|
Dhevalapally B. Ramachary
Dhevalapally B. Ramachary FTAS, FRSC, FASc, FNASc, also known as D. B. Ramachary (born 1973), is an Indian chemist and professor at the School of Chemistry, University of Hyderabad. He has made numerous contributions in various fields of chemical science. Early life and education D. B. Ramachary was born to Shri Ramalingaiahchary and Ramalingamma in 1973, at Thatikal village, Nakrekal mandal, Nalgonda district of Telangana. He did his early schooling at ZPHS schools at Thatikal and Nakrekal, and later joined for an under graduate BSc programme at Nagarjuna Government College (Autonomous) at Nalgonda (1991–1994). Later, he moved to School of Chemistry, University of Hyderabad for pursuing MSc chemistry (1994–96), and then he obtained a PhD in synthetic organic chemistry under the guidance of Prof. A. Srikrishna at Indian Institute of Science in 1996–2001 for the total synthesis of sesquiterpenes. Soon after his PhD in 2001, he moved to US as a Skaggs Postdoctoral ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nalgonda
Nalgonda is a city and municipality in the Indian state of Telangana. It is the headquarters of the Nalgonda district, as well as the headquarters of the Nalgonda mandal in the Nalgonda revenue division. It is located about from the state capital Hyderabad. Etymology In the past, Nalgonda was referred to as ''Nilagiri''. During the medieval Bahamani kingdom, it was renamed Nalgunda. The name was changed to "Nalgonda" for official uses during the rule of the later Nizam kings. History Paleolithic Age There is archaeological evidence that Paleolithic people lived in the area that is now Nalgonda, fashioning tools and weapons out of stone. Some of these implements have been found in the Nalgonda area, similar to those discovered at the Sloan archaeological site in Arkansas. Neolithic Age Traces of Neolithic culture were found at Chota Yelupu, where sling stones and other contemporary objects were excavated. Evidence of Megalithic culture was also found via the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Professor
Professor (commonly abbreviated as Prof.) is an Academy, academic rank at university, universities and other post-secondary education and research institutions in most countries. Literally, ''professor'' derives from Latin as a "person who professes". Professors are usually experts in their field and teachers of the highest rank. In most systems of List of academic ranks, academic ranks, "professor" as an unqualified title refers only to the most senior academic position, sometimes informally known as "full professor". In some countries and institutions, the word "professor" is also used in titles of lower ranks such as associate professor and assistant professor; this is particularly the case in the United States, where the unqualified word is also used colloquially to refer to associate and assistant professors as well. This usage would be considered incorrect among other academic communities. However, the otherwise unqualified title "Professor" designated with a capital let ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
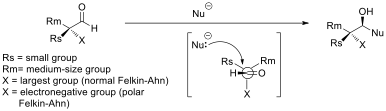
Asymmetric Induction
In stereochemistry, asymmetric induction (also enantioinduction) describes the preferential formation in a chemical reaction of one enantiomer or diastereoisomer over the other as a result of the influence of a chiral feature present in the substrate, reagent, catalyst or environment. Asymmetric induction is a key element in asymmetric synthesis. Asymmetric induction was introduced by Hermann Emil Fischer based on his work on carbohydrates. Several types of induction exist. Internal asymmetric induction makes use of a chiral center bound to the reactive center through a covalent bond and remains so during the reaction. The starting material is often derived from chiral pool synthesis. In relayed asymmetric induction the chiral information is introduced in a separate step and removed again in a separate chemical reaction. Special synthons are called chiral auxiliaries. In external asymmetric induction chiral information is introduced in the transition state through a catalyst o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Michael Reaction
In organic chemistry, the Michael reaction or Michael addition is a reaction between a Michael donor (an enolate or other nucleophile) and a Michael acceptor (usually an α,β-unsaturated carbonyl) to produce a Michael adduct by creating a carbon-carbon bond at the acceptor's β-carbon. It belongs to the larger class of conjugate additions and is widely used for the mild formation of carbon-carbon bonds. The Michael addition is an important atom-economical method for diastereoselective and enantioselective C–C bond formation, and many asymmetric variants exist : In this general Michael addition scheme, either or both of R and R' on the nucleophile (the Michael donor) represent electron-withdrawing substituents such as acyl, cyano, nitro, or sulfone groups, which make the adjacent methylene hydrogen acidic enough to form a carbanion when reacted with the base, ''B:''. For the alkene (the Michael acceptor), the R" substituent is usually a carbonyl, which makes the compound ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Asymmetric Reaction
Enantioselective synthesis, also called asymmetric synthesis, is a form of chemical synthesis. It is defined by IUPAC as "a chemical reaction (or reaction sequence) in which one or more new elements of chirality are formed in a substrate molecule and which produces the stereoisomeric ( enantiomeric or diastereomeric) products in unequal amounts." Put more simply: it is the synthesis of a compound by a method that favors the formation of a specific enantiomer or diastereomer. Enantiomers are stereoisomers that have opposite configurations at every chiral center. Diastereomers are stereoisomers that differ at one or more chiral centers. Enantioselective synthesis is a key process in modern chemistry and is particularly important in the field of pharmaceuticals, as the different enantiomers or diastereomers of a molecule often have different biological activity. Overview Many of the building blocks of biological systems such as sugars and amino acids are produced exclusive ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitrosobenzene
Nitrosobenzene is the organic compound with the formula C6H5NO. It is one of the prototypical organic nitroso compounds. Characteristic of its functional group, it is a dark green species that exists in equilibrium with its pale yellow dimer. Both monomer and dimer are diamagnetic. Monomer-dimer equilibrium Nitrosobenzene and other nitrosoarenes typically participate in a monomer-dimer equilibrium. The dimers are often favored in the solid state, whereas the deeply colored monomers are favored in dilute solution or at higher temperatures. The dimers can be formulated as Ar(O−)N+=N+(O−)Ar. They exist as ''cis''- and ''trans''-isomers due to the presence of the N–N double bond. The dimers are sometimes called azobenzenedioxides. The cis-trans isomerization occurs via the intermediacy of the monomer. In the case of nitrosobenzene itself, the metastable monomeric form could be prepared by sublimation onto a cold finger. The monomeric material is selectively sublimed due to ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ketones
In organic chemistry, a ketone is a functional group with the structure R–C(=O)–R', where R and R' can be a variety of carbon-containing substituents. Ketones contain a carbonyl group –C(=O)– (which contains a carbon-oxygen double bond C=O). The simplest ketone is acetone (where R and R' is methyl), with the formula . Many ketones are of great importance in biology and in industry. Examples include many sugars (ketoses), many steroids (e.g., testosterone), and the solvent acetone. Nomenclature and etymology The word ''ketone'' is derived from ''Aketon'', an old German word for ''acetone''. According to the rules of IUPAC nomenclature, ketone names are derived by changing the suffix ''-ane'' of the parent alkane to ''-anone''. Typically, the position of the carbonyl group is denoted by a number, but traditional nonsystematic names are still generally used for the most important ketones, for example acetone and benzophenone. These nonsystematic names are considered ret ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dipole-dipole Interactions
An intermolecular force (IMF) (or secondary force) is the force that mediates interaction between molecules, including the electromagnetic forces of attraction or repulsion which act between atoms and other types of neighbouring particles, e.g. atoms or ions. Intermolecular forces are weak relative to intramolecular forces – the forces which hold a molecule together. For example, the covalent bond, involving sharing electron pairs between atoms, is much stronger than the forces present between neighboring molecules. Both sets of forces are essential parts of force fields frequently used in molecular mechanics. The investigation of intermolecular forces starts from macroscopic observations which indicate the existence and action of forces at a molecular level. These observations include non-ideal-gas thermodynamic behavior reflected by virial coefficients, vapor pressure, viscosity, superficial tension, and absorption data. The first reference to the nature of microscopic for ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Stereoselective Reactions
Enantioselective synthesis, also called asymmetric synthesis, is a form of chemical synthesis. It is defined by IUPAC as "a chemical reaction (or reaction sequence) in which one or more new elements of chirality are formed in a substrate molecule and which produces the stereoisomeric (enantiomeric or diastereomeric) products in unequal amounts." Put more simply: it is the synthesis of a compound by a method that favors the formation of a specific enantiomer or diastereomer. Enantiomers are stereoisomers that have opposite configurations at every chiral center. Diastereomers are stereoisomers that differ at one or more chiral centers. Enantioselective synthesis is a key process in modern chemistry and is particularly important in the field of pharmaceuticals, as the different enantiomers or diastereomers of a molecule often have different biological activity. Overview Many of the building blocks of biological systems such as sugars and amino acids are produced exclusively as ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organocatalyst
In organic chemistry, organocatalysis is a form of catalysis in which the rate of a chemical reaction is increased by an Organic compound, organic catalyst. This "organocatalyst" consists of carbon, hydrogen, sulfur and other nonmetal elements found in organic compounds.Special Issue: Because of their similarity in composition and description, they are often mistaken as a misnomer for enzymes due to their comparable effects on reaction rates and forms of catalysis involved. Organocatalysts which display secondary amine functionality can be described as performing either enamine catalysis (by forming catalytic quantities of an active enamine nucleophile) or imine, iminium catalysis (by forming catalytic quantities of an activated iminium electrophile). This mechanism is typical for covalent organocatalysis. Covalent binding of substrate normally requires high catalyst loading (for proline-catalysis typically 20–30 mol%). Noncovalent interactions such as hydrogen-bonding fa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organocatalysis
In organic chemistry, organocatalysis is a form of catalysis in which the rate of a chemical reaction is increased by an organic catalyst. This "organocatalyst" consists of carbon, hydrogen, sulfur and other nonmetal elements found in organic compounds.Special Issue: Because of their similarity in composition and description, they are often mistaken as a misnomer for enzymes due to their comparable effects on reaction rates and forms of catalysis involved. Organocatalysts which display secondary amine functionality can be described as performing either enamine catalysis (by forming catalytic quantities of an active enamine nucleophile) or iminium catalysis (by forming catalytic quantities of an activated iminium electrophile). This mechanism is typical for covalent organocatalysis. Covalent binding of substrate normally requires high catalyst loading (for proline-catalysis typically 20–30 mol%). Noncovalent interactions such as hydrogen-bonding facilitates low catalyst l ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Click Chemistry
In chemical synthesis, click chemistry is a class of biocompatible small molecule reactions commonly used in bioconjugation, allowing the joining of substrates of choice with specific biomolecules. Click chemistry is not a single specific reaction, but describes a way of generating products that follow examples in nature, which also generates substances by joining small modular units. In many applications, click reactions join a biomolecule and a reporter molecule. Click chemistry is not limited to biological conditions: the concept of a "click" reaction has been used in chemoproteomic, pharmacological, and various biomimetic applications. However, they have been made notably useful in the detection, localization and qualification of biomolecules. Click reactions occur in one pot, are not disturbed by water, generate minimal and inoffensive byproducts, and are "spring-loaded"—characterized by a high thermodynamic driving force that drives it quickly and irreversibly to high yi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |