistance, or Ohmic heating, is the process by which the passage of an electric current through a conductor produces heat. Joule's first law (also just Joule's law), also known in countries of former USSR as the Joule–Lenz law, Assuming the element behaves as a perfect resistor and that the power is completely converted into heat, the formula can be re-written by substituting Ohm's law, V = I R , into the generalized power equation: P = IV = I^2R = V^2/R where ''R'' is the resistance. Alternating current When current varies, as it does in AC circuits, P(t) = U(t) I(t) where ''t'' is time and ''P'' is the instantaneous power being converted from electrical energy to heat. Far more often, the ''average'' power is of more interest than the instantaneous power: P_ = U_\text I_\text = I_\text^2 R = U_\text^2 / R where "avg" denotes average (mean) over one or more cycles, and "rms" denotes root mean square. These formulas are valid fo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Kinetic Energy
In physics, the kinetic energy of an object is the energy that it possesses due to its motion. It is defined as the work needed to accelerate a body of a given mass from rest to its stated velocity. Having gained this energy during its acceleration, the body maintains this kinetic energy unless its speed changes. The same amount of work is done by the body when decelerating from its current speed to a state of rest. Formally, a kinetic energy is any term in a system's Lagrangian which includes a derivative with respect to time. In classical mechanics, the kinetic energy of a non-rotating object of mass ''m'' traveling at a speed ''v'' is \fracmv^2. In relativistic mechanics, this is a good approximation only when ''v'' is much less than the speed of light. The standard unit of kinetic energy is the joule, while the English unit of kinetic energy is the foot-pound. History and etymology The adjective ''kinetic'' has its roots in the Greek word κίνησις ''kines ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Energy Transformation
Energy transformation, also known as energy conversion, is the process of changing energy from one form to another. In physics, energy is a quantity that provides the capacity to perform work or moving, (e.g. Lifting an object) or provides heat. In addition to being converted, according to the law of conservation of energy, energy is transferable to a different location or object, but it cannot be created or destroyed. The energy in many of its forms may be used in natural processes, or to provide some service to society such as heating, refrigeration, lighting or performing mechanical work to operate machines. For example, to heat a home, the furnace burns fuel, whose chemical potential energy is converted into thermal energy, which is then transferred to the home's air to raise its temperature. Limitations in the conversion of thermal energy Conversions to thermal energy from other forms of energy may occur with 100% efficiency. Conversion among non-thermal forms of ener ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Energy
In physics, energy (from Ancient Greek: ἐνέργεια, ''enérgeia'', “activity”) is the quantitative property that is transferred to a body or to a physical system, recognizable in the performance of work and in the form of heat and light. Energy is a conserved quantity—the law of conservation of energy states that energy can be converted in form, but not created or destroyed. The unit of measurement for energy in the International System of Units (SI) is the joule (J). Common forms of energy include the kinetic energy of a moving object, the potential energy stored by an object (for instance due to its position in a field), the elastic energy stored in a solid object, chemical energy associated with chemical reactions, the radiant energy carried by electromagnetic radiation, and the internal energy contained within a thermodynamic system. All living organisms constantly take in and release energy. Due to mass–energy equivalence, any object th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Entropy Production
Entropy production (or generation) is the amount of entropy which is produced in any irreversible processes such as heat and mass transfer processes including motion of bodies, heat exchange, fluid flow, substances expanding or mixing, anelastic deformation of solids, and any irreversible thermodynamic cycle, including thermal machines such as power plants, heat engines, refrigerators, heat pumps, and air conditioners. In the dual representation entropy–exergy for accounting the second law of thermodynamics it can be expressed in equivalent terms of exergy disruption. Short history Entropy is produced in irreversible processes. The importance of avoiding irreversible processes (hence reducing the entropy production) was recognized as early as 1824 by Carnot. In 1865 Rudolf Clausius expanded his previous work from 1854 on the concept of "unkompensierte Verwandlungen" (uncompensated transformations), which, in our modern nomenclature, would be called the entropy production. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Power (physics)
In physics, power is the amount of energy transferred or converted per unit time. In the International System of Units, the unit of power is the watt, equal to one joule per second. In older works, power is sometimes called ''activity''. Power is a scalar quantity. Power is related to other quantities; for example, the power involved in moving a ground vehicle is the product of the aerodynamic drag plus traction force on the wheels, and the velocity of the vehicle. The output power of a motor is the product of the torque that the motor generates and the angular velocity of its output shaft. Likewise, the power dissipated in an electrical element of a circuit is the product of the current flowing through the element and of the voltage across the element. Definition Power is the rate with respect to time at which work is done; it is the time derivative of work: P =\frac where is power, is work, and is time. If a constant force F is applied throughout a distance x, t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Second Law Of Thermodynamics
The second law of thermodynamics is a physical law based on universal experience concerning heat and energy interconversions. One simple statement of the law is that heat always moves from hotter objects to colder objects (or "downhill"), unless energy in some form is supplied to reverse the direction of heat flow. Another definition is: "Not all heat energy can be converted into work in a cyclic process."Young, H. D; Freedman, R. A. (2004). ''University Physics'', 11th edition. Pearson. p. 764. The second law of thermodynamics in other versions establishes the concept of entropy as a physical property of a thermodynamic system. It can be used to predict whether processes are forbidden despite obeying the requirement of conservation of energy as expressed in the first law of thermodynamics and provides necessary criteria for spontaneous processes. The second law may be formulated by the observation that the entropy of isolated systems left to spontaneous evolution cannot decr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Measure-preserving Dynamical System
In mathematics, a measure-preserving dynamical system is an object of study in the abstract formulation of dynamical systems, and ergodic theory in particular. Measure-preserving systems obey the Poincaré recurrence theorem, and are a special case of conservative systems. They provide the formal, mathematical basis for a broad range of physical systems, and, in particular, many systems from classical mechanics (in particular, most non-dissipative systems) as well as systems in thermodynamic equilibrium. Definition A measure-preserving dynamical system is defined as a probability space and a measure-preserving transformation on it. In more detail, it is a system :(X, \mathcal, \mu, T) with the following structure: *X is a set, *\mathcal B is a σ-algebra over X, *\mu:\mathcal\rightarrow ,1/math> is a probability measure, so that \mu (X) = 1, and \mu(\varnothing) = 0, * T:X \rightarrow X is a measurable transformation which preserves the measure \mu, i.e., \forall A\in \m ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |