|
Cerium Anomaly
The cerium anomaly, in geochemistry, is the phenomenon whereby cerium (Ce) concentration is either depleted or enriched in a rock relative to the other rare-earth elements (REEs). A Ce anomaly is said to be "negative" if Ce is depleted relative to the other REEs and is said to be "positive" if Ce is enriched relative to the other REEs. Cerium oxidation states Cerium is a rare-earth element ( lanthanide) characterized by two different redox states: III and IV. Contrary to other lanthanide elements, which are only trivalent (with the notable exception of Eu2+), Ce3+ can be oxidized by atmospheric oxygen (O2) to Ce4+ under alkaline conditions. The cerium anomaly relates to the decrease in solubility, which accompanies the oxidation of Ce(III) to Ce(IV). Under reducing conditions, Ce3+ is relatively soluble, while under oxidizing conditions CeO2 precipitates. Sediments deposited under oxic or anoxic conditions can preserve on the long term the geochemical signature of Ce3+ or Ce4+ ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Geochemistry
Geochemistry is the science that uses the tools and principles of chemistry to explain the mechanisms behind major geological systems such as the Earth's crust and its oceans. The realm of geochemistry extends beyond the Earth, encompassing the entire Solar System, and has made important contributions to the understanding of a number of processes including mantle convection, the formation of planets and the origins of granite and basalt. It is an integrated field of chemistry and geology. History The term ''geochemistry'' was first used by the Swiss-German chemist Christian Friedrich Schönbein in 1838: "a comparative geochemistry ought to be launched, before geognosy can become geology, and before the mystery of the genesis of our planets and their inorganic matter may be revealed." However, for the rest of the century the more common term was "chemical geology", and there was little contact between geologists and chemists. Geochemistry emerged as a separate discipline after ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Zircon
Zircon () is a mineral belonging to the group of nesosilicates and is a source of the metal zirconium. Its chemical name is zirconium(IV) silicate, and its corresponding chemical formula is Zr SiO4. An empirical formula showing some of the range of substitution in zircon is (Zr1–y, REEy)(SiO4)1–x(OH)4x–y. Zircon precipitates from silicate melts and has relatively high concentrations of high field strength incompatible elements. For example, hafnium is almost always present in quantities ranging from 1 to 4%. The crystal structure of zircon is tetragonal crystal system. The natural color of zircon varies between colorless, yellow-golden, red, brown, blue, and green. The name derives from the Persian ''zargun'', meaning "gold-hued". This word is corrupted into "jargoon", a term applied to light-colored zircons. The English word "zircon" is derived from ''Zirkon'', which is the German adaptation of this word. Yellow, orange, and red zircon is also known as "hyacinth", ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Rare-earth Element
The rare-earth elements (REE), also called the rare-earth metals or (in context) rare-earth oxides or sometimes the lanthanides (yttrium and scandium are usually included as rare earths), are a set of 17 nearly-indistinguishable lustrous silvery-white soft heavy metals. Compounds containing rare earths have diverse applications in electrical and electronic components, lasers, glass, magnetic materials, and industrial processes. Scandium and yttrium are considered rare-earth elements because they tend to occur in the same ore deposits as the lanthanides and exhibit similar chemical properties, but have different electronic and magnetic properties. These metals tarnish slowly in air at room temperature and react slowly with cold water to form hydroxides, liberating hydrogen. They react with steam to form oxides, and at elevated temperature (400°C) ignite spontaneously. These elements and their compounds have no biological function other than in several specialized enzymes, s ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lanthanide
The lanthanide () or lanthanoid () series of chemical elements comprises the 15 metallic chemical elements with atomic numbers 57–71, from lanthanum through lutetium. These elements, along with the chemically similar elements scandium and yttrium, are often collectively known as the rare-earth elements or rare-earth metals. The informal chemical symbol Ln is used in general discussions of lanthanide chemistry to refer to any lanthanide. All but one of the lanthanides are f-block elements, corresponding to the filling of the 4f electron shell. There is some dispute on whether lanthanum or lutetium is a d-block element, but lutetium is usually considered so by those who study the matter; it is included due to its chemical similarities with the other 14. All lanthanide elements form trivalent cations, Ln3+, whose chemistry is largely determined by the ionic radius, which decreases steadily from lanthanum to lutetium. These elements are called lanthanides because the elements i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Europium
Europium is a chemical element with the symbol Eu and atomic number 63. Europium is the most reactive lanthanide by far, having to be stored under an inert fluid to protect it from atmospheric oxygen or moisture. Europium is also the softest lanthanide, as it can be dented with a fingernail and easily cut with a knife. When oxidation is removed a shiny-white metal is visible. Europium was isolated in 1901 and is named after the continent of Europe. Being a typical member of the lanthanide series, europium usually assumes the oxidation state +3, but the oxidation state +2 is also common. All europium compounds with oxidation state +2 are slightly reducing. Europium has no significant biological role and is relatively non-toxic as compared to other heavy metals. Most applications of europium exploit the phosphorescence of europium compounds. Europium is one of the rarest of the rare-earth elements on Earth.Stwertka, Albert. ''A Guide to the Elements'', Oxford University Press, 1996, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cerium
Cerium is a chemical element with the symbol Ce and atomic number 58. Cerium is a soft, ductile, and silvery-white metal that tarnishes when exposed to air. Cerium is the second element in the lanthanide series, and while it often shows the +3 oxidation state characteristic of the series, it also has a stable +4 state that does not oxidize water. It is also considered one of the rare-earth elements. Cerium has no known biological role in humans but is not particularly toxic, except with intense or continued exposure. Despite always occurring in combination with the other rare-earth elements in minerals such as those of the monazite and bastnäsite groups, cerium is easy to extract from its ores, as it can be distinguished among the lanthanides by its unique ability to be oxidized to the +4 state in aqueous solution. It is the most common of the lanthanides, followed by neodymium, lanthanum, and praseodymium. It is the 25th-most abundant element, making up 66 ppm of the Ear ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tuff
Tuff is a type of rock made of volcanic ash ejected from a vent during a volcanic eruption. Following ejection and deposition, the ash is lithified into a solid rock. Rock that contains greater than 75% ash is considered tuff, while rock containing 25% to 75% ash is described as tuffaceous (for example, ''tuffaceous sandstone''). Tuff composed of sandy volcanic material can be referred to as volcanic sandstone. Tuff is a relatively soft rock, so it has been used for construction since ancient times. Because it is common in Italy, the Romans used it often for construction. The Rapa Nui people used it to make most of the ''moai'' statues on Easter Island. Tuff can be classified as either igneous or sedimentary rock. It is usually studied in the context of igneous petrology, although it is sometimes described using sedimentological terms. Tuff is often erroneously called tufa in guidebooks and in television programmes. Volcanic ash The material that is expelled in a volcanic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
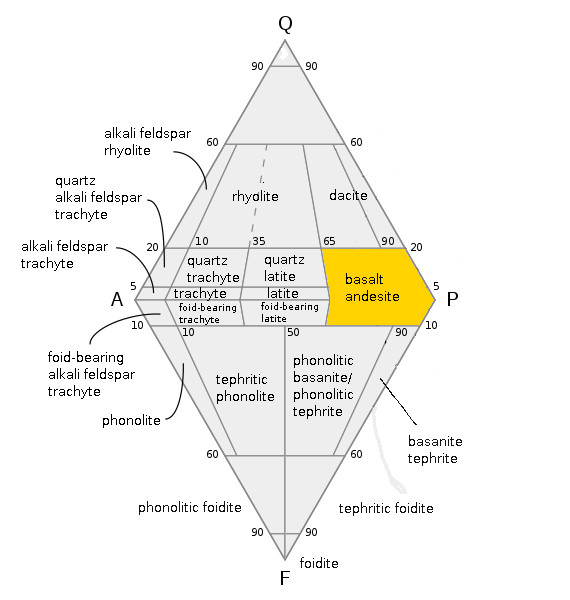
Basalt
Basalt (; ) is an aphanite, aphanitic (fine-grained) extrusive igneous rock formed from the rapid cooling of low-viscosity lava rich in magnesium and iron (mafic lava) exposed at or very near the planetary surface, surface of a terrestrial planet, rocky planet or natural satellite, moon. More than 90% of all volcanic rock on Earth is basalt. Rapid-cooling, fine-grained basalt is chemically equivalent to slow-cooling, coarse-grained gabbro. The eruption of basalt lava is observed by geologists at about 20 volcanoes per year. Basalt is also an important rock type on other planetary bodies in the Solar System. For example, the bulk of the plains of volcanism on Venus, Venus, which cover ~80% of the surface, are basaltic; the lunar mare, lunar maria are plains of flood-basaltic lava flows; and basalt is a common rock on the surface of Mars. Molten basalt lava has a low viscosity due to its relatively low silica content (between 45% and 52%), resulting in rapidly moving lava flo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mafic
A mafic mineral or rock is a silicate mineral or igneous rock rich in magnesium and iron. Most mafic minerals are dark in color, and common rock-forming mafic minerals include olivine, pyroxene, amphibole, and biotite. Common mafic rocks include basalt, diabase and gabbro. Mafic rocks often also contain calcium-rich varieties of plagioclase feldspar. Mafic materials can also be described as ferromagnesian. History The term ''mafic'' is a portmanteau of "magnesium" and "ferric" and was coined by Charles Whitman Cross, Joseph P. Iddings, Louis Valentine Pirsson, and Henry Stephens Washington in 1912. Cross' group had previously divided the major rock-forming minerals found in igneous rocks into ''salic'' minerals, such as quartz, feldspars, or feldspathoids, and ''femic'' minerals, such as olivine and pyroxene. However, micas and aluminium-rich amphiboles were excluded, while some calcium minerals containing little iron or magnesium, such as wollastonite or apatite, were included ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fugacity
In chemical thermodynamics, the fugacity of a real gas is an effective partial pressure which replaces the mechanical partial pressure in an accurate computation of the chemical equilibrium constant. It is equal to the pressure of an ideal gas which has the same temperature and molar Gibbs free energy as the real gas. Fugacities are determined experimentally or estimated from various models such as a Van der Waals gas that are closer to reality than an ideal gas. The real gas pressure and fugacity are related through the dimensionless fugacity coefficient . \varphi = \frac For an ideal gas, fugacity and pressure are equal and so . Taken at the same temperature and pressure, the difference between the molar Gibbs free energies of a real gas and the corresponding ideal gas is equal to . The fugacity is closely related to the thermodynamic activity. For a gas, the activity is simply the fugacity divided by a reference pressure to give a dimensionless quantity. This reference press ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Zirconium
Zirconium is a chemical element with the symbol Zr and atomic number 40. The name ''zirconium'' is taken from the name of the mineral zircon, the most important source of zirconium. The word is related to Persian '' zargun'' (zircon; ''zar-gun'', "gold-like" or "as gold"). It is a lustrous, grey-white, strong transition metal that closely resembles hafnium and, to a lesser extent, titanium. Zirconium is mainly used as a refractory and opacifier, although small amounts are used as an alloying agent for its strong resistance to corrosion. Zirconium forms a variety of inorganic and organometallic compounds such as zirconium dioxide and zirconocene dichloride, respectively. Five isotopes occur naturally, four of which are stable. Zirconium compounds have no known biological role. Characteristics Zirconium is a lustrous, greyish-white, soft, ductile, malleable metal that is solid at room temperature, though it is hard and brittle at lesser purities. In powder form, zirconium is highl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Felsic
In geology, felsic is a modifier describing igneous rocks that are relatively rich in elements that form feldspar and quartz.Marshak, Stephen, 2009, ''Essentials of Geology,'' W. W. Norton & Company, 3rd ed. It is contrasted with mafic rocks, which are relatively richer in magnesium and iron. Felsic refers to silicate minerals, magma, and rocks which are enriched in the lighter elements such as silicon, oxygen, aluminium, sodium, and potassium. Felsic magma or lava is higher in viscosity than mafic magma/lava. Felsic rocks are usually light in color and have specific gravities less than 3. The most common felsic rock is granite. Common felsic minerals include quartz, muscovite, orthoclase, and the sodium-rich plagioclase feldspars (albite-rich). Terminology In modern usage, the term ''acid rock'', although sometimes used as a synonym, normally now refers specifically to a high-silica-content (greater than 63% SiO2 by weight) volcanic rock, such as rhyolite. Older, broader usage ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |