|
Nitrosoureas
Nitrosourea is both the name of a molecule, and a class of compounds that include a nitroso (R-NO) group and a urea. Examples Examples include: * Arabinopyranosyl-''N''-methyl-''N''-nitrosourea (Aranose) * Carmustine (BCNU, BiCNU) * Chlorozotocin * Ethylnitrosourea (ENU) * Fotemustine * Lomustine (CCNU) * Nimustine * ''N''-Nitroso-''N''-methylurea (NMU) * Ranimustine (MCNU) * Semustine * Streptozocin (Streptozotocin) Nitrosourea compounds are DNA alkylating agents and are often used in chemotherapy. They are lipophilic and thus can cross the blood–brain barrier, making them useful in the treatment of brain tumors such as glioblastoma multiforme. File:Aranose (Haworth).svg, Arabinopyranosyl-''N''-methyl-''N''-nitrosourea File:Carmustine.svg, Carmustine File:Chlorozotocin (Haworth).svg, Chlorozotocin File:ENU.svg, Ethylnitrosourea File:Fotemustine.svg, Fotemustine File:Lomustine.svg, Lomustine File:N-Nitroso-N-methylurea.svg, ''N''-Nitroso-''N''-methylurea File:Nimust ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Arabinopyranosyl-N-methyl-N-nitrosourea
Arabinopyranosyl-''N''-methyl-''N''-nitrosourea, also known as Aranose (Араноза) is a cytostatic anticancer chemotherapeutic drug of an alkylating type. Chemically it is a nitrosourea derivative. It was developed in the Soviet Union in the 1970s. It was claimed by its developers that its advantages over other nitrosoureas are a relatively low hematological toxicity (compared to other nitrosoureas available at that time) and a wider therapeutic index, which allows for its outpatient administration. History It was first synthesized in late 1970s in the Laboratory of Organic Synthesis of Soviet Cancer Research Institute (which belonged to Academy of Medical Sciences of the USSR). Its first clinical trials in USSR were conducted in the late 1980s. Those trials confirmed its potential clinical efficacy in melanoma and better relative safety & improved tolerability over other nitrosourea antineoplastic compounds available at that time. In 1996 the compound obtained a Russia ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Semustine
Semustine (1- (2-Chloroethyl)-3-(trans-4-methylcyclohexyl)- 1-nitrosourea, MeCCNU) is an alkylating nitrosourea compound used in chemotherapy treatment of various types of tumours. Due to its lipophilic property, semustine can cross the blood-brain barrier for the chemotherapy of brain tumours, where it interferes with the cellular DNA of the highly dividing cells. Semustine, just as lomustine, is administered orally. It has been reported that semustine can perform the main mechanism of action at molecular level via the alkylation of DNA nitrogenous bases in duplex by results of inhibition of DNA replication, transcription, and translation by way of alkylation of DNA, under pH-dependent conditions by hydrolysis, forming chloro-carbonium ions. Sufficient evidence was found that treatment with semustine can cause acute leukaemia as a delayed effect in very rare cases. History During the past two decades, massive developments of various categories of anti-cancer drugs have been obs ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lomustine
Lomustine (INN); abbreviated as CCNU; original brand name CeeNU, now marketed as Gleostine) is an alkylating nitrosourea compound used in chemotherapy. It is closely related to semustine and is in the same family as streptozotocin. It is a highly lipid-soluble drug, thus it crosses the blood–brain barrier. This property makes it ideal for treating brain tumors, which is its primary use, although it is also used to treat Hodgkin lymphoma as a second-line option. Lomustine has a long time to nadir (the time when white blood cells reach their lowest number). Unlike carmustine, lomustine is administered orally. It is a bifunctional alkylating agent, alkylates both DNA and RNA, has the ability to created interstrand cross-links (ICLs) in DNA. As with other nitrosoureas, it may also inhibit several key enzymatic processes by carbamoylation of amino acids in proteins. Lomustine is cell-cycle nonspecific. It has also been used in veterinary practice as a treatment for mast cell tum ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemotherapy
Chemotherapy (often abbreviated to chemo and sometimes CTX or CTx) is a type of cancer treatment that uses one or more anti-cancer drugs (chemotherapeutic agents or alkylating agents) as part of a standardized chemotherapy regimen. Chemotherapy may be given with a curative intent (which almost always involves combinations of drugs) or it may aim to prolong life or to reduce symptoms ( palliative chemotherapy). Chemotherapy is one of the major categories of the medical discipline specifically devoted to pharmacotherapy for cancer, which is called ''medical oncology''. The term ''chemotherapy'' has come to connote non-specific usage of intracellular poisons to inhibit mitosis (cell division) or induce DNA damage, which is why inhibition of DNA repair can augment chemotherapy. The connotation of the word chemotherapy excludes more selective agents that block extracellular signals (signal transduction). The development of therapies with specific molecular or genetic targets, wh ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitroso
In organic chemistry, nitroso refers to a functional group in which the nitric oxide () group is attached to an organic moiety. As such, various nitroso groups can be categorized as ''C''-nitroso compounds (e.g., nitrosoalkanes; ), ''S''-nitroso compounds ( nitrosothiols; ), ''N''-nitroso compounds (e.g., nitrosamines, ), and ''O''-nitroso compounds ( alkyl nitrites; ). Synthesis Nitroso compounds can be prepared by the reduction of nitro compounds or by the oxidation of hydroxylamines. Ortho-nitrosophenols may be produced by the Baudisch reaction. In the Fischer–Hepp rearrangement aromatic 4-nitrosoanilines are prepared from the corresponding nitrosamines. Properties Nitrosoarenes typically participate in a monomer–dimer equilibrium. The dimers, which are often pale yellow, are often favored in the solid state, whereas the deep-green monomers are favored in dilute solution or at higher temperatures. They exist as ''cis'' and ''trans'' isomers. Due to the stability o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
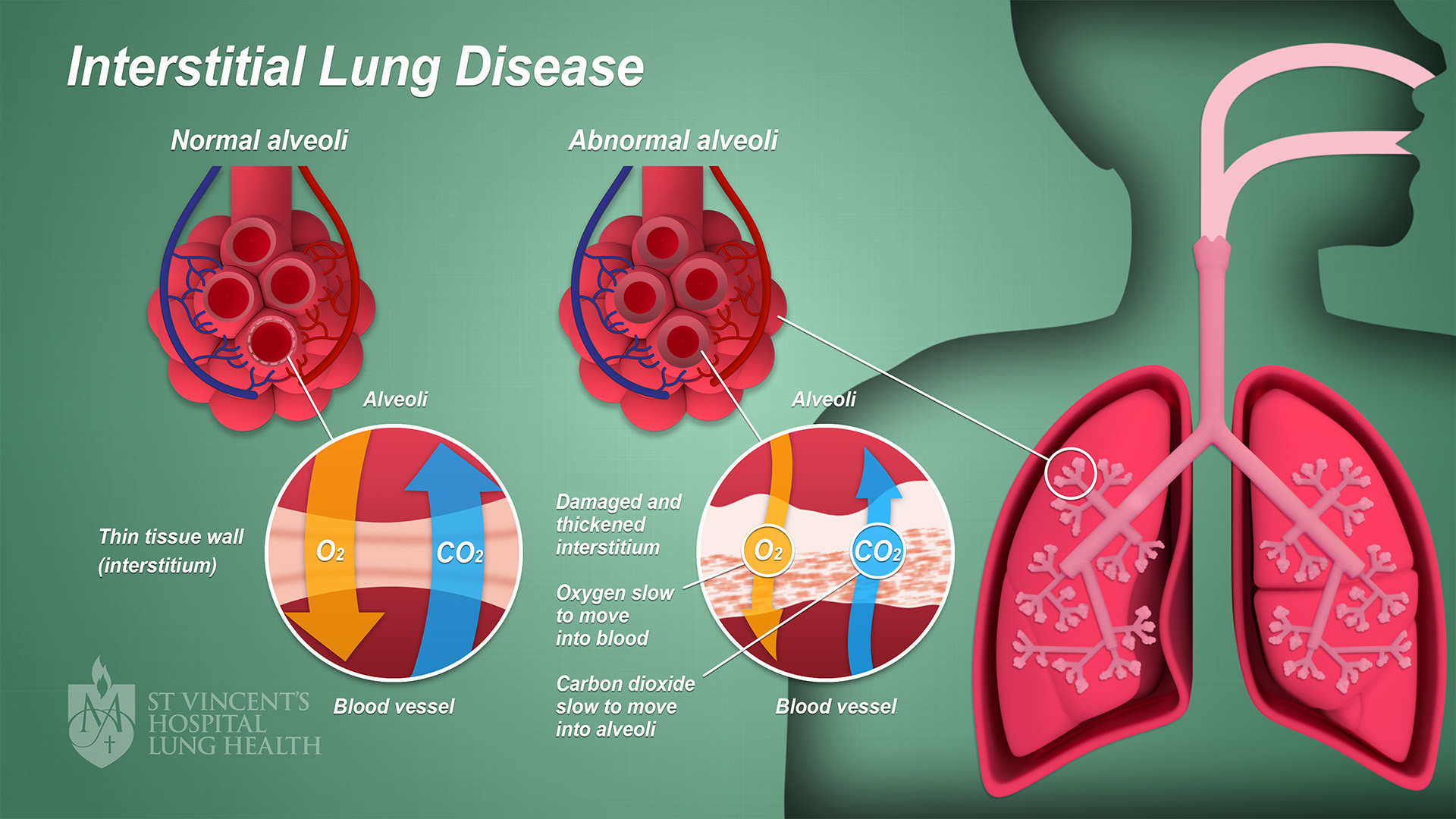
Interstitial Lung Disease
Interstitial lung disease (ILD), or diffuse parenchymal lung disease (DPLD), is a group of respiratory diseases affecting the interstitium (the tissue and space around the alveoli (air sacs)) of the lungs. It concerns alveolar epithelium, pulmonary capillary endothelium, basement membrane, and perivascular and perilymphatic tissues. It may occur when an injury to the lungs triggers an abnormal healing response. Ordinarily, the body generates just the right amount of tissue to repair damage, but in interstitial lung disease, the repair process is disrupted, and the tissue around the air sacs (alveoli) becomes scarred and thickened. This makes it more difficult for oxygen to pass into the bloodstream. The disease presents itself with the following symptoms: shortness of breath, nonproductive coughing, fatigue, and weight loss, which tend to develop slowly, over several months. The average rate of survival for someone with this disease is between three and five years. The term ILD i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glioblastoma Multiforme
Glioblastoma, previously known as glioblastoma multiforme (GBM), is one of the most aggressive types of cancer that begin within the brain. Initially, signs and symptoms of glioblastoma are nonspecific. They may include headaches, personality changes, nausea, and symptoms similar to those of a stroke. Symptoms often worsen rapidly and may progress to unconsciousness. The cause of most cases of glioblastoma is not known. Uncommon risk factors include genetic disorders, such as neurofibromatosis and Li–Fraumeni syndrome, and previous radiation therapy. Glioblastomas represent 15% of all brain tumors. They can either start from normal brain cells or develop from an existing low-grade astrocytoma. The diagnosis typically is made by a combination of a CT scan, MRI scan, and tissue biopsy. There is no known method of preventing the cancer. Treatment usually involves surgery, after which chemotherapy and radiation therapy are used. The medication temozolomide is frequently used ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Brain Tumor
A brain tumor occurs when abnormal cells form within the brain. There are two main types of tumors: malignant tumors and benign (non-cancerous) tumors. These can be further classified as primary tumors, which start within the brain, and secondary tumors, which most commonly have spread from tumors located outside the brain, known as brain metastasis tumors. All types of brain tumors may produce symptoms that vary depending on the size of the tumor and the part of the brain that is involved. Where symptoms exist, they may include headaches, seizures, problems with vision, vomiting and mental changes. Other symptoms may include difficulty walking, speaking, with sensations, or unconsciousness. The cause of most brain tumors is unknown. Uncommon risk factors include exposure to vinyl chloride, Epstein–Barr virus, ionizing radiation, and inherited syndromes such as neurofibromatosis, tuberous sclerosis, and von Hippel-Lindau Disease. Studies on mobile phone exposure hav ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Blood–brain Barrier
The blood–brain barrier (BBB) is a highly selective semipermeable membrane, semipermeable border of endothelium, endothelial cells that prevents solutes in the circulating blood from ''non-selectively'' crossing into the extracellular fluid of the central nervous system where neurons reside. The blood–brain barrier is formed by endothelial cells of the Capillary, capillary wall, astrocyte end-feet ensheathing the capillary, and pericytes embedded in the capillary basement membrane. This system allows the passage of some small molecules by passive transport, passive diffusion, as well as the selective and active transport of various nutrients, ions, organic anions, and macromolecules such as glucose and amino acids that are crucial to neural function. The blood–brain barrier restricts the passage of pathogens, the diffusion of solutes in the blood, and Molecular mass, large or Hydrophile, hydrophilic molecules into the cerebrospinal fluid, while allowing the diffusion of Hydr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lipophile
Lipophilicity (from Greek λίπος "fat" and φίλος "friendly"), refers to the ability of a chemical compound to dissolve in fats, oils, lipids, and non-polar solvents such as hexane or toluene. Such non-polar solvents are themselves lipophilic (translated as "fat-loving" or "fat-liking"), and the axiom that "like dissolves like" generally holds true. Thus lipophilic substances tend to dissolve in other lipophilic substances, but hydrophilic ("water-loving") substances tend to dissolve in water and other hydrophilic substances. Lipophilicity, hydrophobicity, and non-polarity may describe the same tendency towards participation in the London dispersion force, as the terms are often used interchangeably. However, the terms "lipophilic" and "hydrophobic" are not synonymous, as can be seen with silicones and fluorocarbons, which are hydrophobic but not lipophilic. __TOC__ Surfactants Hydrocarbon-based surfactants are compounds that are amphiphilic (or amphipathic), having a h ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkylating Antineoplastic Agent
An alkylating antineoplastic agent is an alkylating agent used in cancer treatment that attaches an alkyl group (CnH2n+1) to DNA. The alkyl group is attached to the guanine base of DNA, at the number 7 nitrogen atom of the purine ring. Since cancer cells, in general, proliferate faster and with less error-correcting than healthy cells, cancer cells are more sensitive to DNA damage—such as being alkylated. Alkylating agents are used to treat several cancers. However, they are also toxic to normal cells ( cytotoxic), particularly cells that divide frequently, such as those in the gastrointestinal tract, bone marrow, testicles and ovaries, which can cause loss of fertility. Most of the alkylating agents are also carcinogenic. History Before their use in chemotherapy, alkylating agents were better known for their use as sulfur mustard, ("mustard gas") and related chemical weapons in World War I. The nitrogen mustards were the first alkylating agents used medically, as well ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ranimustine
Ranimustine (INN, marketed under the tradename Cymerin; also known as MCNU) is a nitrosourea alkylating agent approved in Japan for the treatment of chronic myelogenous leukemia and polycythemia vera. It has never been filed for FDA evaluation in the United States The United States of America (U.S.A. or USA), commonly known as the United States (U.S. or US) or America, is a country primarily located in North America. It consists of 50 states, a federal district, five major unincorporated territori ..., where it is not marketed. References *Cymerin サイメリン(PDF) Mitsubishi Tanabe Pharma. October 2007. Alkylating antineoplastic agents Nitrosamines Nitrosoureas Ureas Chloroethyl compounds {{antineoplastic-drug-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |