|
Chromium(III) Sulfate
Chromium(III) sulfate usually refers to the inorganic compounds with the formula Cr2(SO4)3.x(H2O), where x can range from 0 to 18. Additionally, ill-defined but commercially important "basic chromium sulfates" are known. These salts are usually either violet or green solids that are soluble in water. It is commonly used in tanning leather. Chromium(III) sulfates Three chromium(III) sulfates are well characterized: *Anhydrous chromium(III) sulfate, Cr2(SO4)3, (CAS #10101-53-8) is a violet solid that dissolves in water upon addition of a reducing agent, which generates chromium(II) sulfates. *Hydrated chromium(III) sulfate, Cr2(SO4)3·18H2O, (CAS #13520-66-6) is a violet solid that readily dissolves in water to give the metal aquo complex, r(H2O)6sup>3+. The formula of this compound can be written more descriptively as r(H2O)6sub>2(SO4)3·6H2O. Six of the eighteen water molecules in this formula unit are water of crystallization. *Hydrated chromium(III) sulfate, Cr2(SO4)3·15(H2 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chromic Acid
The term chromic acid is usually used for a mixture made by adding concentrated sulfuric acid to a dichromate, which may contain a variety of compounds, including solid chromium trioxide. This kind of chromic acid may be used as a cleaning mixture for glass. Chromic acid may also refer to the molecular species, H2CrO4 of which the trioxide is the anhydride. Chromic acid features chromium in an oxidation state of +6 (or VI). It is a strong and corrosive oxidising agent. Molecular chromic acid Molecular chromic acid, H2CrO4, has much in common with sulfuric acid, H2SO4. Only sulfuric acid can be classified as part of the 7 strong acids list. Due to the laws pertinent to the concept of "first order ionization energy", the first proton is lost most easily. It behaves extremely similar to sulfuric acid deprotonation. Since the process of polyvalent acid-base titrations have more than one proton (especially when the acid is starting substance and the base is the titrant), protons ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfur Dioxide
Sulfur dioxide (IUPAC-recommended spelling) or sulphur dioxide (traditional Commonwealth English) is the chemical compound with the formula . It is a toxic gas responsible for the odor of burnt matches. It is released naturally by volcanic activity and is produced as a by-product of copper extraction and the burning of sulfur- bearing fossil fuels. Structure and bonding SO2 is a bent molecule with ''C''2v symmetry point group. A valence bond theory approach considering just ''s'' and ''p'' orbitals would describe the bonding in terms of resonance between two resonance structures. The sulfur–oxygen bond has a bond order of 1.5. There is support for this simple approach that does not invoke ''d'' orbital participation. In terms of electron-counting formalism, the sulfur atom has an oxidation state of +4 and a formal charge of +1. Occurrence Sulfur dioxide is found on Earth and exists in very small concentrations and in the atmosphere at about 1 ppm. On other p ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bentorite
Bentorite is a mineral with the chemical formula Ca6(Cr,Al)2(SO4)3(OH)12·26H2O. It is colored violet to light violet. Its crystals are hexagonal to dihexagonal dipyramidal. It is transparent and has vitreous luster. It has perfect cleavage. It is not radioactive. Bentorite is rated 2 on the Mohs Scale. The mineral was first described in 1980 by Shulamit Gross for an occurrence in the Hatrurim Formation of Danian age along the western margin of the Dead Sea, Israel. It was named by its discoverer, Shulamit Gross, for Yaakov Ben-Tor (1910–2002), Professor at the Hebrew University of Jerusalem and the University of California, San Diego, California, US, for his contributions to geology and mineralogy in Israel. Formation The only naturally occurring bentorite that has been discovered is in the Hatrurim Formation near the Dead Sea in Israel. The formation consists of a mixture of metamorphosed clays, limestones, and marls. The original sediments were enriched in chromium, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hexavalent Chromium
Hexavalent chromium (chromium(VI), Cr(VI), chromium 6) is chromium in any chemical compound that contains the element in the +6 oxidation state (thus hexavalent). Virtually all chromium ore is processed via hexavalent chromium, specifically the salt sodium dichromate. Hexavalent chromium is key to all materials made from chromium. Approximately of hexavalent chromium were produced in 1985. Additional hexavalent chromium compounds include chromium trioxide and various salts of chromate and dichromate, among others. Hexavalent chromium is used in textile dyes, wood preservation, anti-corrosion products, chromate conversion coatings, and a variety of niche uses. Industrial uses of hexavalent chromium compounds include chromate pigments in dyes, paints, inks, and plastics; chromates added as anticorrosive agents to paints, primers, and other surface coatings; and chromic acid electroplated onto metal parts to provide a decorative or protective coating. Hexavalent chromium can be ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Formate
Sodium formate, HCOONa, is the sodium salt of formic acid, HCOOH. It usually appears as a white deliquescent powder. Preparation For commercial use, sodium formate is produced by absorbing carbon monoxide under pressure in solid sodium hydroxide at 130 °C and 6-8 bar pressure:Arnold Willmes, ''Taschenbuch Chemische Substanzen'', Harri Deutsch, Frankfurt (M.), 2007. :CO + NaOH → HCO2Na Because of the low-cost and large-scale availability of formic acid by carbonylation of methanol and hydrolysis of the resulting methyl formate, sodium formate is usually prepared by neutralizing formic acid with sodium hydroxide. Sodium formate is also unavoidably formed as a by-product in the final step of the pentaerythritol synthesis and in the crossed Cannizzaro reaction of formaldehyde with the aldol reaction product trimethylol acetaldehyde -hydroxy-2,2-bis(hydroxymethyl)propanal In the laboratory, sodium formate can be prepared by neutralizing formic acid with sodium carbonate ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Carbonate
Sodium carbonate, , (also known as washing soda, soda ash and soda crystals) is the inorganic compound with the formula Na2CO3 and its various hydrates. All forms are white, odourless, water-soluble salts that yield moderately alkaline solutions in water. Historically, it was extracted from the ashes of plants growing in sodium-rich soils. Because the ashes of these sodium-rich plants were noticeably different from ashes of wood (once used to produce potash), sodium carbonate became known as "soda ash". It is produced in large quantities from sodium chloride and limestone by the Solvay process. Hydrates Sodium carbonate is obtained as three hydrates and as the anhydrous salt: * sodium carbonate decahydrate ( natron), Na2CO3·10H2O, which readily effloresces to form the monohydrate. * sodium carbonate heptahydrate (not known in mineral form), Na2CO3·7H2O. * sodium carbonate monohydrate ( thermonatrite), Na2CO3·H2O. Also known as crystal carbonate. * anhydrous sodium carbonate ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Jargon
Jargon is the specialized terminology associated with a particular field or area of activity. Jargon is normally employed in a particular communicative context and may not be well understood outside that context. The context is usually a particular occupation (that is, a certain trade, profession, vernacular or academic field), but any ingroup can have jargon. The main trait that distinguishes jargon from the rest of a language is special vocabulary—including some words specific to it and often different senses or meanings of words, that outgroups would tend to take in another sense—therefore misunderstanding that communication attempt. Jargon is sometimes understood as a form of technical slang and then distinguished from the official terminology used in a particular field of activity. The terms ''jargon'', ''slang,'' and ''argot'' are not consistently differentiated in the literature; different authors interpret these concepts in varying ways. According to one definition, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Anion
An ion () is an atom or molecule with a net electrical charge. The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton, which is considered to be positive by convention. The net charge of an ion is not zero because its total number of electrons is unequal to its total number of protons. A cation is a positively charged ion with fewer electrons than protons while an anion is a negatively charged ion with more electrons than protons. Opposite electric charges are pulled towards one another by electrostatic force, so cations and anions attract each other and readily form ionic compounds. Ions consisting of only a single atom are termed atomic or monatomic ions, while two or more atoms form molecular ions or polyatomic ions. In the case of physical ionization in a fluid (gas or liquid), "ion pairs" are created by spontaneous molecule collisions, where each generated pair consists of a free electron ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
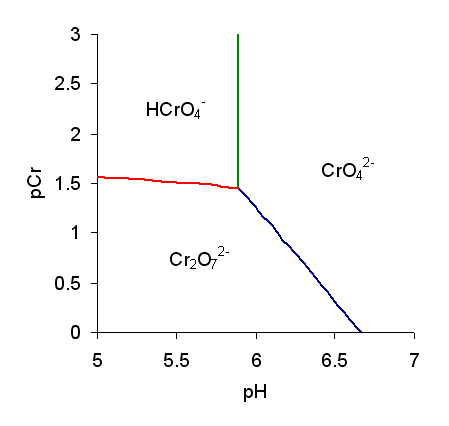
Chromate Ion
Chromate salts contain the chromate anion, . Dichromate salts contain the dichromate anion, . They are oxyanions of chromium in the +6 oxidation state and are moderately strong oxidizing agents. In an aqueous solution, chromate and dichromate ions can be interconvertible. Chemical properties Potassium-chromate-sample.jpg, potassium chromate Potassium-dichromate-sample.jpg, potassium dichromate Chromates react with hydrogen peroxide, giving products in which peroxide, , replaces one or more oxygen atoms. In acid solution the unstable blue peroxo complex Chromium(VI) oxide peroxide, CrO(O2)2, is formed; it is an uncharged covalent molecule, which may be extracted into ether. Addition of pyridine results in the formation of the more stable complex CrO(O2)2py. Acid–base properties In aqueous solution, chromate and dichromate anions exist in a chemical equilibrium. :2 + 2 H+ + H2O The predominance diagram shows that the position of the equilibrium depends on ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ethanol
Ethanol (abbr. EtOH; also called ethyl alcohol, grain alcohol, drinking alcohol, or simply alcohol) is an organic compound. It is an alcohol with the chemical formula . Its formula can be also written as or (an ethyl group linked to a hydroxyl group). Ethanol is a volatile, flammable, colorless liquid with a characteristic wine-like odor and pungent taste. It is a psychoactive recreational drug, the active ingredient in alcoholic drinks. Ethanol is naturally produced by the fermentation process of sugars by yeasts or via petrochemical processes such as ethylene hydration. It has medical applications as an antiseptic and disinfectant. It is used as a chemical solvent and in the synthesis of organic compounds, and as a fuel source. Ethanol also can be dehydrated to make ethylene, an important chemical feedstock. As of 2006, world production of ethanol was , coming mostly from Brazil and the U.S. Etymology ''Ethanol'' is the systematic name defined by the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
-oxid.jpg)




